Scalable Synthesis of 2-Chloro-4-Fluorobenzoic Acid: A Cost-Effective Route for Herbicide Intermediates
Introduction to Advanced Synthetic Methodologies
The landscape of fine chemical manufacturing is constantly evolving, driven by the dual imperatives of cost efficiency and environmental sustainability. A pivotal development in this sector is documented in Chinese patent CN114790149A, which outlines a robust method for synthesizing 2-chloro-4-fluorobenzoic acid using 2-chloro-4-fluoroaniline as the starting material. This compound serves as a critical building block in the production of saflufenacil, a high-potential herbicide, making its efficient synthesis a priority for the global agrochemical industry. The patented approach addresses significant limitations found in legacy technologies by replacing hazardous oxidants and expensive noble metal catalysts with a streamlined diazotization-cyanation sequence followed by hydrolysis. For R&D directors and procurement specialists, understanding this shift is vital, as it represents a move towards safer, more economically viable supply chains for high-purity agrochemical intermediates without compromising on yield or quality standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
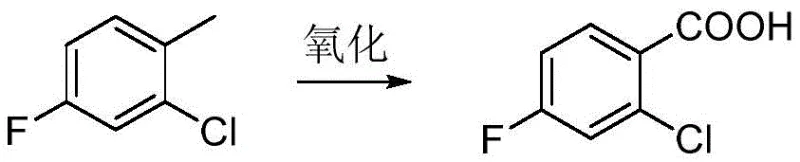
Historically, the production of 2-chloro-4-fluorobenzoic acid has relied on several chemically intensive pathways that pose substantial operational and environmental risks. One traditional domestic method involves the oxidation of 2-chloro-4-fluorotoluene using dichromates as the primary oxidant. While conceptually straightforward, this process generates massive amounts of toxic chromium waste, creating severe environmental compliance burdens and disposal costs. Furthermore, alternative routes described in literature, such as those utilizing rhodium catalysts for oxidation or palladium-catalyzed carbonylation of iodo-substituted precursors, introduce prohibitive raw material costs. These precious metal-dependent processes not only inflate the bill of materials but also necessitate complex downstream purification to remove trace metal residues, which is unacceptable for pharmaceutical-grade intermediates. Additionally, methods employing strong oxidants like sodium chlorite carry inherent safety risks due to potential explosivity, complicating reactor design and safety protocols for large-scale operations.
The Novel Approach
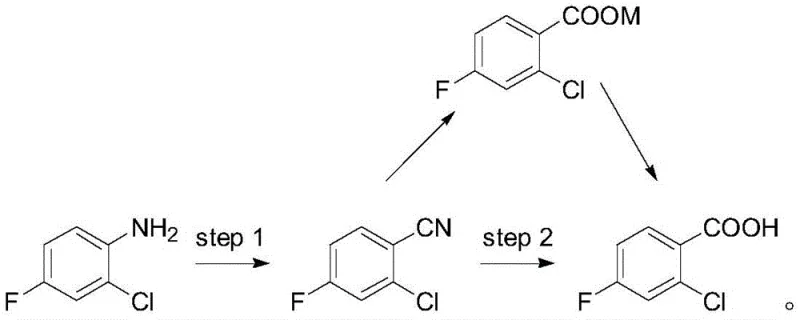
In stark contrast to these legacy methods, the novel approach detailed in the patent leverages a classic yet optimized Sandmeyer-type transformation to achieve superior outcomes. By starting with readily available 2-chloro-4-fluoroaniline, the process converts the amino group into a nitrile functionality via a diazonium salt intermediate using cuprous cyanide. This nitrile is subsequently hydrolyzed under either acidic or alkaline conditions to yield the target carboxylic acid. This strategy elegantly bypasses the need for toxic chromium reagents and expensive noble metals entirely. The reaction conditions are notably mild, with the diazotization step carefully controlled at low temperatures between 0-3°C to ensure stability, followed by hydrolysis at manageable temperatures of 80-100°C. This transition from high-risk oxidation chemistry to controlled substitution and hydrolysis represents a paradigm shift in process safety and economic feasibility for manufacturers seeking a reliable agrochemical intermediate supplier.
