Advanced Silver-Catalyzed Synthesis of Alpha Alpha Gem Difluoro Carbonyl Compounds for Pharma
Advanced Silver-Catalyzed Synthesis of Alpha Alpha Gem Difluoro Carbonyl Compounds for Pharma
The landscape of organofluorine chemistry is undergoing a significant transformation driven by the critical need for efficient access to fluorinated structural motifs found in modern pharmaceuticals and agrochemicals. Patent CN114249679A introduces a groundbreaking methodology for the preparation of α,α-gem-difluoro carbonyl compounds, a class of molecules renowned for their metabolic stability and bioactivity enhancement properties. This innovation leverages a novel silver-catalyzed C-F bond cleavage and C-C bond formation strategy, utilizing readily available trifluoromethyl precursors to construct these valuable scaffolds with exceptional atom economy. By addressing the longstanding challenge of controlling chemical selectivity during defluorination, this technology offers a robust pathway for generating high-purity intermediates essential for drug discovery pipelines.
For research and development teams focused on complex molecule synthesis, the ability to install gem-difluoro groups directly from trifluoromethyl sources represents a paradigm shift in retrosynthetic planning. The disclosed method operates under mild alkaline conditions using specialized metal silver salts, effectively bypassing the harsh reagents often required in traditional fluorination protocols. This approach not only streamlines the synthetic route but also aligns with green chemistry principles by minimizing waste and energy consumption. As the demand for fluorinated active pharmaceutical ingredients continues to surge, mastering this specific catalytic transformation becomes a strategic imperative for maintaining competitiveness in the fine chemical sector.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of α,α-gem-difluoro carbonyl skeletons has been plagued by significant synthetic hurdles that impede efficient large-scale production. Traditional methods often rely on direct fluorination of carbonyl precursors using hazardous reagents like DAST or Deoxo-Fluor, which pose severe safety risks and generate difficult-to-handle waste streams. Furthermore, alternative strategies involving the coupling of pre-functionalized difluoro building blocks frequently suffer from poor atom economy and limited substrate scope, restricting their utility in diverse medicinal chemistry campaigns. A particularly persistent issue is the difficulty in controlling the degree of defluorination; the bond energy of carbon-fluorine bonds weakens progressively, leading to uncontrolled over-defluorination and the formation of unwanted mono-fluorinated or non-fluorinated byproducts.
These selectivity issues drastically reduce the yield of the target compound and complicate downstream purification processes, thereby inflating the overall cost of goods. In an industrial setting, the inability to precisely halt the reaction at the gem-difluoro stage necessitates extensive chromatographic separation or recrystallization steps, which are time-consuming and solvent-intensive. Moreover, many conventional protocols require cryogenic temperatures or strictly anhydrous conditions that are challenging to maintain in multi-kilogram reactors. These operational complexities create bottlenecks in supply chains, extending lead times for critical intermediates and hindering the rapid iteration required in modern drug development cycles.
The Novel Approach
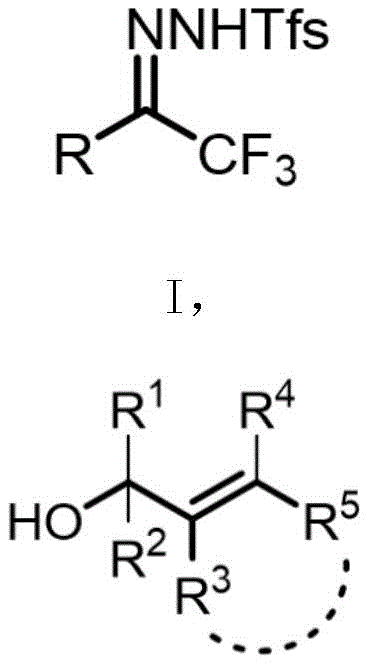
The methodology described in the patent data presents a sophisticated solution to these entrenched problems by employing a metal silver salt catalyst to mediate the selective activation of C-F bonds. This novel approach utilizes trifluoromethyl benzenesulfonylhydrazones as stable and accessible starting materials, which react with various nucleophiles such as allylic alcohols or heterocyclic methanols in a one-pot fashion. The use of a tailored silver catalyst, specifically complexes like TpBr3Ag, allows for precise modulation of the reaction pathway, ensuring that only two fluorine atoms are retained while the third is cleaved to facilitate carbon-carbon bond formation. This level of control effectively eliminates the formation of over-defluorinated impurities, resulting in crude reaction mixtures of significantly higher purity.
Operationally, this new route is remarkably user-friendly, proceeding in common organic solvents like toluene at moderate temperatures around 80°C, which eliminates the need for expensive cooling infrastructure. The reaction system is environmentally friendly, avoiding the generation of corrosive hydrogen fluoride gas typically associated with other defluorinative coupling reactions. By integrating the C-F bond cleavage and C-C bond formation into a single catalytic cycle, the process achieves superior step economy, reducing the number of unit operations required to reach the final product. This streamlined workflow translates directly into enhanced throughput and reliability, making it an ideal candidate for the commercial scale-up of complex fluorinated intermediates needed for next-generation therapeutics.
Mechanistic Insights into Silver-Catalyzed C-F Bond Cleavage
The efficacy of this transformation hinges on the unique electronic properties of the tris(pyrazolyl)borate silver catalysts, which serve as the cornerstone of the reaction mechanism. Unlike simple silver salts, these scorpionate ligand-stabilized complexes provide a specific coordination environment that facilitates the oxidative addition or single-electron transfer processes necessary for activating the strong carbon-fluorine bond. The catalyst likely interacts with the diazo species generated in situ from the hydrazone precursor, forming a reactive silver-carbene or silver-radical intermediate that initiates the defluorinative functionalization. This mechanistic pathway is distinct from traditional nucleophilic substitutions, allowing for the tolerance of sensitive functional groups that would otherwise decompose under basic or acidic conditions.
