Scalable Synthesis of Phosphorylated Indole-Isoquinolinones for Advanced Pharmaceutical Intermediates
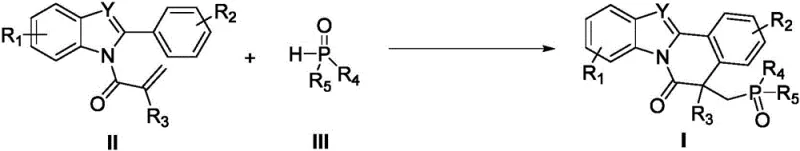
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies to construct complex polycyclic heterocycles, which serve as critical scaffolds for bioactive molecules and functional materials. A significant breakthrough in this domain is detailed in Chinese Patent CN110467640B, which discloses a novel preparation method for indole or benzimidazole-isoquinolinone fused heterocyclic derivatives. This technology addresses the growing demand for efficient synthetic routes to access phosphorus-containing heterocycles, a class of compounds increasingly valued in medicinal chemistry for their unique biological profiles and potential applications in luminescent materials. The core innovation lies in the utilization of an economical manganese promoter to facilitate a radical cascade cyclization, offering a distinct alternative to traditional precious metal-catalyzed processes.
For R&D directors and process chemists, the ability to introduce phosphorus motifs into rigid heterocyclic frameworks opens new avenues for structure-activity relationship (SAR) studies. The patent highlights a versatile approach that merges N-methacryloyl-2-phenylindole or benzimidazole precursors with P-H bond-containing compounds. This strategy not only constructs the isoquinolinone core but simultaneously installs a phosphorus functionality, effectively achieving two synthetic goals in a single operation. Such atom-economic transformations are highly desirable for streamlining the synthesis of advanced intermediates used in the development of next-generation therapeutics and agrochemicals.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art strategies for synthesizing indolo-isoquinoline or benzimidazolo-isoquinoline derivatives have often relied on sophisticated and costly catalytic systems that pose challenges for large-scale implementation. For instance, previous reports have utilized photocatalytic conditions involving free radical serial cyclization with aroyl chlorides, which necessitates specialized irradiation equipment and careful control of light intensity, complicating the scale-up process. Other methods have employed silver-catalyzed decarboxylative radical cyclization, which, while effective, relies on expensive noble metals that significantly inflate the raw material costs and introduce concerns regarding heavy metal residues in the final active pharmaceutical ingredients (APIs). Furthermore, many of these conventional routes exhibit limited substrate tolerance, often failing when electron-deficient or sterically hindered substrates are employed, thereby restricting the chemical space accessible to medicinal chemists.
The Novel Approach
In stark contrast, the methodology described in patent CN110467640B leverages manganese(III) acetate dihydrate [Mn(OAc)3·2H2O] as a cost-effective promoter to drive the reaction forward under mild thermal conditions. This novel approach eliminates the need for expensive noble metals or complex photochemical setups, utilizing simple heating at 60°C in acetic acid solvent. The reaction proceeds via a radical mechanism that tolerates a broad spectrum of functional groups, including halogens, cyano, nitro, and various alkoxy substituents on the aromatic rings. By shifting from precious metal catalysis to base metal promotion, this method drastically reduces the cost of goods sold (COGS) and simplifies the purification workflow, making it an ideal candidate for the commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into Mn-Promoted Radical Cyclization
The success of this transformation hinges on the unique redox properties of the manganese(III) species, which acts as a single-electron oxidant to initiate the radical cascade. The mechanism likely begins with the coordination of the manganese promoter to the P-H bond containing compound or the activation of the methacryloyl double bond, generating a carbon-centered radical species. This radical subsequently undergoes an intramolecular addition to the pendant phenyl ring, forming the new C-C bond that establishes the isoquinolinone skeleton. The resulting cyclohexadienyl radical is then oxidized by another equivalent of Mn(III) to restore aromaticity, followed by the trapping of the cationic intermediate or direct radical coupling with the phosphorus species. This intricate dance of electron transfer and bond formation occurs with high regioselectivity, ensuring that the phosphorus group is installed at the desired position on the newly formed ring system.
From an impurity control perspective, the use of Mn(OAc)3 offers a cleaner reaction profile compared to oxidative conditions that might lead to over-oxidation or polymerization of the methacryloyl moiety. The mild acidity of the acetic acid solvent helps to stabilize intermediates and suppresses side reactions such as hydrolysis of the amide bond, which is a common degradation pathway in basic or strongly acidic media. Moreover, the broad substrate scope demonstrated in the patent suggests that the radical intermediate is sufficiently robust to withstand electronic variations, meaning that electron-withdrawing groups like trifluoromethyl or cyano do not inhibit the cyclization efficiency. This mechanistic resilience is crucial for maintaining high purity standards required by regulatory bodies, as it minimizes the formation of difficult-to-remove structural analogs.
