Advanced Manufacturing of Asenapine Intermediates: Technical Upgrades and Commercial Scalability
Advanced Manufacturing of Asenapine Intermediates: Technical Upgrades and Commercial Scalability
The pharmaceutical industry continuously demands more efficient and safer pathways for the production of critical active pharmaceutical ingredient (API) precursors. Patent CN102976929A introduces a significant methodological advancement in the synthesis of (4-chloro-2-phenoxyphenyl)acetic acid, a pivotal intermediate in the manufacturing of Asenapine, an atypical antipsychotic agent. This technical disclosure addresses long-standing challenges in process chemistry by optimizing reaction conditions to enhance yield while simultaneously mitigating operational hazards. For R&D directors and procurement strategists, understanding the nuances of this patented route is essential for evaluating supply chain resilience and cost structures. The innovation lies not merely in the chemical transformation but in the strategic substitution of reagents that alters the economic and safety profile of the entire manufacturing campaign. By shifting from hazardous, high-cost bases to commodity alkaline materials, the process demonstrates a clear commitment to sustainable and scalable chemical engineering. This report analyzes the technical merits of this approach and its implications for commercial procurement, offering a roadmap for integrating this superior synthetic route into global supply networks.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the diaryl ether backbone required for this intermediate relied heavily on the use of sodium hydride (NaH) as a strong base in aprotic solvents such as tetrahydrofuran (THF). While chemically effective, this conventional approach presents severe limitations for industrial application. Sodium hydride is pyrophoric, requiring stringent handling protocols, inert atmosphere gloveboxes, and specialized quenching procedures to prevent fire hazards. These safety requirements translate directly into elevated operational expenditures (OPEX) and increased insurance liabilities for manufacturing facilities. Furthermore, the necessity for anhydrous conditions and expensive aprotic solvents complicates the solvent recovery process, generating significant hazardous waste streams that require costly disposal. From a supply chain perspective, reliance on such dangerous reagents introduces fragility; any disruption in the supply of high-purity NaH or THF can halt production entirely. Additionally, the multi-step preparation of sodium phenolate prior to coupling adds time and complexity, reducing the overall throughput of the reactor train and limiting the ability to respond quickly to market demand fluctuations.
The Novel Approach
The methodology disclosed in the patent data fundamentally reengineers the etherification step by replacing sodium hydride with sodium hydroxide (NaOH) in the presence of copper powder. This substitution is transformative, as it allows the reaction to proceed under solvent-free or minimal solvent conditions at elevated temperatures ranging from 125°C to 130°C. By eliminating the need for pre-formation of sodium phenolate, the process condenses multiple operational steps into a single, streamlined reaction vessel. The use of NaOH, a commodity chemical, drastically reduces raw material costs and removes the pyrophoric risk profile associated with NaH. This novel approach not only simplifies the work-up procedure but also enhances the safety culture of the manufacturing plant, aligning with modern environmental, health, and safety (EHS) standards. The robustness of this copper-catalyzed system under nitrogen protection ensures consistent quality while allowing for the use of standard stainless steel reactors rather than specialized glass-lined or Hastelloy equipment required for highly corrosive or reactive alternatives. This shift represents a paradigm change from laboratory-scale feasibility to industrial-scale viability.
Mechanistic Insights into Cu-Catalyzed Etherification and Oxidative Hydrolysis
The core of this synthetic strategy involves a copper-mediated Ullmann-type coupling followed by a unique oxidative cleavage. In the first stage, 2,4-dichloroacetophenone reacts with phenol in the presence of copper powder and sodium hydroxide. The copper acts as a catalyst to facilitate the nucleophilic aromatic substitution, where the phenoxide ion, generated in situ, attacks the chlorinated aromatic ring. The high temperature (125°C to 130°C) provides the necessary activation energy to overcome the kinetic barrier of the aryl chloride bond cleavage. This mechanism is superior because it tolerates the presence of water generated from the neutralization of NaOH, which would typically deactivate stronger bases like NaH. The result is the formation of 1-(5-chloro-2-phenoxy-phenyl)-ethanone with high purity, eliminating the need for intermediate purification steps that often lead to yield loss. The crude product can be directly transferred to the next stage, maximizing atom economy and reducing solvent consumption.
The second stage involves the conversion of the acetyl group to the acetic acid moiety using sublimed sulfur and morpholine, followed by acid hydrolysis. This transformation is critical for establishing the correct side-chain functionality required for Asenapine. The reaction proceeds through the formation of a thiomorpholine intermediate, which is subsequently hydrolyzed under acidic conditions using concentrated hydrochloric acid and glacial acetic acid. This specific pathway avoids the use of harsh oxidizing agents like potassium permanganate or chromic acid, which generate heavy metal waste. Instead, the sulfur-morpholine system offers a cleaner profile with byproducts that are easier to manage. The hydrolysis step, conducted under reflux for 18 hours, ensures complete conversion of the intermediate to the final acid. The final isolation involves pH adjustment and crystallization, yielding a tan solid with high purity suitable for downstream coupling reactions.
