Scalable Palladium-Catalyzed Synthesis of Aryl-Substituted Triphenylene Compounds for Advanced Electronics
Scalable Palladium-Catalyzed Synthesis of Aryl-Substituted Triphenylene Compounds for Advanced Electronics
The rapidly evolving landscape of organic electronics demands robust, scalable, and cost-effective synthetic routes for high-performance materials. A significant breakthrough in this domain is detailed in patent CN109942361B, which discloses a highly efficient preparation method for aryl-substituted triphenylene compounds. These compounds serve as critical building blocks for advanced optoelectronic applications, including organic light-emitting diodes (OLEDs) and organic field-effect transistors (OFETs). The disclosed technology leverages a palladium-catalyzed cyclization strategy that operates under relatively mild thermal conditions, ranging from 70°C to 160°C, utilizing inexpensive inorganic bases and readily available organic solvents. This innovation addresses the longstanding challenges associated with constructing rigid polycyclic aromatic hydrocarbon skeletons, offering a pathway that is not only chemically elegant but also commercially viable for large-scale manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of symmetric bi-aromatic and polycyclic compounds has relied heavily on classical methodologies such as the Ullmann reaction, which often necessitates harsh reaction conditions, stoichiometric amounts of copper, and extended reaction times that can degrade sensitive functional groups. Furthermore, more modern approaches, such as those reported by the Itami group, typically involve a multi-step sequence where chlorinated polybiphenyls are first synthesized via Suzuki-Miyaura cross-coupling before undergoing a separate palladium-catalyzed cyclodimerization. While effective, these traditional pathways suffer from limited functional group compatibility and require complex derivatization steps that increase the overall cost of goods and extend the production lead time. The reliance on pre-functionalized substrates and multiple isolation steps introduces significant inefficiencies in material throughput and generates substantial chemical waste, posing challenges for both environmental compliance and economic feasibility in an industrial setting.
The Novel Approach
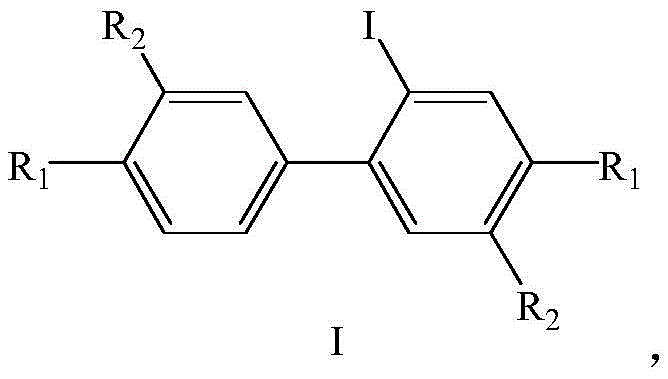
In stark contrast to these cumbersome legacy processes, the novel approach described in the patent utilizes a direct palladium-catalyzed intramolecular cyclization of 2-iodobiphenyl derivatives, designated as Formula I compounds. This streamlined methodology eliminates the need for pre-synthesizing complex polybiphenyl intermediates, allowing for the rapid construction of the triphenylene core in a single operational step. The versatility of this route is underscored by its tolerance for a wide array of substituents, including alkyl, alkoxy, haloalkoxy, fluorine, and chlorine groups, provided that at least one position remains a hydrogen atom to facilitate the cyclization event. By simplifying the synthetic sequence and utilizing cheap, commercially accessible starting materials, this method drastically reduces the complexity of unit operations and lowers the barrier for equipment requirements, making it an ideal candidate for the rapid and economical construction of triphenylene and fused ring compounds.
Mechanistic Insights into Pd-Catalyzed Cyclization and Scholl Reaction
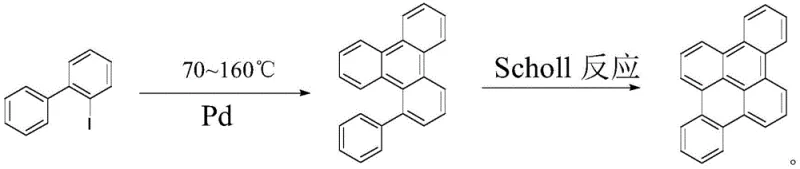
The core of this technological advancement lies in the efficient palladium catalytic cycle that drives the formation of the new carbon-carbon bonds necessary to close the triphenylene ring system. The mechanism likely initiates with the oxidative addition of the palladium catalyst into the carbon-iodine bond of the 2-iodobiphenyl substrate, generating a reactive aryl-palladium species. This intermediate subsequently undergoes a concerted metalation-deprotonation or electrophilic palladation at the ortho-position of the adjacent phenyl ring, facilitated by the presence of the inorganic base such as potassium bicarbonate. The final reductive elimination step releases the cyclized aryl-substituted triphenylene product and regenerates the active palladium catalyst, allowing the cycle to continue with high turnover numbers. This mechanistic pathway is remarkably robust, accommodating various electronic environments on the aromatic rings without compromising the integrity of the catalytic cycle or the yield of the final product.
