Advanced One-Pot Synthesis of Benzo Dibenzofuran Intermediates for High-Performance OLED Manufacturing
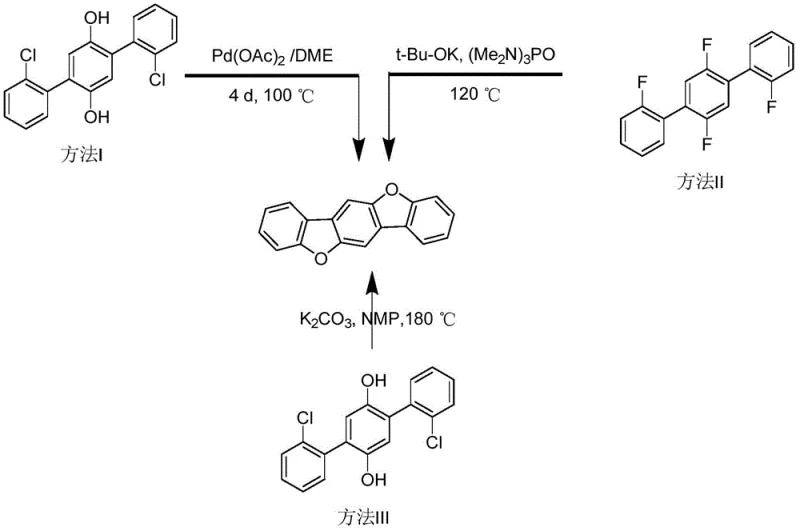
The rapid evolution of the organic light-emitting diode (OLED) industry demands increasingly sophisticated molecular architectures capable of delivering superior charge transport and luminescence properties. A pivotal breakthrough in this domain is documented in patent CN113262816A, which discloses a highly efficient catalytic system for synthesizing benzo[1,2-b:4,5-b']dibenzofuran, a critical scaffold for next-generation optoelectronic materials. This innovation addresses the longstanding challenges associated with constructing fused heterocyclic systems by employing a synergistic palladium and silver co-catalytic strategy. Unlike conventional routes that rely on tedious multi-step precursor synthesis, this method utilizes readily available 1,4-cyclohexanedione and o-dichlorobenzene in a direct, one-pot transformation. For R&D directors and procurement specialists alike, this represents a paradigm shift towards more sustainable and cost-effective manufacturing of high-value electronic chemical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of benzo[1,2-b:4,5-b']dibenzofuran derivatives has been plagued by inefficient synthetic routes that severely impact overall process economics and scalability. Traditional methodologies, such as those reported by Kawaguchi et al., typically necessitate the prior construction of complex polybiphenyl compounds via Suzuki coupling reactions. These multi-step sequences not only suffer from low atom utilization but also require the handling of sensitive organoboron reagents and expensive halogenated precursors. Furthermore, alternative approaches involving nucleophilic substitution under high-temperature conditions often result in darkened reaction mixtures due to thermal degradation, thereby complicating downstream purification and drastically reducing final product purity. The reliance on pre-functionalized starting materials inherently inflates raw material costs and extends lead times, creating significant bottlenecks for supply chain managers aiming to secure reliable sources of high-purity OLED material.
The Novel Approach
In stark contrast, the novel methodology outlined in the patent leverages a direct C-H activation strategy that bypasses the need for pre-synthesized biphenyl or fluorobenzene intermediates entirely. By utilizing 1,4-cyclohexanedione and o-dichlorobenzene as fundamental building blocks, the process achieves the construction of two furan ring fragments in a single operational step. This approach not only simplifies the synthetic workflow but also operates under remarkably mild conditions, typically at 100°C, which preserves the integrity of the molecular structure and minimizes side reactions. The dual role of o-dichlorobenzene as both a reactant and a solvent further streamlines the process engineering, reducing the volume of auxiliary solvents required and enhancing the overall green chemistry profile of the manufacturing route.
Mechanistic Insights into Pd-Ag Co-Catalyzed C-H Activation and Cyclization
The core innovation of this technology lies in the intricate interplay between the palladium and silver catalysts, which orchestrate a complex cascade of bond-forming events. The mechanism initiates with the reduction of the palladium(II) salt to active palladium(0) species by the phosphine ligand, followed by oxidative addition into the carbon-chlorine bond of o-dichlorobenzene. Simultaneously, the silver salt acts as a Lewis acid to activate the alpha-position C-H bond of the 1,4-cyclohexanedione, generating a reactive silver-carbon intermediate. This intermediate subsequently undergoes transmetallation with the aryl-palladium species, facilitating the critical carbon-carbon bond formation known as Suzuki-type coupling without the need for boronic acids. Following this, the silver cation promotes the formation of a phenoxide intermediate, which undergoes intramolecular cyclization to construct the first furan ring, setting the stage for the second cycle of activation and aromatization.
Furthermore, the precise control of impurity profiles is achieved through the specific selection of ligands and the balanced ratio of metal catalysts. The ligand, such as tri-tert-butylphosphine or bipyridine derivatives, serves to stabilize the low-valent palladium species and prevent aggregation into inactive palladium black, ensuring sustained catalytic turnover throughout the reaction duration. The synergistic effect of the silver salt in promoting beta-hydride elimination is crucial for the final aromatization step, converting the partially reduced intermediate into the fully conjugated benzo[1,2-b:4,5-b']dibenzofuran system. This mechanistic precision ensures that side products, such as partially cyclized species or dehalogenated byproducts, are minimized, resulting in crude product purities exceeding 99% as confirmed by HPLC analysis in the patent examples.
