Scalable Synthesis of 3-Quinolyl-5-Trifluoromethyl-1,2,4-Triazoles via Metal-Free Oxidative Cyclization
Scalable Synthesis of 3-Quinolyl-5-Trifluoromethyl-1,2,4-Triazoles via Metal-Free Oxidative Cyclization
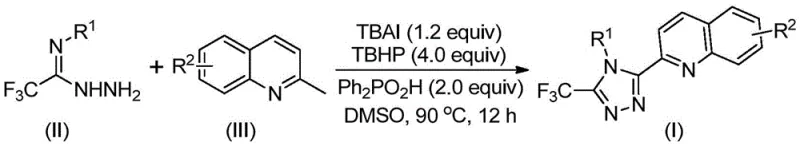
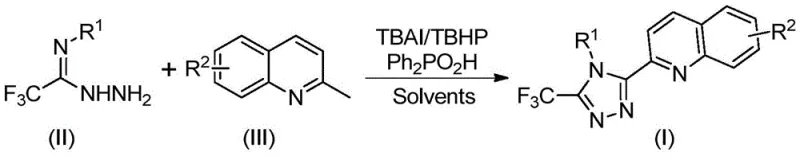
The pharmaceutical and fine chemical industries are constantly seeking more efficient pathways to construct complex heterocyclic scaffolds, particularly those containing nitrogen-rich motifs like 1,2,4-triazoles. A groundbreaking development in this sector is detailed in Chinese Patent CN113307790B, which discloses a highly efficient preparation method for 3-quinolyl-5-trifluoromethyl substituted 1,2,4-triazole compounds. This technology represents a significant leap forward by utilizing a metal-free oxidative cyclization strategy that couples trifluoroacetimidohydrazides with 2-methylquinolines. Unlike traditional multi-step syntheses that plague R&D departments with low yields and harsh conditions, this novel approach leverages a robust catalytic system comprising tetrabutylammonium iodide (TBAI) and tert-butyl hydroperoxide (TBHP). The result is a streamlined process that operates under relatively mild thermal conditions without the need for stringent anhydrous or oxygen-free environments, offering a compelling solution for reliable pharmaceutical intermediate supplier networks aiming to optimize their production pipelines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of quinolyl-substituted 1,2,4-triazoles has been fraught with inefficiencies that hinder commercial viability. The prior art predominantly relies on quinoline-2-carboxylic acid as the foundational starting material. This conventional route is notoriously cumbersome, typically requiring a sequence of at least five distinct chemical transformations to arrive at the target heterocyclic structure. Each step in this linear synthesis introduces potential yield losses, cumulative impurities, and significant operational overhead. Furthermore, the reaction conditions associated with these older methodologies are often severe, necessitating specialized equipment and rigorous safety protocols that drive up capital expenditure. The most critical bottleneck, however, is the abysmal overall yield; literature indicates that the total yield for these five-step sequences rarely exceeds 17%. Such poor atom economy renders these methods unsuitable for large-scale synthetic applications, creating a supply chain vulnerability for manufacturers who require consistent, high-volume access to these bioactive intermediates for drug discovery and development programs.
