Advanced Nickel-Catalyzed Synthesis of Monofluoroalkyl Intermediates for Pharmaceutical Manufacturing
The introduction of fluorine atoms into organic molecules is a cornerstone strategy in modern medicinal chemistry, profoundly influencing the metabolic stability, lipophilicity, and bioavailability of drug candidates. As the demand for fluorinated active pharmaceutical ingredients (APIs) continues to surge, the development of efficient, scalable, and cost-effective synthetic methodologies has become a critical priority for the global fine chemical industry. Patent CN106278847B discloses a groundbreaking preparation method for monofluoroalkyl-containing compounds, addressing long-standing challenges in the field. This technology leverages a nickel-catalyzed Suzuki coupling reaction to introduce monofluoroalkyl groups in a single step, offering a robust alternative to traditional methods that often suffer from harsh conditions and limited substrate scope. For R&D directors and procurement managers seeking reliable pharmaceutical intermediate supplier solutions, this innovation represents a significant leap forward in process efficiency and economic viability.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of fluoroalkyl-substituted aromatic compounds has relied heavily on transition metal-catalyzed cross-coupling reactions, predominantly utilizing palladium catalysts. While effective in certain contexts, these conventional approaches are plagued by substantial drawbacks that hinder their widespread industrial adoption. A primary limitation is the economic burden imposed by the high cost of palladium catalysts, which significantly inflates the production costs of high-purity OLED material and pharmaceutical precursors. Furthermore, many existing protocols require drastic reaction conditions, such as elevated temperatures or the use of strong bases, which can lead to the decomposition of sensitive functional groups. Critically, some prior art methods necessitate the use of excessive amounts of coupling partners; for instance, certain palladium-catalyzed reactions require up to 40-fold equivalents of arylboronic esters to achieve acceptable conversion, resulting in poor atom economy and generating substantial chemical waste. These inefficiencies create bottlenecks in cost reduction in electronic chemical manufacturing and complicate the supply chain for complex intermediates.
The Novel Approach
In stark contrast, the methodology described in patent CN106278847B utilizes an earth-abundant nickel catalyst system to drive the Suzuki coupling between aryl boronic acids and fluoroalkyl halides. This novel approach eliminates the need for expensive noble metals, thereby offering a pathway for substantial cost savings in large-scale production. The reaction operates under remarkably mild conditions, typically proceeding at temperatures between 70°C and 80°C, which preserves the integrity of delicate functional groups such as aldehydes, ketones, and esters. The protocol demonstrates exceptional broad-spectrum capability, accommodating a wide array of substrates including substituted phenyl, naphthyl, and heteroaryl boronic acids. By achieving high conversion rates and isolated yields often exceeding 70-90% with only catalytic amounts of nickel (5-10 mol%), this method streamlines the commercial scale-up of complex polymer additives and drug intermediates. The simplicity of the post-processing operation further enhances its appeal, allowing for straightforward isolation of the target monofluoroalkyl compounds without the need for cumbersome purification steps.
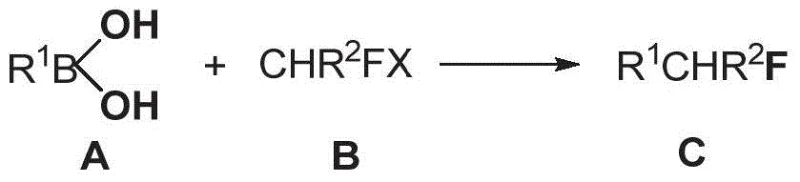
Mechanistic Insights into Nickel-Catalyzed Suzuki Coupling
The success of this transformation lies in the intricate interplay between the nickel catalyst, the nitrogen-containing ligands, and the specific additives employed. The catalytic cycle likely initiates with the oxidative addition of the fluoroalkyl halide to the low-valent nickel species, forming a key organonickel intermediate. This step is facilitated by the presence of bidentate nitrogen ligands such as 1,10-phenanthroline or substituted bipyridines, which stabilize the active nickel center and prevent the formation of inactive nickel black. Subsequent transmetallation with the aryl boronic acid, activated by the base (typically potassium carbonate), transfers the aryl group to the nickel center. The final reductive elimination step releases the desired monofluoroalkyl product and regenerates the active catalyst. A crucial aspect of this mechanism is the inclusion of additives like 4-dimethylaminopyridine (DMAP), which plays a pivotal role in modulating the electronic environment of the catalyst and suppressing side reactions. This precise control over the catalytic cycle ensures high selectivity and minimizes the formation of homocoupling byproducts or defluorinated impurities, which is essential for meeting the stringent purity specifications required in the life sciences sector.
Impurity control is another critical dimension where this nickel-catalyzed system excels. The mild reaction conditions prevent the degradation of sensitive moieties, such as the fluoroalkyl group itself, which can be prone to elimination under harsher basic or thermal conditions. The compatibility with diverse functional groups means that complex molecules can be synthesized without extensive protecting group manipulation, reducing the overall step count and potential points of failure. Analytical data from the patent examples confirms that the resulting products consistently achieve purity levels greater than 95% as determined by NMR spectroscopy. This high level of chemical fidelity is paramount for reducing lead time for high-purity pharmaceutical intermediates, as it minimizes the need for resource-intensive recrystallization or chromatographic purification. The robustness of the catalyst system against moisture and oxygen, relative to other sensitive organometallic processes, further contributes to a cleaner reaction profile and more predictable outcomes in a manufacturing setting.