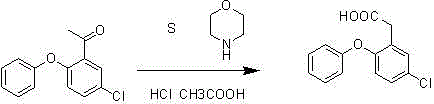
How to Synthesize (4-chloro-2-phenoxyphenyl)acetic acid Efficiently
Implementing this synthesis requires precise control over reaction parameters to ensure reproducibility and safety at scale. The process begins with the charging of 2,4-dichloroacetophenone, phenol, sodium hydroxide, and copper powder into a reactor under nitrogen protection. The mixture is heated to 125°C to 130°C and stirred for 10 hours to complete the etherification. Upon cooling, the crude ketone is extracted and carried forward without purification to maintain efficiency. The second phase involves refluxing the crude ketone with sublimed sulfur and morpholine for 5 hours. Finally, the mixture is subjected to acidic hydrolysis using HCl and acetic acid for 18 hours. Detailed standard operating procedures (SOPs) regarding addition rates, temperature ramping, and quenching protocols are essential for safe execution. The detailed standardized synthesis steps are provided in the guide below.
- Perform copper-catalyzed etherification of 2,4-dichloroacetophenone with phenol using sodium hydroxide at 125-130°C.
- React the resulting ketone intermediate with sublimed sulfur and morpholine under reflux conditions.
- Hydrolyze the reaction mixture using concentrated hydrochloric acid and glacial acetic acid to isolate the final acid product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this patented process offers tangible strategic benefits beyond mere chemical yield. The primary advantage lies in the drastic simplification of the raw material portfolio. By substituting specialized, hazardous reagents with commodity chemicals like sodium hydroxide and copper powder, the procurement team can leverage existing vendor relationships and bulk purchasing power to negotiate better pricing. This reduces the risk of supply disruption caused by the scarcity of niche reagents. Furthermore, the elimination of pyrophoric materials lowers the regulatory burden and insurance costs associated with storage and transport. The process design inherently supports continuous manufacturing or large-batch processing, which enhances supply continuity for downstream API production. These factors collectively contribute to a more resilient and cost-effective supply chain architecture.
- Cost Reduction in Manufacturing: The replacement of sodium hydride with sodium hydroxide results in substantial cost savings, as NaOH is a globally available commodity with a stable price point compared to the volatile market for specialized bases. Additionally, the ability to skip the purification of the intermediate ketone reduces solvent usage and labor hours, further driving down the cost of goods sold (COGS). The simplified work-up procedure minimizes waste treatment expenses, as there are no heavy metal oxidants or pyrophoric quench wastes to manage. These cumulative efficiencies allow for a more competitive pricing structure without compromising margin, providing a significant advantage in tender negotiations for API intermediates.
- Enhanced Supply Chain Reliability: Reliance on common reagents ensures that production is not bottlenecked by the availability of exotic chemicals. Sodium hydroxide and copper powder are produced in massive quantities globally, ensuring a steady supply even during market fluctuations. The robustness of the reaction conditions, which tolerate minor variations in moisture and temperature better than anhydrous NaH routes, reduces the rate of batch failures. This reliability translates to consistent on-time delivery performance for customers, strengthening long-term partnerships. The reduced safety risk also means fewer unplanned shutdowns due to safety incidents, ensuring a stable flow of material to the market.
- Scalability and Environmental Compliance: The process is inherently designed for scale-up, utilizing standard heating and stirring equipment available in most multipurpose chemical plants. The absence of cryogenic conditions or high-pressure requirements simplifies the engineering controls needed for expansion. From an environmental perspective, the avoidance of heavy metal oxidants and the reduction in solvent volume align with green chemistry principles. This facilitates easier compliance with increasingly stringent environmental regulations in key manufacturing regions. The waste stream is less hazardous, reducing disposal costs and environmental liability, making the facility more sustainable and attractive to eco-conscious partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These answers are derived directly from the patent specifications and practical engineering assessments of the process. Understanding these details is crucial for technical teams evaluating the feasibility of technology transfer. The responses highlight the safety, efficiency, and scalability aspects that differentiate this method from traditional approaches.
Q: Why is sodium hydroxide preferred over sodium hydride in this synthesis?
A: Sodium hydroxide eliminates the pyrophoric hazards associated with sodium hydride, significantly reducing safety risks and operational costs while maintaining high reaction efficiency.
Q: What is the role of sulfur and morpholine in the second step?
A: Sulfur and morpholine facilitate the oxidative transformation of the methyl ketone group, preparing the molecule for subsequent hydrolysis into the acetic acid derivative.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the use of commodity reagents like sodium hydroxide and the elimination of cryogenic or anhydrous requirements make this route highly scalable for industrial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (4-chloro-2-phenoxyphenyl)acetic acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the successful development of novel therapeutics like Asenapine. Our technical team has extensively analyzed the route disclosed in CN102976929A and possesses the expertise to implement this optimized process at an industrial scale. We have extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. Our facilities are equipped with stringent purity specifications and rigorous QC labs to guarantee that every batch of (4-chloro-2-phenoxyphenyl)acetic acid meets the highest international standards. We are committed to delivering not just a chemical product, but a reliable supply solution that supports your regulatory filings and commercial launch timelines.
We invite you to collaborate with us to optimize your supply chain for this critical intermediate. Our team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments. By partnering with NINGBO INNO PHARMCHEM, you gain access to a robust manufacturing platform dedicated to advancing your pharmaceutical projects with efficiency and integrity.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