Mechanistic Insights into Sandmeyer Cyanation and Nitrile Hydrolysis
The core of this synthetic innovation lies in the precise execution of the diazotization and subsequent cyanation steps. The mechanism begins with the formation of the diazonium salt from 2-chloro-4-fluoroaniline using sodium nitrite and hydrochloric acid. Maintaining the reaction temperature strictly between 0-3°C is critical during this phase to prevent the decomposition of the unstable diazonium species, which could lead to phenol byproducts and reduced yields. Once formed, the diazonium salt reacts with cuprous cyanide (CuCN) to install the cyano group. The patent specifies optimized molar ratios, such as 1:1.2-1.6 for the aniline-to-CuCN ratio, which ensures complete conversion while minimizing excess reagent waste. Following the cyanation, the organic phase undergoes a crucial washing step with saturated aqueous ammonia. This step is mechanistically significant as it complexes residual copper ions, preventing them from carrying over into the final product and ensuring the high purity required for downstream applications.
The second mechanistic phase involves the hydrolysis of the 2-chloro-4-fluorobenzonitrile intermediate to the final acid. This can be achieved through two distinct pathways: acid-catalyzed hydrolysis using sulfuric acid or base-catalyzed hydrolysis using sodium or potassium hydroxide. In the alkaline pathway, the nitrile is converted to the corresponding carboxylate salt at temperatures around 80°C, followed by acidification to pH 1-3 to precipitate the free acid. The choice between acid or base hydrolysis offers flexibility depending on the specific equipment and waste treatment capabilities of the manufacturing facility. Both pathways avoid the harsh conditions associated with direct methyl oxidation, resulting in cleaner reaction profiles and simplified isolation procedures. This mechanistic robustness allows for consistent production of high-purity 2-chloro-4-fluorobenzoic acid with mass yields exceeding 90% in the hydrolysis step, demonstrating the practical viability of the route.
How to Synthesize 2-Chloro-4-Fluorobenzoic Acid Efficiently
Implementing this synthesis requires strict adherence to the thermal and stoichiometric parameters outlined in the patent to maximize efficiency and safety. The process is divided into two main stages: the formation of the nitrile intermediate and its subsequent conversion to the acid. Operators must prioritize temperature control during the exothermic diazotization phase and ensure thorough mixing during the heterogeneous cyanation reaction. The following guide summarizes the standardized operational protocol derived from the patent examples, providing a clear roadmap for laboratory validation and pilot plant scale-up. Detailed standard operating procedures for each unit operation are essential to maintain the high quality and reproducibility demanded by international regulatory standards.
- Perform diazotization of 2-chloro-4-fluoroaniline with sodium nitrite and hydrochloric acid at 0-3°C, followed by reaction with cuprous cyanide to form 2-chloro-4-fluorobenzonitrile.
- Hydrolyze the resulting nitrile under acidic (sulfuric acid) or alkaline (sodium/potassium hydroxide) conditions at elevated temperatures (80-100°C).
- Acidify the reaction mixture to pH 1-3 using mineral acids to precipitate the final 2-chloro-4-fluorobenzoic acid product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers transformative benefits that extend beyond simple chemical yield. The primary advantage is the drastic simplification of the supply chain by eliminating dependency on volatile and expensive noble metal markets. By removing the need for rhodium or palladium catalysts, manufacturers can insulate their production costs from the fluctuations of precious metal prices, leading to more stable long-term pricing for customers. Furthermore, the use of commodity chemicals like sodium nitrite, hydrochloric acid, and cuprous cyanide ensures a robust and continuous supply of raw materials, reducing the risk of production stoppages due to sourcing bottlenecks. This reliability is crucial for maintaining the continuity of supply for key agrochemical actives like saflufenacil.