Furthermore, the utility of these synthesized triphenylene compounds extends beyond their immediate application, as they serve as superior precursors for the construction of even larger fused ring systems via the Scholl reaction. As illustrated in the reaction scheme, the aryl-substituted triphenylene can undergo oxidative cyclodehydrogenation in the presence of Lewis acids like ferric chloride to form fully fused polycyclic aromatic hydrocarbons, such as dibenzo[fg,op]tetracene. This two-stage strategy—first establishing the triphenylene core through Pd-catalysis and then expanding the pi-system through Scholl chemistry—provides a modular and powerful platform for synthesizing graphene nanoribbons and other advanced carbon nanostructures. The ability to control the substitution pattern in the first step allows for precise tuning of the electronic and optical properties of the final fused materials, which is critical for optimizing performance in electronic devices.
How to Synthesize Aryl-Substituted Triphenylene Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires careful attention to reaction parameters to maximize yield and purity. The process begins by charging a dried reaction vessel, such as a Schlenk tube, with the palladium catalyst precursor, the 2-iodobiphenyl substrate, and the inorganic base under a protective atmosphere of argon or nitrogen to prevent catalyst deactivation by oxygen. An appropriate organic solvent, with N,N-dimethylaniline being the preferred choice due to its high boiling point and solvating power, is added to the mixture. The reaction is then heated to a temperature between 70°C and 160°C and stirred for a duration ranging from 24 to 96 hours, depending on the specific steric and electronic nature of the substituents. Upon completion, the solvent is removed under reduced pressure, and the crude product is purified using standard silica gel column chromatography to afford the target triphenylene compound as a high-purity white solid.
- Charge a reaction vessel with palladium catalyst precursor, 2-iodobiphenyl derivative substrate, and inorganic base under inert gas protection.
- Add organic solvent such as N,N-dimethylaniline and heat the mixture to a temperature range of 70-160°C.
- Maintain stirring for 24-96 hours, then cool, remove solvent, and purify the resulting white solid via silica gel chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, this patented methodology offers transformative advantages that directly impact the bottom line and operational reliability. The shift from multi-step, resource-intensive syntheses to a direct cyclization process significantly simplifies the manufacturing workflow, reducing the number of unit operations and the associated labor and energy costs. By eliminating the need for complex pre-functionalized intermediates and expensive transition metal removal steps often associated with cross-coupling reactions, the overall cost of manufacturing is substantially decreased. This efficiency gain allows for a more competitive pricing structure for high-purity electronic chemical intermediates, enabling downstream manufacturers to optimize their own production costs without sacrificing material quality or performance specifications.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven by the use of cheap and easily obtainable raw materials, such as simple 2-iodobiphenyl derivatives, which are widely available in the global chemical market. The elimination of expensive ligands and the use of catalytic rather than stoichiometric amounts of palladium further contribute to significant cost savings. Additionally, the simplified workup procedure, which involves basic solvent evaporation and chromatography, reduces the consumption of auxiliary chemicals and minimizes waste disposal costs, leading to a leaner and more cost-effective production model for aryl-substituted triphenylene compounds.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the robustness of the reaction conditions and the accessibility of the starting materials. Since the process does not rely on exotic or hard-to-source reagents, the risk of supply disruptions is minimized, ensuring a continuous flow of materials for production schedules. The moderate temperature range and the use of common solvents mean that the process can be easily transferred between different manufacturing sites or scaled up using standard reactor configurations, providing flexibility and security for long-term supply agreements with key partners in the electronics industry.
- Scalability and Environmental Compliance: The simplicity of the unit operations and the low equipment requirements make this process highly scalable from gram-scale laboratory synthesis to multi-ton commercial production. The reduced generation of hazardous waste and the avoidance of harsh reagents align with increasingly stringent environmental regulations and corporate sustainability goals. This green chemistry profile not only facilitates regulatory approval but also enhances the brand value of the final products, appealing to environmentally conscious consumers and stakeholders in the high-tech sector.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this synthesis technology. These answers are derived directly from the experimental data and technical disclosures within the patent documentation, providing a reliable foundation for decision-making. Understanding these details is crucial for R&D teams evaluating process feasibility and procurement officers assessing supplier capabilities for critical electronic materials.
Q: What are the primary advantages of this Pd-catalyzed method over traditional Ullmann coupling?
A: This method operates under milder conditions (70-160°C) compared to classical Ullmann reactions and avoids the multi-step complexity of prior Suzuki-coupling strategies, resulting in higher atom economy and simplified purification.
Q: Can this synthesis route be scaled for commercial production of OLED intermediates?
A: Yes, the process utilizes cheap and easily available raw materials with simple unit operations and low equipment requirements, making it highly suitable for rapid construction and commercial scale-up of triphenylene derivatives.
Q: What subsequent reactions can be performed on the synthesized triphenylene compounds?
A: The resulting aryl-substituted triphenylene compounds serve as excellent precursors for further Scholl reactions to construct fully fused polycyclic aromatic hydrocarbons and graphene nanoribbons.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Aryl-Substituted Triphenylene Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development of next-generation electronic materials. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. We are committed to delivering aryl-substituted triphenylene compounds that meet stringent purity specifications, supported by our rigorous QC labs and state-of-the-art analytical capabilities. Our expertise in palladium-catalyzed transformations allows us to optimize this specific patent-protected route for maximum yield and minimal impurity profiles, guaranteeing a consistent supply of premium materials for your OLED and semiconductor applications.
We invite you to collaborate with us to leverage this advanced synthetic technology for your specific project needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how our optimized processes can reduce your overall material costs. Please contact us today to request specific COA data for our triphenylene inventory and to discuss route feasibility assessments for your custom synthesis projects, ensuring that your supply chain is built on a foundation of technical excellence and commercial reliability.
