Efficient Lithium Iodide Catalyzed Synthesis of Aza Bridged Ring Frameworks for Commercial Scale-up
Efficient Lithium Iodide Catalyzed Synthesis of Aza Bridged Ring Frameworks for Commercial Scale-up
The pharmaceutical industry constantly seeks robust methodologies for constructing complex nitrogen-containing heterocycles, which serve as the structural backbone for numerous bioactive agents. As detailed in patent CN114057721A, a groundbreaking approach has been developed for the efficient construction of aza[n.2.1] (n=3,4) bridged ring framework compounds. This technology leverages a lithium iodide-promoted intramolecular [3+2] cycloaddition reaction, offering a streamlined pathway to highly functionalized scaffolds that are pivotal in medicinal chemistry. The significance of this innovation lies not only in its chemical elegance but also in its potential to simplify the supply chain for high-value pharmaceutical intermediates. By enabling the rapid assembly of these complex cores from readily available aziridine precursors, this method addresses critical bottlenecks in the synthesis of analgesic drug candidates like homoepibxidine.
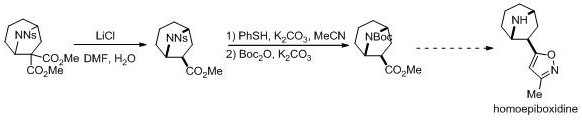
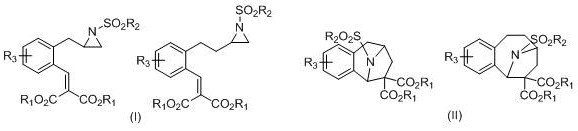
The versatility of this synthetic route is demonstrated by its applicability to a wide range of substrates bearing different electronic and steric properties. The resulting aza[n.2.1] bridged ring skeletons possess a high degree of functionalization, making them ideal building blocks for further derivatization. For procurement managers and supply chain directors, the ability to access such complex intermediates through a concise, high-yielding process translates directly into enhanced supply security and reduced lead times. The method's compatibility with various substituents, including halogens and sulfonyl groups, ensures that diverse chemical space can be explored without necessitating entirely new process development campaigns for each analog.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of aza[n.2.1] bridged ring skeletons has relied heavily on intermolecular cycloaddition reactions, such as [3+2] cycloadditions of ylides or [6+2] cycloadditions involving azapyridines. While these traditional strategies have provided access to certain target molecules, they are often plagued by significant limitations that hinder large-scale manufacturing. Conventional methods frequently require harsh reaction conditions, including elevated temperatures or the use of sensitive organometallic reagents that demand strict inert atmosphere protection. Furthermore, intermolecular reactions often suffer from issues related to regioselectivity and stereoselectivity, leading to complex mixtures of products that are difficult and costly to separate. The reliance on expensive transition metal catalysts in some prior art routes also introduces concerns regarding residual metal contamination, necessitating additional purification steps that drive up production costs and extend cycle times.
The Novel Approach
In stark contrast to these legacy methods, the novel lithium iodide-promoted strategy described in the patent offers a paradigm shift towards efficiency and simplicity. This approach utilizes an intramolecular [3+2] cycloaddition of aziridines containing electron-deficient olefins, which inherently favors the formation of the desired bridged architecture with high fidelity. The reaction proceeds under remarkably mild conditions, typically ranging from room temperature to 60°C, and crucially, does not require inert gas protection, thereby drastically reducing operational complexity. The use of ethyl acetate as a solvent further underscores the green chemistry credentials of this process, replacing hazardous chlorinated solvents often found in older protocols. By streamlining the bond-forming events into a single pot operation involving sequential ring opening, Michael addition, and nucleophilic substitution, this method achieves superior atom economy and bonding efficiency.
Mechanistic Insights into Lithium Iodide-Promoted Cyclization
The mechanistic pathway of this transformation is a testament to the power of simple Lewis acid catalysis in driving complex molecular rearrangements. The reaction initiates with the coordination of the lithium cation to the nitrogen atom of the aziridine ring, which significantly enhances the electrophilicity of the adjacent carbon atoms. This activation facilitates the nucleophilic attack by the iodide anion, leading to the regioselective ring-opening of the strained aziridine moiety. The resulting open-chain intermediate, now possessing a nucleophilic nitrogen and an electrophilic carbon center activated by the electron-deficient olefin, is perfectly poised for the subsequent intramolecular cyclization. The iodide ion acts not merely as a nucleophile but effectively shuttles the reaction forward, promoting the Michael addition followed by an intramolecular nucleophilic substitution that closes the bridge to form the stable aza[n.2.1] skeleton.
From an impurity control perspective, the mildness of the lithium iodide promotion is a critical factor in ensuring high product purity. Because the reaction avoids extreme thermal stress or highly reactive radical species, the formation of decomposition by-products or polymerization side-reactions is minimized. The chemoselectivity of the iodide-mediated ring opening ensures that other sensitive functional groups on the aromatic rings or ester moieties remain intact throughout the process. This high level of control over the reaction trajectory means that the crude reaction mixture is cleaner, reducing the burden on downstream purification processes like silica gel chromatography. For R&D directors, this mechanistic clarity provides confidence in the scalability of the process, as the risk of runaway exotherms or unpredictable side-reactions during scale-up is substantially mitigated by the benign nature of the reagents involved.