- Cost Reduction in Manufacturing: The elimination of expensive precious metal catalysts directly translates to significant cost savings in the bill of materials. Unlike carbonylation routes that require costly palladium acetate or oxidation routes needing rhodium, this method utilizes copper, which is orders of magnitude cheaper. Additionally, the avoidance of toxic dichromates reduces the financial burden associated with hazardous waste disposal and environmental compliance fees. The simplified workup procedures, such as the ammonia wash for copper removal, further reduce processing time and utility consumption, contributing to a leaner and more cost-effective manufacturing process overall.
- Enhanced Supply Chain Reliability: Sourcing 2-chloro-4-fluoroaniline is generally more straightforward and stable compared to specialized precursors like 2-chloro-4-fluoroiodobenzene or protected aniline derivatives. The reliance on widely available industrial reagents minimizes the risk of supply disruptions caused by geopolitical factors or limited producer capacity. This stability allows supply chain planners to forecast production schedules with greater confidence, ensuring that downstream customers receive their orders on time. The robustness of the chemistry also means that the process is less sensitive to minor variations in raw material quality, further enhancing supply security.
- Scalability and Environmental Compliance: From an EHS (Environment, Health, and Safety) perspective, this route is vastly superior to traditional methods. The absence of hexavalent chromium and explosive sodium chlorite significantly lowers the safety risk profile of the plant, potentially reducing insurance premiums and regulatory scrutiny. The process generates less hazardous waste, aligning with modern green chemistry principles and corporate sustainability goals. The mild reaction conditions and standard pressure requirements make the technology easily scalable from pilot batches to multi-ton commercial production without the need for specialized high-pressure reactors or exotic containment systems.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis of 2-chloro-4-fluorobenzoic acid. These answers are derived directly from the technical specifications and beneficial effects described in the patent documentation, providing clarity on process capabilities and advantages. Understanding these details is essential for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: Why is the Sandmeyer cyanation route preferred over direct oxidation for 2-chloro-4-fluorobenzoic acid?
A: Direct oxidation using dichromates involves highly toxic heavy metals and often results in lower yields and significant environmental pollution. The Sandmeyer route described in CN114790149A avoids these toxic oxidants and expensive precious metal catalysts like rhodium or palladium, offering a greener and more cost-effective alternative suitable for large-scale production.
Q: How does this process handle copper residue removal to ensure high purity?
A: The patent specifies a critical purification step where the organic phase containing the nitrile intermediate is washed with saturated aqueous ammonia solution. This effectively complexes and removes residual copper compounds before the hydrolysis step, ensuring the final acid product meets stringent purity specifications required for pharmaceutical and agrochemical applications.
Q: Is this synthesis method scalable for industrial manufacturing?
A: Yes, the process is designed for scalability. It utilizes common industrial reagents such as hydrochloric acid, sodium nitrite, and cuprous cyanide, and operates under relatively mild conditions (0-3°C for diazotization, 60-100°C for hydrolysis). The avoidance of high-pressure carbonylation or explosive oxidants like sodium chlorite significantly enhances operational safety and ease of scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Chloro-4-Fluorobenzoic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of efficient intermediate synthesis in the global agrochemical market. Our team of expert chemists has thoroughly analyzed the methodology presented in CN114790149A and possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. We are committed to delivering high-purity 2-chloro-4-fluorobenzoic acid that meets stringent purity specifications, utilizing our rigorous QC labs to ensure every batch complies with international standards. Our state-of-the-art facilities are equipped to handle the specific thermal and safety requirements of diazotization and hydrolysis reactions, guaranteeing a consistent and reliable supply for your herbicide manufacturing needs.
We invite you to collaborate with us to optimize your supply chain and reduce manufacturing costs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our advanced synthesis capabilities can support your long-term business goals and enhance your competitive edge in the market.
