Optimized Industrial Synthesis of p-Methylsulfonyl Benzaldehyde for High-Purity Pharmaceutical Intermediates
Optimized Industrial Synthesis of p-Methylsulfonyl Benzaldehyde for High-Purity Pharmaceutical Intermediates
The production of high-value pharmaceutical intermediates requires robust, scalable, and economically viable synthetic pathways that minimize environmental impact while maximizing yield. Patent CN102675167B introduces a significant advancement in the preparation of p-methylsulfonyl benzaldehyde, a critical precursor for the broad-spectrum antibiotic thiamphenicol and its derivatives. This innovative methodology departs from conventional, labor-intensive routes by utilizing p-chlorobenzaldehyde as a readily available starting material, thereby streamlining the manufacturing process into two highly efficient stages. The first stage employs a phase-transfer catalyzed nucleophilic substitution to install the methylthio group, while the second stage utilizes a controlled catalytic oxidation to achieve the desired sulfone functionality. For global procurement teams and R&D directors seeking a reliable pharmaceutical intermediate supplier, this technology represents a paradigm shift towards greener chemistry and superior cost-efficiency in API manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of p-methylsulfonyl benzaldehyde has been plagued by complex multi-step sequences that originate from toluene. These legacy processes typically necessitate harsh reaction conditions, including chlorosulfonation, reduction, methylation, bromination, and hydrolysis, each step introducing potential yield losses and generating substantial hazardous waste. The cumulative effect of these sequential transformations results in a low overall plant factor and high unit consumption of raw materials, making the process economically unattractive for large-scale commercial production. Furthermore, alternative methods reported in literature, such as those starting from p-toluenesulfonyl chloride or utilizing potassium hydrogen persulfate as an oxidant, have demonstrated disappointing yields ranging from merely 21% to 53%. These inefficient routes not only inflate the cost of goods sold but also impose severe environmental protection pressures due to the generation of toxic by-products and the requirement for extensive purification protocols that are difficult to manage in a continuous manufacturing setting.
The Novel Approach
In stark contrast to these cumbersome traditional methodologies, the novel approach detailed in the patent leverages the inherent reactivity of p-chlorobenzaldehyde to construct the target molecule with remarkable efficiency. By initiating the synthesis with a direct nucleophilic displacement of the chlorine atom using sodium methyl mercaptide, the process bypasses the need for multiple functional group interconversions required in the toluene route. This strategic shortcut drastically reduces the number of unit operations, thereby lowering energy consumption and simplifying equipment requirements. The subsequent oxidation step is equally refined, employing hydrogen peroxide as a clean oxidant in the presence of specific catalysts to selectively convert the sulfide to the sulfone without compromising the integrity of the aldehyde group. This streamlined two-step sequence not only enhances the overall throughput but also aligns perfectly with modern green chemistry principles, offering a compelling value proposition for cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into Phase-Transfer Catalyzed Substitution and Oxidation
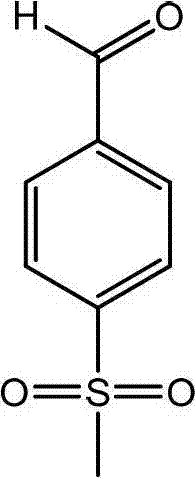
The success of this synthetic route hinges on the precise control of reaction kinetics and selectivity in both transformation steps. In the initial substitution phase, the reaction between p-chlorobenzaldehyde and sodium methyl mercaptide occurs in a biphasic system, where the organic aldehyde and the aqueous thiolate salt have limited mutual solubility. The addition of a phase-transfer catalyst, such as tetrabutylammonium bromide or iodide, facilitates the transport of the nucleophilic methyl mercaptide anion into the organic phase, significantly accelerating the reaction rate at moderate temperatures of 30 to 50°C. This mechanistic advantage ensures complete conversion of the starting material while minimizing side reactions, a critical factor for maintaining high purity in the crude intermediate. The optimization of molar ratios, specifically maintaining a slight excess of the thiolate reagent, drives the equilibrium towards the formation of p-methylthio benzaldehyde, setting a solid foundation for the subsequent oxidation step.
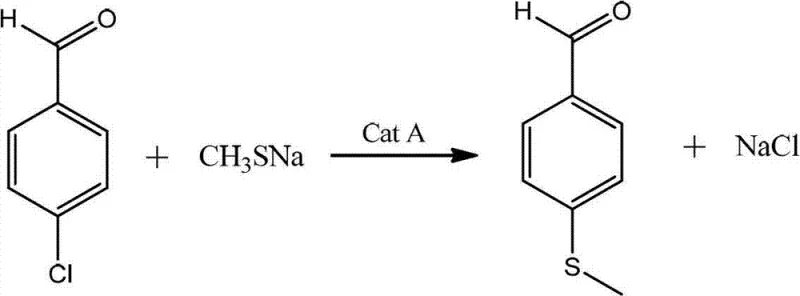
The second mechanistic pillar of this process is the selective oxidation of the sulfide moiety to the sulfone using hydrogen peroxide under acidic conditions. The presence of an oxidation catalyst, such as sodium tungstate or manganese sulfate, activates the hydrogen peroxide, generating reactive oxygen species that efficiently attack the sulfur atom. Crucially, the reaction conditions, including temperature control between 30 and 60°C and the careful regulation of acid concentration, prevent the over-oxidation of the sensitive aldehyde group to the corresponding carboxylic acid. This chemoselectivity is paramount, as it eliminates the need for protecting group strategies that would otherwise add complexity and cost to the synthesis. The final workup involves a simple pH adjustment to neutralize the acid and convert any trace acidic impurities into water-soluble salts, which are easily removed during crystallization, ensuring the final product meets stringent purity specifications required for pharmaceutical applications.