The Novel Approach
In stark contrast to the laborious traditional pathways, the methodology disclosed in patent CN113307790B offers a direct, one-pot oxidative cyclization route that fundamentally reshapes the manufacturing landscape. By employing cheap and easily obtainable 2-methylquinoline derivatives alongside trifluoroacetimidohydrazides, this novel approach bypasses the need for pre-functionalized carboxylic acid precursors. The reaction is driven by a synergistic catalytic system where TBAI and TBHP promote the oxidative transformation of the methyl group on the quinoline ring, facilitating its coupling with the hydrazide moiety. This innovation not only drastically reduces the number of unit operations but also eliminates the reliance on toxic heavy metal catalysts, which are often difficult to remove to ppm levels required for pharmaceutical grades. The operational simplicity is further enhanced by the tolerance for ambient atmospheric conditions, meaning reactions can be conducted in standard glassware without expensive inert gas manifolds. This shift from a five-step, low-yield process to a concise, high-yielding oxidative cyclization provides a robust platform for cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into TBAI/TBHP Promoted Oxidative Cyclization
The core of this technological breakthrough lies in the intricate mechanistic pathway facilitated by the TBAI/TBHP catalytic couple. The reaction initiates with the oxidation of the 2-methylquinoline substrate. Under the influence of tert-butyl hydroperoxide and the iodide source, the methyl group at the 2-position of the quinoline ring is oxidatively activated, effectively generating a reactive 2-quinoline carbaldehyde equivalent in situ. This transient aldehyde species then undergoes a condensation reaction with the trifluoroacetimidohydrazide to form a dehydrated hydrazone intermediate. This step is critical as it aligns the nitrogen nucleophile and the electrophilic carbon for the subsequent ring closure. Following condensation, the system undergoes an oxidative iodination event, likely mediated by in situ generated hypoiodite species, which activates the hydrazone for intramolecular electrophilic substitution. The final aromatization step releases the iodine species back into the catalytic cycle, yielding the stable 3-quinolyl-5-trifluoromethyl substituted 1,2,4-triazole core. Alternatively, the patent suggests that a free radical process may also contribute to the reaction progression, highlighting the versatility of the radical species generated by TBHP decomposition in DMSO.
From an impurity control perspective, this mechanism offers distinct advantages over transition-metal catalyzed cross-couplings. The absence of palladium, copper, or nickel eliminates the risk of metal leaching, a common regulatory hurdle in API synthesis. The primary byproducts are derived from the oxidant (tert-butanol) and the iodide salt, both of which are water-soluble and easily removed during the aqueous work-up or silica gel filtration described in the post-processing procedures. The use of diphenylphosphinic acid as an additive further stabilizes the reaction environment, potentially suppressing side reactions such as over-oxidation of the quinoline ring or polymerization of the hydrazide. This clean reaction profile ensures that the crude product contains fewer structurally related impurities, thereby simplifying the final purification via column chromatography and enhancing the overall purity profile of the high-purity OLED material or pharmaceutical intermediate produced.
How to Synthesize 3-Quinolyl-5-Trifluoromethyl-1,2,4-Triazole Efficiently
Implementing this synthesis route requires careful attention to reagent stoichiometry and solvent selection to maximize the benefits of the oxidative cyclization. The patent specifies that dimethyl sulfoxide (DMSO) is the preferred organic solvent due to its ability to effectively dissolve both the organic substrates and the ionic catalyst while promoting high conversion rates. The molar ratio of reagents is optimized to ensure complete consumption of the limiting reagent, typically the 2-methylquinoline derivative. A typical protocol involves mixing tetrabutylammonium iodide, 70% tert-butyl hydroperoxide aqueous solution, diphenylphosphinic acid, the trifluoroacetimidohydrazide, and the 2-methylquinoline substrate in DMSO. The detailed standardized synthesis steps for replicating this high-efficiency process are outlined below.
- Combine tetrabutylammonium iodide (TBAI), tert-butyl hydroperoxide (TBHP), diphenylphosphinic acid, trifluoroacetimidohydrazide, and 2-methylquinoline in an organic solvent such as DMSO.
- Heat the reaction mixture to a temperature range of 80-100°C and maintain stirring for 8 to 14 hours to ensure complete conversion without requiring anhydrous or anaerobic conditions.
- Upon completion, filter the mixture, mix with silica gel, and perform column chromatography purification to isolate the high-purity 3-quinolyl-5-trifluoromethyl substituted 1,2,4-triazole product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented methodology translates into tangible strategic benefits beyond mere chemical curiosity. The shift away from multi-step syntheses involving expensive carboxylic acid precursors to a direct coupling of commodity chemicals like 2-methylquinoline significantly lowers the raw material cost base. Moreover, the elimination of heavy metal catalysts removes the need for specialized scavenging resins or complex extraction protocols designed to meet strict residual metal specifications. This simplification of the downstream processing directly correlates to reduced manufacturing cycle times and lower utility consumption. The robustness of the reaction conditions, which do not require cryogenic temperatures or absolute exclusion of moisture, allows for the utilization of standard reactor vessels, thereby increasing the available capacity within existing manufacturing facilities without the need for capital-intensive retrofitting.
