Advanced Electrochemical Manufacturing of Fluorinated Indole Pharmaceutical Intermediates
Introduction to Electrochemical Fluorination Technology
The pharmaceutical industry is constantly seeking innovative synthetic methodologies to access complex fluorinated scaffolds, which are pivotal in modern drug design for enhancing metabolic stability and bioavailability. Patent CN111170924B introduces a groundbreaking electrochemical approach for synthesizing hexafluoroisopropoxyindole compounds, addressing the long-standing challenges associated with introducing fluorine-containing substituents into heterocyclic systems. This technology leverages electricity as a clean reagent to drive the oxidative coupling of indoles with hexafluoroisopropanol (HFIP), bypassing the need for toxic stoichiometric oxidants. For R&D directors and procurement specialists, this represents a significant shift towards greener, more sustainable manufacturing processes that align with global regulatory trends. The method operates under remarkably mild conditions, utilizing simple undivided cells and commercially available electrode materials, making it highly attractive for both laboratory-scale optimization and eventual industrial deployment. By enabling the direct functionalization of the indole core, this patent opens new avenues for generating diverse libraries of bioactive molecules with potential applications in oncology and beyond.
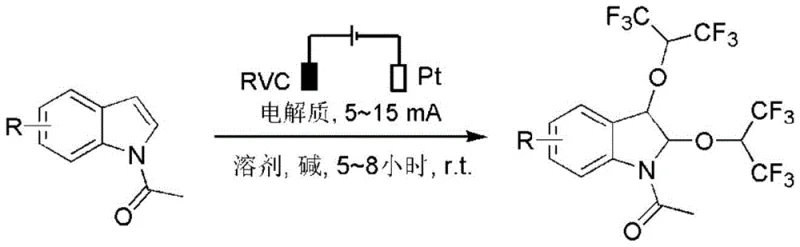
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for installing alkoxy groups onto indole rings often rely on harsh chemical oxidants such as hypervalent iodine reagents, lead tetraacetate, or transition metal catalysts which pose significant environmental and safety hazards. These conventional methods frequently require strict anhydrous conditions, elevated temperatures, and complex workup procedures to remove heavy metal residues, leading to increased production costs and lower overall atom economy. Furthermore, the selective introduction of bulky fluorinated groups like hexafluoroisopropoxy is particularly challenging due to steric hindrance and the potential for over-oxidation or polymerization of the sensitive indole nucleus. The reliance on stoichiometric amounts of expensive oxidants not only drives up the raw material costs but also generates substantial quantities of chemical waste, creating a burden on waste management systems and complicating regulatory compliance for pharmaceutical manufacturers. Consequently, there is a critical need for alternative strategies that can achieve these transformations with higher efficiency and reduced environmental impact.
The Novel Approach
The electrochemical method disclosed in the patent offers a transformative solution by replacing chemical oxidants with electrons, thereby fundamentally changing the reaction paradigm. In this novel approach, the indole substrate undergoes anodic oxidation to generate a reactive radical cation intermediate, which is subsequently trapped by hexafluoroisopropanol to form the desired C-O bonds. This process is conducted in an undivided cell using a Reticulated Vitreous Carbon (RVC) anode and a Platinum cathode, ensuring robust performance and longevity of the equipment. The reaction proceeds smoothly at room temperature with constant current electrolysis, typically requiring only 5 to 8 hours to reach completion. By utilizing electricity as the primary driving force, the method drastically reduces the consumption of chemical reagents and minimizes the generation of hazardous byproducts. This streamlined protocol not only simplifies the operational workflow but also enhances the safety profile of the synthesis, making it an ideal candidate for scale-up in a GMP environment where process safety and purity are paramount concerns for supply chain stakeholders.
Mechanistic Insights into Electrochemical Oxidative Functionalization
The core mechanism involves the anodic oxidation of the N-acetylindole substrate, where the removal of an electron generates a radical cation species that is delocalized across the indole ring system. This electrophilic intermediate is highly susceptible to nucleophilic attack by the oxygen atom of hexafluoroisopropanol, leading to the formation of a C-O bond at the C3 position initially, followed by further oxidation and nucleophilic addition at the C2 position to yield the 2,3-disubstituted product. The choice of electrolyte, such as tetrabutylammonium iodide or hexafluorophosphate, plays a crucial role in maintaining conductivity and stabilizing the charged intermediates without interfering with the reaction pathway. The use of a base like potassium carbonate or cesium carbonate helps to deprotonate the alcohol nucleophile, enhancing its reactivity towards the electro-generated cation. This precise control over the oxidation potential allows for high chemoselectivity, preventing the degradation of sensitive functional groups that might be present on the indole scaffold. Understanding this mechanistic nuance is essential for process chemists aiming to optimize reaction parameters such as current density and electrode spacing to maximize yield and minimize side reactions during large-scale production.