How to Synthesize Monofluoroalkyl Compounds Efficiently
The practical implementation of this synthesis route is designed for ease of operation and scalability, making it accessible for both laboratory research and industrial production. The process begins with the charging of the aryl boronic acid substrate, the nickel catalyst precursor (such as NiCl2·DME), the selected ligand, and the DMAP additive into a reaction vessel containing a mixed solvent system of dimethoxyethane (DME) and 1,4-dioxane. Following the addition of the inorganic base, the fluoroalkyl halide coupling partner is introduced, and the mixture is heated to the optimal temperature range. The reaction progress is monitored using standard analytical techniques until completion. Detailed standardized synthesis steps are provided in the guide below to ensure reproducibility and safety during execution.
- Prepare the reaction mixture by combining the aryl boronic acid substrate, nickel catalyst (e.g., NiCl2·DME), nitrogen-containing ligand (e.g., phenanthroline), and additive (e.g., DMAP) in a suitable solvent system like DME/dioxane.
- Add the base (e.g., K2CO3) and the fluoroalkyl halide coupling partner to the reaction vessel under inert atmosphere to prevent catalyst oxidation.
- Heat the reaction mixture to 70°C-80°C and stir for 12-24 hours until the starting material is consumed, followed by standard aqueous workup and purification to isolate the high-purity product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this nickel-catalyzed technology offers transformative benefits that extend beyond mere chemical efficiency. The shift from precious metal catalysts to base metal nickel directly addresses the volatility of raw material costs, providing a more stable and predictable pricing structure for long-term contracts. The elimination of expensive palladium not only reduces the direct cost of goods sold but also simplifies the regulatory burden associated with heavy metal residue limits in final drug substances. This aligns perfectly with the industry's drive for cost reduction in pharmaceutical intermediate manufacturing, allowing companies to allocate resources more effectively towards innovation and market expansion. Furthermore, the high atom economy and reduced waste generation contribute to a greener manufacturing footprint, supporting corporate sustainability goals and environmental compliance standards.
- Cost Reduction in Manufacturing: The replacement of palladium with nickel catalysts results in a drastic reduction in catalyst costs, as nickel is significantly more abundant and affordable. Additionally, the low catalyst loading (5-10 mol%) and the ability to use stoichiometric amounts of boronic acids rather than large excesses minimize raw material consumption. This efficiency translates into substantial cost savings across the entire production lifecycle, enhancing the overall profit margin for high-volume chemical products without compromising on quality or performance metrics.
- Enhanced Supply Chain Reliability: The reliance on commercially available and stable reagents ensures a resilient supply chain that is less susceptible to geopolitical disruptions or market shortages often associated with rare earth metals. The robustness of the reaction conditions allows for flexible manufacturing schedules and reduces the risk of batch failures due to sensitive operational parameters. This reliability is crucial for maintaining continuous production flows and meeting tight delivery deadlines for critical API intermediates, thereby strengthening the partnership between suppliers and pharmaceutical manufacturers.
- Scalability and Environmental Compliance: The process has been successfully demonstrated at gram scales with consistent results, indicating a clear path for commercial scale-up to multi-kilogram or ton quantities. The mild reaction temperatures and the use of relatively benign solvents simplify the engineering requirements for large-scale reactors, reducing capital expenditure on specialized equipment. Moreover, the avoidance of highly toxic reagents and the generation of less hazardous waste streamline the environmental permitting process and reduce the costs associated with waste disposal, ensuring full compliance with increasingly stringent global environmental regulations.
Frequently Asked Questions (FAQ)
Understanding the technical nuances and commercial implications of this synthesis method is vital for stakeholders evaluating its integration into their supply chains. The following questions address common inquiries regarding the catalyst system, substrate scope, and scalability, drawing directly from the experimental data and technical specifications outlined in the patent documentation. These insights are intended to clarify the operational advantages and assist in decision-making processes for sourcing and production planning.
Q: What are the primary advantages of using nickel catalysts over palladium for fluorination?
A: Nickel catalysts offer significant cost advantages due to the lower price of nickel compared to palladium. Furthermore, the patented nickel system demonstrates superior functional group tolerance and operates under milder conditions, avoiding the need for excessive equivalents of boronic esters often required in traditional palladium methods.
Q: Is this synthesis method scalable for industrial production?
A: Yes, the method has been validated at gram-scale levels with consistent high yields and purity. The use of commercially available reagents, mild temperatures (70°C-80°C), and simple workup procedures makes it highly suitable for commercial scale-up of complex pharmaceutical intermediates.
Q: What types of functional groups are compatible with this reaction?
A: The reaction exhibits broad substrate scope, tolerating diverse functional groups including esters, ketones, aldehydes, nitriles, sulfones, and halogens. This compatibility allows for the late-stage functionalization of complex drug molecules without extensive protecting group strategies.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Monofluoroalkyl Compounds Supplier
As the pharmaceutical and agrochemical industries continue to evolve, the demand for specialized fluorinated intermediates remains at an all-time high. NINGBO INNO PHARMCHEM stands at the forefront of this sector, leveraging advanced technologies like the nickel-catalyzed Suzuki coupling to deliver superior value to our global partners. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements with precision and consistency. We are committed to maintaining stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of monofluoroalkyl compounds meets the highest industry standards for safety and efficacy.
We invite you to explore how our innovative manufacturing capabilities can optimize your supply chain and reduce your overall production costs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific project needs. We are ready to provide specific COA data and comprehensive route feasibility assessments to support your R&D and commercialization efforts. Let us be your trusted partner in navigating the complexities of fine chemical synthesis and delivering high-quality solutions for the global market.