- Cost Reduction in Manufacturing: The economic impact of this process is driven primarily by the drastic reduction in step count and the use of inexpensive, bulk-available reagents. By collapsing a five-step sequence into a single pot operation, manufacturers eliminate the yield losses inherent in isolation and purification between steps. The catalyst system, relying on TBAI and TBHP, is substantially cheaper than precious metal complexes like palladium or rhodium. Furthermore, the avoidance of anhydrous solvents reduces the cost associated with solvent drying and storage. These factors combine to deliver substantial cost savings per kilogram of finished product, making the commercial scale-up of complex pharmaceutical intermediates much more financially viable.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of widely available starting materials. 2-Methylquinoline and various substituted aromatic amines used to prepare the hydrazides are commodity chemicals with established global supply chains, reducing the risk of bottlenecks associated with custom-synthesized precursors. The operational flexibility of the reaction, which tolerates ambient atmosphere and moderate temperatures, minimizes the risk of batch failures due to equipment malfunction or environmental excursions. This reliability ensures consistent delivery schedules for downstream customers, reducing lead time for high-purity pharmaceutical intermediates and allowing for more accurate inventory planning and demand forecasting.
- Scalability and Environmental Compliance: From an environmental and safety standpoint, this method aligns well with green chemistry principles. The absence of toxic heavy metals simplifies waste stream management and reduces the environmental footprint of the manufacturing process. The primary waste products are organic salts and tert-butanol, which are easier to treat than heavy metal sludge. The scalability is evidenced by the successful execution of the reaction in standard Schlenk tubes and the potential for translation to larger reactors given the homogeneous nature of the reaction mixture. This ease of scale-up facilitates the rapid transition from gram-scale R&D batches to multi-kilogram pilot runs and eventually to 100 MT annual commercial production, ensuring a steady supply for growing market demands.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this oxidative cyclization technology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity for technical teams evaluating this route for integration into their existing portfolios. Understanding these nuances is essential for assessing the feasibility of adopting this method for specific target molecules within the 1,2,4-triazole class.
Q: What are the primary advantages of the TBAI/TBHP catalytic system over traditional heavy metal catalysts?
A: The TBAI/TBHP system eliminates the need for toxic transition metals, significantly simplifying downstream purification and reducing heavy metal residue risks in the final API. Furthermore, it operates under ambient atmospheric conditions, removing the costly requirement for inert gas protection or strict anhydrous environments.
Q: How does the yield of this novel oxidative cyclization compare to conventional quinoline-2-carboxylic acid routes?
A: Traditional methods involving quinoline-2-carboxylic acid typically suffer from low overall yields (around 17%) across five steps. In contrast, this patented one-pot oxidative cyclization achieves isolated yields up to 97% for specific substrates, representing a massive improvement in atom economy and process efficiency.
Q: Is this synthesis method suitable for large-scale commercial production?
A: Yes, the method is highly scalable. It utilizes cheap, commercially available starting materials like 2-methylquinoline and avoids sensitive reagents. The robust reaction conditions (80-100°C in DMSO) and simple work-up procedures make it ideal for kilogram-to-ton scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Quinolyl-5-Trifluoromethyl-1,2,4-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the metal-free oxidative cyclization technology described in CN113307790B for the production of advanced heterocyclic intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent are realized in practical, industrial settings. Our state-of-the-art facilities are equipped to handle the specific solvent systems and thermal profiles required for this TBAI/TBHP promoted reaction, while our rigorous QC labs enforce stringent purity specifications to guarantee that every batch meets the exacting standards of the global pharmaceutical industry.
We invite R&D directors and procurement specialists to collaborate with us to leverage this efficient synthesis route for their upcoming projects. By partnering with our technical team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements and purity needs. We encourage you to contact our technical procurement team today to request specific COA data for our triazole intermediates and to discuss detailed route feasibility assessments that can accelerate your drug development timelines and optimize your supply chain economics.