How to Synthesize Phosphorylated Indole-Isoquinolinones Efficiently
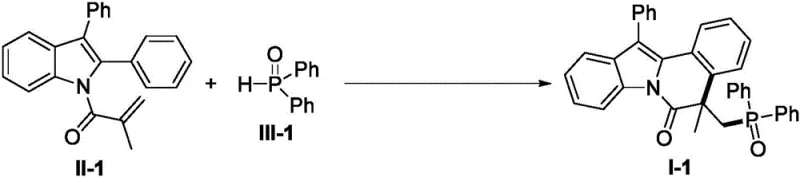
To implement this synthesis in a laboratory or pilot plant setting, operators should adhere to the optimized parameters established in the patent examples to ensure maximum yield and reproducibility. The process involves charging a closed-loop reactor, such as a Schlenk tube, with the specific N-methacryloyl-2-phenylindole substrate and the chosen phosphine oxide or phosphonate reagent. The stoichiometry is critical, with a molar ratio of approximately 1:2 for the substrate to phosphorus reagent, and 3 equivalents of the manganese promoter being preferred to drive the reaction to completion. The choice of solvent is also pivotal; while acetonitrile and DCE were tested, glacial acetic acid (HOAc) was identified as the superior medium, likely due to its ability to solubilize the manganese salt and participate in proton transfer steps during the radical termination phase.
- Charge a Schlenk reactor with N-methacryloyl-2-phenylindole substrate, P-H compound, and Mn(OAc)3·2H2O promoter in acetic acid solvent.
- Heat the reaction mixture to 60°C under an inert argon atmosphere and stir for 8 to 12 hours while monitoring progress via TLC.
- Concentrate the mixture under vacuum and purify the crude residue using silica gel column chromatography with n-hexane/ethyl acetate eluent.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this manganese-promoted protocol represents a strategic opportunity to optimize the sourcing of high-value heterocyclic intermediates. The primary driver for cost reduction is the substitution of expensive catalysts; replacing silver salts or specialized photocatalysts with commodity-grade manganese acetate results in substantial raw material savings. Additionally, the elimination of specialized equipment for photochemistry reduces capital expenditure (CAPEX) for manufacturing facilities, allowing existing standard reactors to be utilized for production. The simplicity of the work-up procedure, involving standard vacuum concentration and silica gel chromatography, further lowers operational expenditures (OPEX) by reducing processing time and solvent consumption compared to multi-step sequences required by older methods.
- Cost Reduction in Manufacturing: The economic impact of this process is profound due to the drastic reduction in catalyst costs. Manganese acetate is a bulk chemical available at a fraction of the price of silver or iridium-based catalysts, directly lowering the variable cost per kilogram of the produced intermediate. Furthermore, the high yields reported across a wide range of substrates mean less starting material is wasted, improving the overall mass balance and reducing the cost associated with raw material procurement and waste disposal. This efficiency translates into a more competitive pricing structure for the final API or functional material.
- Enhanced Supply Chain Reliability: Relying on widely available, non-proprietary reagents enhances supply chain security. Unlike specialized ligands or custom-synthesized photocatalysts that may have long lead times or single-source risks, manganese acetate and acetic acid are globally sourced commodities. This availability ensures that production schedules are not disrupted by raw material shortages. Additionally, the robustness of the reaction conditions allows for flexible manufacturing, enabling suppliers to respond quickly to fluctuating market demands without the need for complex process re-validation.
- Scalability and Environmental Compliance: The process is inherently scalable, moving seamlessly from gram-scale laboratory optimization to multi-kilogram pilot runs. The use of acetic acid, while requiring appropriate handling, is a well-understood solvent in the industry with established recovery and recycling protocols, minimizing environmental impact. The absence of heavy metals like silver simplifies the wastewater treatment process and reduces the burden of meeting stringent residual metal limits in pharmaceutical products, thereby accelerating regulatory approval timelines and ensuring compliance with green chemistry principles.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this manganese-promoted synthesis route. These insights are derived directly from the experimental data and scope analysis provided in the patent documentation, aiming to clarify the operational feasibility and strategic benefits for potential partners and licensees.
Q: What are the advantages of using Mn(OAc)3 over silver or photocatalysts for this synthesis?
A: The use of Mn(OAc)3·2H2O offers significant economic and operational advantages. Unlike expensive silver catalysts or complex photocatalytic systems requiring specialized light sources, manganese acetate is inexpensive, commercially abundant, and operates under mild thermal conditions (60°C), simplifying the equipment requirements and reducing overall production costs.
Q: Does this method support a wide range of substrates for diverse derivative synthesis?
A: Yes, the protocol demonstrates exceptional substrate universality. It tolerates various functional groups on both the indole/benzimidazole core (including halogens, cyano, nitro, and alkoxy groups) and the phosphine oxide component (aryl, alkyl, and ester substituents), allowing for the rapid generation of diverse chemical libraries for drug discovery.
Q: Is the reaction sensitive to atmospheric conditions?
A: While the reaction can proceed under air, optimal yields are achieved under an inert atmosphere, specifically argon. This ensures the controlled generation of radicals by the manganese promoter without interference from ambient oxygen, leading to consistent high purity and reproducibility essential for GMP manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Phosphorylated Indole-Isoquinolinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this manganese-catalyzed technology for the production of high-purity pharmaceutical intermediates. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from bench-scale discovery to industrial manufacturing is seamless. Our state-of-the-art facilities are equipped to handle radical chemistry safely and efficiently, and our rigorous QC labs enforce stringent purity specifications to guarantee that every batch meets the exacting standards required for global regulatory submissions.
We invite you to collaborate with us to leverage this cost-effective synthesis for your pipeline projects. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out today to obtain specific COA data for our catalog of heterocyclic building blocks and to discuss route feasibility assessments that could accelerate your time-to-market for novel therapeutic candidates.