How to Synthesize Benzo[1,2-b:4,5-b']dibenzofuran Efficiently
Implementing this advanced synthetic route requires careful attention to the stoichiometric balance of the co-catalysts and the maintenance of an inert atmosphere to prevent ligand oxidation. The process is designed for operational simplicity, allowing for the simultaneous charging of all reagents including the ketone substrate, the aryl halide, and the catalytic system into a standard pressure-rated reactor. Detailed standard operating procedures regarding specific molar ratios, agitation speeds, and workup protocols are essential for reproducing the high yields reported in the patent literature, particularly when scaling from gram-scale laboratory experiments to pilot plant batches.
- Charge 1,4-cyclohexanedione, o-dichlorobenzene, palladium salt, silver salt, and ligand into a reactor under inert atmosphere.
- Heat the mixture to 100°C and maintain reaction for 6-10 hours with stirring to facilitate multi-step coupling.
- Recover solvent, extract with ethyl acetate/water, desolventize, and recrystallize from toluene to obtain high-purity product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this catalytic system offers transformative benefits that extend far beyond simple yield improvements. The elimination of pre-synthesis steps for complex precursors fundamentally alters the cost structure of the supply chain, removing the dependency on multiple external vendors for specialized starting materials. This vertical integration of the synthetic pathway significantly reduces the risk of supply disruptions and allows for greater flexibility in sourcing bulk commodity chemicals like 1,4-cyclohexanedione and o-dichlorobenzene, which are widely available in the global market. Consequently, this leads to substantial cost savings in electronic chemical manufacturing by compressing the value chain and minimizing logistics overhead associated with transporting hazardous intermediates.
- Cost Reduction in Manufacturing: The economic impact of this technology is driven primarily by the drastic reduction in step count and the high atom economy of the transformation. By avoiding the use of expensive organoboron reagents and eliminating the need for separate Suzuki coupling stages, the process significantly lowers the consumption of precious metal catalysts per kilogram of final product. Additionally, the ability to recover and recycle o-dichlorobenzene, which serves as the solvent, further enhances the process mass intensity metrics, driving down waste disposal costs and raw material expenditure without compromising on the quality of the final OLED intermediate.
- Enhanced Supply Chain Reliability: From a logistical perspective, the reliance on stable, shelf-stable commodity chemicals rather than moisture-sensitive or thermally unstable intermediates greatly enhances supply chain resilience. The robustness of the reaction conditions, operating at a moderate 100°C rather than extreme cryogenic or high-temperature regimes, reduces the technical barriers for contract manufacturing organizations (CMOs) to adopt the process. This accessibility ensures a more diversified supplier base and reduces lead time for high-purity electronic chemical intermediates, enabling faster response to market fluctuations in the display and lighting sectors.
- Scalability and Environmental Compliance: The environmental footprint of this synthesis is markedly lower than traditional methods, aligning with stringent global regulatory standards for chemical manufacturing. The one-pot nature of the reaction minimizes solvent usage and waste generation, while the absence of toxic tin or boron byproducts simplifies effluent treatment processes. This green chemistry profile facilitates easier permitting for commercial scale-up of complex polymer additives and OLED materials, ensuring long-term operational continuity and compliance with evolving environmental, social, and governance (ESG) mandates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Pd-Ag co-catalytic technology. These insights are derived directly from the experimental data and mechanistic studies presented in the patent documentation, providing a clear framework for evaluating the feasibility of this route for your specific production needs. Understanding these nuances is critical for making informed decisions about technology transfer and process optimization.
Q: What are the key advantages of the Pd-Ag co-catalytic system over traditional Suzuki coupling?
A: The Pd-Ag co-catalytic system eliminates the need for pre-synthesizing complex polybiphenyl or polyfluorobenzene precursors required in traditional Suzuki coupling. This one-pot approach utilizes commercially available 1,4-cyclohexanedione and o-dichlorobenzene, significantly improving atom utilization and reducing raw material costs while operating under milder conditions (100°C) compared to high-temperature nucleophilic substitutions.
Q: How does the silver salt contribute to the reaction mechanism?
A: The silver salt plays a critical role in activating the sp3 C-H bond at the alpha-position of the ketone (1,4-cyclohexanedione) to form a silver-carbon intermediate. This intermediate then undergoes transmetallation with the aryl-palladium species, facilitating the crucial C-C bond formation and subsequent beta-H elimination aromatization that constructs the furan rings efficiently.
Q: Is this process scalable for industrial production of OLED materials?
A: Yes, the process is highly scalable due to its simple operation and use of o-dichlorobenzene as both reactant and solvent. The mild reaction temperature of 100°C reduces energy consumption risks associated with high-temperature processes, and the straightforward workup involving solvent recovery and recrystallization supports robust commercial scale-up for electronic chemical manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Benzo[1,2-b:4,5-b']dibenzofuran Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of securing a stable and high-quality supply of advanced intermediates for the rapidly growing OLED market. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. We are committed to maintaining stringent purity specifications and utilizing rigorous QC labs to guarantee that every batch of benzo[1,2-b:4,5-b']dibenzofuran meets the exacting standards required for high-performance electronic applications.
We invite you to collaborate with us to leverage this cutting-edge synthetic technology for your product portfolio. Our experts are ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this efficient route can optimize your bottom line. Please contact our technical procurement team today to request specific COA data and comprehensive route feasibility assessments, and let us partner with you to drive innovation in the electronic materials sector.