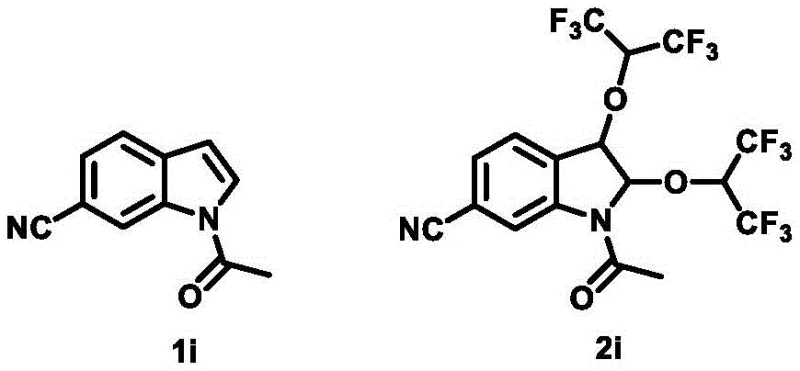
Furthermore, the structural integrity of the final product is maintained through the mild nature of the electrochemical conditions, which avoids the thermal stress often associated with traditional heating methods. The patent highlights the successful synthesis of various derivatives, including those with electron-withdrawing groups like cyano or ester functionalities, demonstrating the broad substrate scope of this methodology. For instance, the conversion of N-acetyl-6-cyanoindole to the corresponding hexafluoroisopropoxy derivative (compound 2i) showcases the tolerance of the system towards nitrile groups, which are common motifs in medicinal chemistry. The ability to introduce two hexafluoroisopropoxy groups simultaneously creates a unique steric and electronic environment around the indole core, which has been shown to significantly enhance biological activity. This mechanistic robustness ensures that the process can be reliably transferred from benchtop to pilot plant, providing a consistent supply of high-quality intermediates for downstream drug development programs.
How to Synthesize Hexafluoroisopropoxyindole Efficiently
To implement this electrochemical synthesis effectively, operators must adhere to specific procedural guidelines regarding cell setup and reaction monitoring to ensure reproducibility and safety. The process begins with the careful preparation of the electrolyte solution, ensuring that the concentration of the supporting salt is sufficient to maintain low cell resistance and uniform current distribution across the RVC anode surface. Continuous stirring is vital to prevent concentration polarization and to facilitate the mass transport of the substrate to the electrode interface. Reaction progress should be monitored regularly using thin-layer chromatography (TLC) to determine the optimal endpoint, preventing over-electrolysis which could lead to decomposition. Once the reaction is deemed complete, the workup procedure involves standard extraction and drying techniques, followed by purification via column chromatography to isolate the pure product. Detailed standardized synthesis steps are provided in the guide below to assist technical teams in replicating these results accurately.
- Prepare the electrolytic cell by placing 0.5 mmol of N-acetylindole derivative, 1.5 equivalents of base (e.g., K2CO3), and 0.5 equivalents of electrolyte (e.g., n-Bu4NI) into a 10 mL three-neck flask with 7 mL of solvent.
- Insert a Reticulated Vitreous Carbon (RVC) anode and a Platinum (Pt) cathode, then apply a constant current of 5-15 mA at room temperature for 5-8 hours while monitoring via TLC.
- Upon completion, extract the mixture with ethyl acetate, dry the organic layer over anhydrous sodium sulfate, and purify the residue via silica gel column chromatography to isolate the target hexafluoroisopropoxyindole.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this electrochemical technology offers substantial strategic benefits for procurement managers and supply chain directors looking to optimize their manufacturing networks. The elimination of expensive and hazardous chemical oxidants translates directly into reduced raw material costs and lower expenses associated with waste disposal and environmental compliance. By relying on electricity, which is generally cheaper and more readily available than specialized reagents, manufacturers can achieve significant cost reductions in API manufacturing without compromising on product quality. Moreover, the simplicity of the reactor design, which does not require high-pressure vessels or complex temperature control systems, lowers the capital expenditure required for setting up production lines. This accessibility makes the technology scalable for both small-batch custom synthesis and large-volume commercial production, providing flexibility to respond to market demands efficiently.
