Industrial Synthesis of 1,2,3-Tri-O-Acetyl-5-Deoxy-Beta-D-Ribose for Oncology Drug Manufacturing
Industrial Synthesis of 1,2,3-Tri-O-Acetyl-5-Deoxy-Beta-D-Ribose for Oncology Drug Manufacturing
The pharmaceutical industry continuously seeks robust and scalable synthetic routes for critical nucleoside intermediates, particularly those serving the oncology sector. Patent CN108727440B discloses a highly efficient preparation method for 1,2,3-tri-O-acetyl-5-deoxy-beta-D-ribose, a key precursor in the synthesis of Capecitabine, a widely prescribed antitumor agent. This specific chemical entity, identified by CAS number 62211-93-2, plays a pivotal role in the manufacturing supply chain for colorectal and breast cancer treatments. The disclosed technology addresses long-standing challenges in nucleoside chemistry by utilizing inosine as a starting material, thereby bypassing the complex purification issues associated with traditional sugar-based routes. By integrating a novel halogenation strategy followed by catalytic hydrogenation and boric acid-mediated acetylation, this method offers a streamlined pathway that is exceptionally well-suited for large-scale commercial production. For R&D directors and procurement specialists, understanding the technical nuances of this patent is essential for securing a reliable supply of high-purity intermediates that meet stringent regulatory standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 1,2,3-tri-O-acetyl-5-deoxy-beta-D-ribose has relied heavily on D-ribose as the primary starting material, a route that presents significant industrial drawbacks. As documented in prior art such as US4340729, the conversion of D-ribose involves multiple steps that generate oily intermediates, making purification exceptionally difficult and costly on a commercial scale. Furthermore, the final triacetylation step in these conventional routes often suffers from low yields, typically ranging between 25% and 30%, and produces an unfavorable ratio of beta to alpha anomers at the 1-position. This stereochemical inefficiency necessitates extensive downstream processing to isolate the desired beta-isomer, drastically increasing production costs and waste generation. Additionally, alternative inosine-based routes, such as those described in CN100432088, have relied on iodine as an iodinating reagent, which results in low molecular utilization and the generation of substantial black iodine-containing wastewater, creating severe environmental compliance burdens for manufacturing facilities.
The Novel Approach
The methodology outlined in patent CN108727440B represents a significant technological leap by optimizing the inosine pathway to overcome these historical inefficiencies. Instead of using elemental iodine or complex tosylation reagents that generate hazardous waste, this novel approach employs a triphenyl phosphite-halogen system to selectively convert the 5-hydroxy group into a halogen while leaving other hydroxy positions unaffected. This selectivity is crucial for maintaining the structural integrity of the nucleoside backbone. Subsequent steps utilize palladium carbon catalysis for hydrogenation, a safer and more controllable alternative to the sodium borohydride reduction methods found in other patents like CN102432642B, which pose safety risks due to hydrogen gas generation during quenching. The final acetylation step is catalyzed by inorganic boric acid, which facilitates high conversion rates without the need for excessive reagents. This integrated approach not only simplifies the operational workflow but also ensures that the final product can be obtained with high purity through straightforward crystallization, making it a superior choice for industrial application.
Mechanistic Insights into Triphenyl Phosphite-Halogen Catalyzed Synthesis
The core of this synthetic innovation lies in the precise control of chemical transformations across three distinct stages, each optimized for maximum yield and minimal impurity formation. The process begins with the reaction of inosine with a triphenyl phosphite-halogen complex in the presence of an organic base such as triethylamine or diisopropylethylamine. This step is conducted at low temperatures, typically between -40°C and -10°C, to ensure regioselectivity, converting the 5-hydroxy group into a halogen (chlorine or bromine) to form compound 2. The reaction mixture is then treated to remove phosphorus-containing byproducts and organic base salts through crystallization in ethanol, a purification step that can be performed without isolating the intermediate, thus saving time and solvent. The second stage involves the catalytic hydrogenation of compound 2 using palladium carbon, which effectively removes the halogen atom to yield 5-deoxyinosine (compound 3). This hydrogenolysis is highly efficient, and the catalyst can be recycled for multiple batches with only minor replenishment, enhancing the economic viability of the process.
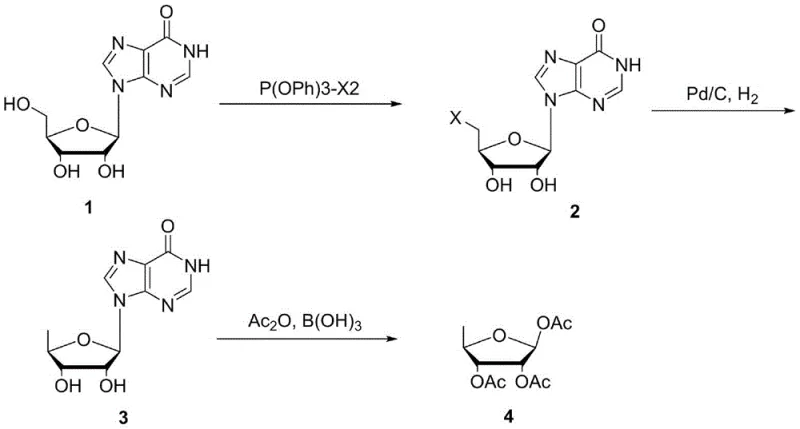
The final and perhaps most critical mechanistic step is the acetylation of 5-deoxyinosine in acetic anhydride catalyzed by inorganic boric acid. Unlike traditional acid catalysts that might promote degradation or side reactions, boric acid facilitates the cleavage of the glycosidic bond and subsequent acetylation of the ribose moiety with exceptional stereoselectivity. The reaction is carried out under reflux conditions, typically around 100°C, ensuring complete conversion. The mechanism allows for the direct removal of the acetylhypoxanthine byproduct through filtration, leaving the desired tri-acetyl ribose in the filtrate. This filtrate is then concentrated and crystallized using isopropanol, yielding the final product with an HPLC purity exceeding 99% and single impurity levels as low as 0.11%. This level of purity is achieved without the need for chromatographic purification, which is a significant advantage for cost reduction in [Pharmaceutical Intermediates] manufacturing.