Impurity control is inherently built into the catalytic cycle through the steric and electronic tuning of the ligand framework. The bulky substituents on the pyrazole rings of the TpBr3Ag catalyst create a protective pocket around the metal center, preventing non-selective interactions that could lead to complete defluorination or polymerization of the reactive intermediates. This steric shielding ensures that the reaction proceeds exclusively through the desired gem-difluoro pathway, suppressing side reactions that typically plague fluorine chemistry. Furthermore, the use of mild inorganic bases like potassium carbonate helps to neutralize any acidic byproducts without promoting the decomposition of the fluorinated scaffold. For quality assurance teams, this inherent selectivity means that the impurity profile of the final API intermediate is much cleaner, simplifying the validation process and reducing the risk of genotoxic impurity formation.
How to Synthesize Alpha Alpha Gem Difluoro Carbonyl Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires adherence to specific procedural parameters to maximize yield and reproducibility. The protocol involves a straightforward one-pot procedure where the trifluoromethyl hydrazone and the alcohol coupling partner are combined with the silver catalyst and base in toluene. The reaction is heated to 80°C under an inert nitrogen atmosphere to prevent oxidation of the sensitive intermediates. Monitoring the reaction progress via thin-layer chromatography allows for precise determination of the endpoint, ensuring that the substrate is fully consumed without prolonged exposure to heat that might degrade the product. Detailed standardized synthetic steps for this transformation are provided in the guide below.
- In a glove box, add trifluoromethyl benzenesulfonylhydrazone 1a, 3-indolylmethanol 2a, TpBr3Ag catalyst, and K2CO3 base to a pressure-resistant tube.
- Add toluene solvent, stir well, and heat the mixture in an oil bath at 80°C under nitrogen atmosphere until substrate disappearance is detected by TLC.
- Pour reaction mixture into saturated brine, extract with dichloromethane, dry over sodium sulfate, and purify via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, this technology offers substantial opportunities for cost optimization and supply chain stabilization. The reliance on cheap and easily obtained trifluoromethyl compounds as starting materials significantly lowers the raw material entry cost compared to purchasing pre-functionalized difluoro building blocks, which often carry high price premiums due to complex manufacturing processes. Additionally, the elimination of hazardous fluorinating reagents reduces the costs associated with specialized containment equipment, waste disposal, and safety compliance measures. The simplified workup procedure, which involves standard aqueous extraction and silica gel chromatography, minimizes solvent consumption and processing time, leading to drastic simplification of the manufacturing workflow and reduced operational expenditures.
Enhanced supply chain reliability is another critical benefit, as the reagents required for this process, including the silver catalyst and potassium carbonate base, are commercially available from multiple global suppliers. This diversification of the supply base mitigates the risk of shortages that can occur with proprietary or single-source reagents. The robustness of the reaction conditions, which tolerate a wide range of substrates and functional groups, ensures consistent batch-to-batch quality even when scaling up production volumes. For supply chain heads, this predictability translates into reduced lead time for high-purity fluorinated intermediates, enabling more agile responses to market demands and clinical trial timelines without compromising on quality standards.
Scalability and environmental compliance are seamlessly integrated into this process design, making it highly attractive for sustainable manufacturing initiatives. The reaction operates at atmospheric pressure and moderate temperatures, removing the need for high-pressure autoclaves or cryogenic cooling systems, which simplifies equipment requirements for commercial scale-up. The use of toluene as a solvent, while requiring proper management, is well-established in industrial settings with existing recovery infrastructure. Furthermore, the high atom economy of the C-F bond cleavage strategy means that less waste is generated per kilogram of product, aligning with increasingly stringent environmental regulations. This combination of operational simplicity and environmental stewardship positions the technology as a future-proof solution for the commercial scale-up of complex polymer additives and pharmaceutical intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this silver-catalyzed defluorination technology. These answers are derived directly from the experimental data and specifications outlined in the patent documentation to provide clarity for potential adopters. Understanding these nuances is essential for evaluating the feasibility of integrating this method into existing production lines or research workflows.
Q: What is the optimal catalyst loading for this silver-catalyzed defluorination?
A: The patent specifies that the metal silver salt amount is preferably 10 mol% relative to the compound a, ensuring high efficiency without excessive metal usage.
Q: Can this method tolerate various functional groups on the substrate?
A: Yes, the method is suitable for synthesizing various highly functionalized compounds, tolerating groups such as halogens, nitro, cyano, and esters on the aryl ring.
Q: What represents the primary advantage of this C-F bond cleavage strategy?
A: The primary advantage is the ability to use cheap and easily obtained trifluoromethyl compounds as starting materials while controlling selectivity to avoid over-defluorination.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alpha Alpha Gem Difluoro Carbonyl Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this advanced silver-catalyzed synthesis for the production of high-value fluorinated intermediates. As a leading CDMO expert, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop discovery to full-scale manufacturing is seamless and efficient. Our state-of-the-art facilities are equipped to handle the specific requirements of organofluorine chemistry, including specialized containment for silver catalysts and rigorous QC labs capable of verifying stringent purity specifications. We are committed to delivering high-purity alpha,alpha-gem-difluoro carbonyls that meet the exacting standards of the global pharmaceutical industry.
We invite you to collaborate with us to leverage this cutting-edge technology for your next project. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and target molecules. Please contact our technical procurement team today to request specific COA data and route feasibility assessments. By partnering with us, you gain access to a reliable supply chain partner dedicated to driving innovation and efficiency in fine chemical manufacturing.