- Cost Reduction in Manufacturing: The primary economic driver of this technology is the substitution of stoichiometric chemical oxidants with electrons, which effectively removes a major cost center from the bill of materials. Traditional oxidation methods often involve costly reagents that are consumed in equimolar amounts, whereas electricity is a utility that can be precisely controlled and optimized for energy efficiency. Additionally, the reduction in waste generation means lower costs for wastewater treatment and hazardous waste disposal, contributing to a leaner and more sustainable operation. The use of durable electrode materials like RVC and Platinum further extends the lifespan of the equipment, reducing the frequency of replacement and maintenance downtime. These factors combined result in a more cost-effective production model that enhances the overall profitability of manufacturing fluorinated pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: Implementing this electrochemical route mitigates supply chain risks associated with the sourcing of specialized chemical reagents that may be subject to market volatility or regulatory restrictions. Hexafluoroisopropanol and common indole derivatives are widely available commodity chemicals, ensuring a stable and continuous supply of starting materials. The robustness of the electrochemical process also means that production is less susceptible to disruptions caused by the unavailability of specific catalysts or oxidants. This reliability is crucial for maintaining consistent delivery schedules to downstream customers and avoiding production delays that can impact the entire drug development timeline. By diversifying the synthetic toolkit with electrochemical methods, companies can build more resilient supply chains capable of withstanding external shocks and ensuring business continuity.
- Scalability and Environmental Compliance: The inherent safety and modularity of electrochemical reactors make them highly suitable for scaling up from laboratory to industrial scales. Unlike batch processes that face heat and mass transfer limitations upon scaling, electrochemical flow cells or larger tank reactors can be designed to maintain efficient electron transfer rates. This scalability ensures that the process can meet increasing demand without the need for extensive re-engineering. Furthermore, the green chemistry credentials of this method, characterized by minimal waste and the absence of heavy metals, align perfectly with stringent environmental regulations and corporate sustainability goals. This compliance reduces the regulatory burden and enhances the company's reputation as a responsible manufacturer, which is increasingly important for securing contracts with major pharmaceutical partners who prioritize ethical and sustainable sourcing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the electrochemical synthesis of hexafluoroisopropoxyindoles, providing clarity for stakeholders evaluating this technology for adoption. These answers are derived directly from the experimental data and technical specifications outlined in the patent documentation, ensuring accuracy and relevance for decision-makers. Understanding these aspects is critical for assessing the feasibility of integrating this method into existing production workflows and for anticipating potential challenges during process validation. We encourage technical teams to review these points carefully to gain a comprehensive understanding of the operational requirements and benefits.
Q: What are the key advantages of using electrochemical synthesis for fluorinated indoles?
A: The electrochemical method described in CN111170924B eliminates the need for harsh chemical oxidants and heavy metal catalysts. It operates under mild conditions (room temperature, constant current), significantly reducing waste generation and simplifying the purification process compared to traditional stoichiometric oxidation methods.
Q: Which electrode materials are critical for this transformation?
A: The patent specifies the use of Reticulated Vitreous Carbon (RVC) as the anode and Platinum (Pt) as the cathode. RVC is crucial due to its high surface area and chemical inertness, which facilitates efficient electron transfer and prevents electrode degradation during the oxidative functionalization of the indole ring.
Q: Does the introduction of hexafluoroisopropoxy groups enhance biological activity?
A: Yes, the patent data indicates that introducing hexafluoroisopropoxy groups significantly improves antitumor activity. Specifically, compound 2i demonstrated superior inhibitory effects against HeLa and T-24 cancer cell lines compared to the starting material and even outperformed the standard drug 5-FU in certain assays.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Hexafluoroisopropoxyindole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of electrochemical synthesis in advancing the production of high-value pharmaceutical intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative technologies like the one described in CN111170924B can be seamlessly transitioned from the lab to the factory floor. Our state-of-the-art facilities are equipped with advanced electrochemical reactors and rigorous QC labs capable of meeting stringent purity specifications required for clinical and commercial supplies. We are committed to delivering high-purity fluorinated indoles that meet the exacting standards of the global pharmaceutical industry, supporting our clients in bringing life-saving therapies to market faster and more efficiently.
We invite procurement leaders and R&D directors to collaborate with us to explore how this electrochemical route can optimize your supply chain and reduce manufacturing costs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific project needs, demonstrating the tangible economic benefits of switching to this greener synthetic method. Please contact us to request specific COA data for our fluorinated indole portfolio and to discuss route feasibility assessments for your target molecules. Together, we can drive innovation and efficiency in the synthesis of complex heterocyclic intermediates, fostering a partnership built on technical excellence and mutual success.