How to Synthesize 1,2,3-Tri-O-Acetyl-5-Deoxy-Beta-D-Ribose Efficiently
Implementing this synthesis route requires careful attention to reaction conditions and reagent ratios to maximize the benefits described in the patent. The process is designed to be telescoped where possible, reducing the need for intermediate isolation and minimizing solvent consumption. Operators must ensure strict temperature control during the initial halogenation phase to prevent side reactions, while the hydrogenation step requires monitoring of catalyst activity to maintain consistent reaction rates across batches. The final acetylation step benefits from the precise molar ratio of boric acid to substrate, which drives the equilibrium towards the desired product. For detailed standard operating procedures and specific parameter settings required for GMP compliance, please refer to the technical guide below.
- React inosine with triphenyl phosphite-halogen in the presence of an organic base to form 5-halogen-inosine.
- Perform catalytic hydrogenation on the halogenated intermediate using palladium carbon to achieve dehalogenation.
- React the deoxy-inosine intermediate with acetic anhydride catalyzed by inorganic boric acid to yield the final tri-acetyl product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this patented synthesis route offers substantial strategic advantages for procurement managers and supply chain directors looking to optimize their sourcing of nucleoside intermediates. The primary benefit lies in the significant reduction of manufacturing costs driven by the use of cheap and readily available raw materials such as inosine and acetic anhydride. Unlike routes that rely on expensive protecting groups or hazardous reagents like sodium borohydride, this method utilizes common industrial chemicals that are easy to source globally, thereby reducing supply chain vulnerability. Furthermore, the ability to recycle the palladium carbon catalyst and the elimination of complex purification steps like chromatography translate directly into lower operational expenditures. The process is also inherently safer, avoiding the generation of large volumes of hazardous waste associated with iodine-based methods, which simplifies environmental compliance and reduces waste disposal costs.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and the ability to recycle the palladium carbon catalyst significantly lowers the raw material cost per kilogram. Additionally, the process avoids the need for energy-intensive purification techniques such as column chromatography, relying instead on simple crystallization and filtration which are far more economical at scale. The high overall yield, which surpasses many conventional methods, means that less starting material is required to produce the same amount of final product, further driving down the cost of goods sold. These factors combined create a robust economic model that allows for competitive pricing without compromising on quality.
- Enhanced Supply Chain Reliability: By utilizing inosine, a stable and commercially abundant starting material, manufacturers can avoid the supply bottlenecks often associated with specialized sugar derivatives. The robustness of the reaction conditions, which tolerate slight variations in temperature and reagent quality, ensures consistent production output even in varying industrial environments. This reliability is critical for maintaining continuous supply to downstream API manufacturers, reducing the risk of production stoppages due to intermediate shortages. The simplified workflow also shortens the overall production cycle time, allowing for faster turnaround on orders and improved responsiveness to market demand fluctuations.
- Scalability and Environmental Compliance: The process is designed with industrial scale-up in mind, utilizing standard reactor equipment and avoiding hazardous exothermic reactions that are difficult to control in large vessels. The reduction in hazardous waste generation, particularly the avoidance of iodine-containing wastewater, aligns with increasingly strict global environmental regulations. This compliance reduces the regulatory burden on manufacturing sites and minimizes the risk of fines or shutdowns. The ability to scale from laboratory to commercial production without significant process re-engineering ensures that supply can be ramped up quickly to meet the needs of large-scale pharmaceutical campaigns.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of this intermediate. These answers are derived directly from the technical specifications and beneficial effects reported in the patent literature, providing a clear understanding of the process capabilities. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this intermediate into their existing supply chains. The data reflects the performance under optimized conditions as described in the specific embodiments of the patent.
Q: What are the advantages of using inosine over D-ribose for this synthesis?
A: Using inosine avoids the formation of oily intermediates common in D-ribose routes, simplifies purification, and significantly improves the beta-to-alpha stereoselectivity ratio, leading to higher overall yields and purity suitable for GMP production.
Q: How is the purity of the final product controlled?
A: The process utilizes a specific boric acid catalyzed acetylation step followed by recrystallization in a methanol-water or isopropanol system, consistently achieving HPLC purity levels exceeding 99% with single impurities controlled below 0.15%.
Q: Is the catalyst system scalable for industrial production?
A: Yes, the process employs palladium carbon which can be filtered and recycled for multiple batches with minimal replenishment, and the reaction conditions avoid hazardous reagents like sodium borohydride, ensuring safety and scalability.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,2,3-Tri-O-Acetyl-5-Deoxy-Beta-D-Ribose Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the development and production of life-saving oncology medications. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the sophisticated chemistry described in patent CN108727440B can be executed with precision and consistency. We are committed to delivering products that meet stringent purity specifications, supported by our rigorous QC labs that utilize advanced analytical techniques to verify every batch. Our capability to manage complex nucleoside synthesis allows us to provide a stable supply of 1,2,3-tri-O-acetyl-5-deoxy-beta-D-ribose that supports your long-term manufacturing goals.
We invite you to collaborate with us to optimize your supply chain for Capecitabine production. By leveraging our technical expertise, we can provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality standards. We encourage potential partners to contact our technical procurement team to request specific COA data and route feasibility assessments, ensuring that our capabilities align perfectly with your project needs. Let us help you secure a reliable and cost-effective source for this essential pharmaceutical intermediate.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