How to Synthesize Aza[n.2.1] Bridged Ring Compounds Efficiently
The practical execution of this synthesis is designed for ease of operation, making it highly suitable for both laboratory optimization and industrial manufacturing. The protocol involves dissolving the aziridine precursor in ethyl acetate and adding the lithium iodide catalyst, followed by stirring at ambient or slightly elevated temperatures. The reaction progress is easily monitored using thin-layer chromatography (TLC) with a standard ethyl acetate and petroleum ether eluent system. Upon completion, the workup procedure is straightforward, involving aqueous quenching, extraction, and standard drying techniques, avoiding the need for specialized quenching reagents or complex phase separations. This simplicity allows for rapid turnover of batches and facilitates the quick generation of material for biological testing or further process development.
- Dissolve the electron-deficient olefin-containing aziridine substrate in ethyl acetate solvent at room temperature without inert gas protection.
- Add lithium iodide catalyst (20-200 mol%) to the reaction system and stir at temperatures ranging from room temperature to 60°C for 3 to 6 hours.
- Monitor reaction completion via TLC, then perform aqueous workup, extraction with ethyl acetate, and silica gel column chromatography purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this lithium iodide-promoted synthesis offers tangible strategic advantages that extend beyond mere chemical yield. The elimination of expensive transition metal catalysts and the removal of the requirement for inert gas protection significantly lower the barrier to entry for manufacturing these intermediates. This translates into a more resilient supply chain where production is less dependent on specialized equipment or scarce catalytic materials. The use of commodity chemicals like lithium iodide and ethyl acetate ensures that raw material sourcing is stable and cost-effective, shielding the supply chain from the volatility often associated with specialty reagents. Furthermore, the simplified workup and purification流程 reduce the overall processing time, allowing for faster throughput and improved responsiveness to market demand fluctuations.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the drastic simplification of the reaction conditions and reagent costs. By utilizing inexpensive lithium iodide instead of precious metal catalysts, the direct material cost per kilogram of product is significantly reduced. Additionally, the ability to run the reaction without inert gas protection eliminates the capital and operational expenditures associated with maintaining nitrogen or argon atmospheres on a large scale. The high atom economy of the intramolecular cyclization means that less raw material is wasted as by-product, further enhancing the overall cost efficiency of the manufacturing campaign. These factors combine to deliver substantial cost savings that can be passed down the supply chain or reinvested into further R&D efforts.
- Enhanced Supply Chain Reliability: Supply chain reliability is bolstered by the robustness and generality of the synthetic method. Since the reaction tolerates a wide variety of substituents on the aromatic rings and the sulfonyl group, a single manufacturing platform can be adapted to produce a library of different analogs without major retooling. This flexibility allows suppliers to respond quickly to changes in client specifications or drug development pipelines. Moreover, the stability of the reagents and the mild reaction conditions reduce the risk of batch failures due to environmental factors or minor procedural deviations. This consistency ensures a steady flow of high-quality intermediates, minimizing the risk of stockouts that could delay downstream drug synthesis.
- Scalability and Environmental Compliance: Scaling this process from gram to ton quantities is facilitated by the absence of hazardous reagents and the use of relatively green solvents. Ethyl acetate is a preferred solvent in the pharmaceutical industry due to its favorable safety profile and ease of recovery, aligning with increasingly stringent environmental regulations. The lack of heavy metal residues simplifies the waste treatment process and reduces the environmental footprint of the manufacturing site. The straightforward exothermic profile of the reaction allows for safe scale-up in standard stainless steel reactors without the need for exotic containment systems. This ease of scale-up ensures that commercial volumes can be achieved rapidly to meet the demands of clinical trials and eventual market launch.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this aza[n.2.1] bridged ring synthesis technology. These answers are derived directly from the experimental data and technical disclosures within the patent literature, providing a clear picture of the process capabilities. Understanding these details is essential for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios. The clarity of the reaction parameters and the robustness of the outcome make this a low-risk, high-reward opportunity for process intensification.
Q: What catalyst is used for the aza[n.2.1] bridged ring synthesis?
A: The process utilizes lithium iodide (LiI) as the primary promoter, though sodium iodide and other alkali metal halides can also be effective. LiI is preferred for its high efficiency and cost-effectiveness.
Q: Does this synthesis require inert gas protection?
A: No, a significant advantage of this method is that it proceeds smoothly at room temperature to 60°C without the need for inert gas protection, simplifying operational requirements.
Q: What are the primary applications of these bridged ring compounds?
A: These compounds serve as critical intermediates for synthesizing biologically active molecules, specifically noted for their utility in producing homoepibxidine, a potent analgesic agent.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Aza[n.2.1] Bridged Ring Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that advanced synthetic methodologies play in accelerating drug discovery and development. Our team of expert chemists has thoroughly analyzed the lithium iodide-promoted cyclization route and is fully prepared to translate this academic innovation into commercial reality. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can move seamlessly from the bench to the plant. Our state-of-the-art facilities are equipped to handle the specific requirements of this chemistry, including rigorous QC labs that enforce stringent purity specifications to guarantee the quality of every batch delivered. We are committed to being a partner that not only supplies materials but also optimizes the process for maximum efficiency and sustainability.
We invite you to engage with our technical procurement team to discuss how this technology can benefit your specific pipeline. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic advantages of switching to this novel synthetic route for your aza[n.2.1] bridged ring needs. We encourage potential partners to contact us for specific COA data and route feasibility assessments tailored to your target molecules. Let us collaborate to secure your supply chain and reduce your manufacturing costs with this cutting-edge chemical technology.