How to Synthesize p-Methylsulfonyl Benzaldehyde Efficiently
Implementing this synthesis protocol requires adherence to specific operational parameters to replicate the high yields and purity reported in the patent data. The process begins with the preparation of the reaction mixture for the substitution step, ensuring the correct stoichiometric balance between the aldehyde, the thiolate source, and the phase-transfer catalyst. Following the isolation of the methylthio intermediate, the oxidation step demands careful thermal management to control the exothermic nature of the peroxide reaction while maintaining the activity of the oxidation catalyst. Operators must monitor the reaction progress via TLC or HPLC to determine the precise endpoint, ensuring complete conversion before proceeding to the crystallization and drying phases. The detailed standardized synthesis steps below outline the critical control points necessary for successful scale-up.
- React p-chlorobenzaldehyde with sodium methyl mercaptide in aqueous solution using a phase-transfer catalyst at 30-50°C to form p-methylthio benzaldehyde.
- Oxidize the resulting p-methylthio benzaldehyde using hydrogen peroxide and sulfuric acid with an oxidation catalyst at 30-60°C to obtain the final sulfone product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this patented synthesis route offers tangible benefits that extend beyond mere technical feasibility. The primary advantage lies in the substantial simplification of the supply chain, as the process relies on p-chlorobenzaldehyde, a commodity chemical that is widely available and cost-effective compared to the specialized reagents required for older methods. By eliminating the need for hazardous chlorosulfonation and bromination steps, the manufacturing facility can operate with reduced safety risks and lower insurance premiums, directly contributing to overhead cost optimization. Furthermore, the high atom economy of the nucleophilic substitution and the use of hydrogen peroxide, which decomposes into water, significantly reduce the volume of hazardous waste requiring treatment, leading to drastic savings in environmental compliance costs.
- Cost Reduction in Manufacturing: The elimination of transition metal catalysts in the first step and the use of inexpensive hydrogen peroxide in the second step drastically lower the raw material costs associated with the synthesis. Unlike previous methods that suffered from yields below 50%, this process consistently achieves total yields exceeding 90%, which effectively halves the raw material consumption per kilogram of finished product. This dramatic improvement in yield efficiency translates directly into a lower cost of goods sold, allowing for more competitive pricing in the global market for antibiotic intermediates without sacrificing margin.
- Enhanced Supply Chain Reliability: The robustness of this two-step process ensures a stable and continuous supply of p-methylsulfonyl benzaldehyde, mitigating the risks associated with complex multi-step syntheses that are prone to bottlenecks. The mild reaction conditions and the use of stable, non-hazardous reagents mean that production can be maintained consistently throughout the year, regardless of seasonal variations or regulatory tightening on hazardous chemical transport. This reliability is crucial for downstream pharmaceutical manufacturers who depend on a steady flow of high-quality intermediates to maintain their own production schedules for essential antibiotics like thiamphenicol.
- Scalability and Environmental Compliance: The process is inherently designed for industrial scale-up, with reaction parameters that are easily controlled in large reactors, ensuring that laboratory success translates seamlessly to commercial production volumes. The reduction in hazardous waste generation and the avoidance of toxic solvents align with increasingly strict global environmental regulations, future-proofing the supply chain against potential regulatory shutdowns. This environmental stewardship not only protects the company's reputation but also ensures long-term operational continuity in regions with rigorous ecological standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of p-methylsulfonyl benzaldehyde, derived directly from the specific innovations and data presented in the patent literature. Understanding these details is essential for technical teams evaluating the feasibility of integrating this intermediate into their existing supply chains or R&D pipelines. The answers provided reflect the optimized conditions and performance metrics achieved through the novel catalytic systems described in the intellectual property.
Q: What are the advantages of this synthesis route over traditional toluene-based methods?
A: Traditional routes starting from toluene involve lengthy steps like chlorosulfonation, reduction, and bromination, resulting in low yields and high environmental pressure. This patented method uses p-chlorobenzaldehyde directly, shortening the route significantly and achieving total yields over 90% with high purity.
Q: Which catalysts are critical for the oxidation step in this process?
A: The oxidation step utilizes specific catalysts such as sodium tungstate, manganese sulfate, or titanium oxide in the presence of sulfuric acid. These catalysts facilitate the efficient conversion of the sulfide group to the sulfone group using hydrogen peroxide while minimizing side reactions.
Q: What is the expected purity of the final p-methylsulfonyl benzaldehyde product?
A: According to the patent embodiments, the final product achieves a purity of 99.5% to 99.8% as analyzed by HPLC. The purification process involves pH regulation and crystallization, effectively removing impurities like thiamphenicol benzoic acid salts.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable p-Methylsulfonyl Benzaldehyde Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-purity intermediates play in the development and manufacture of life-saving antibiotics. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of advanced synthetic routes like the one described in CN102675167B are fully realized in practical, large-scale operations. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of p-methylsulfonyl benzaldehyde supports the efficacy and safety of your final pharmaceutical formulations.
We invite you to collaborate with us to leverage these technological advancements for your specific project needs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our optimized manufacturing capabilities can enhance your supply chain efficiency and reduce your overall production costs.
