Advanced Catalytic Synthesis of Tolfenamic Acid for Commercial Scale-up and Cost Efficiency
The pharmaceutical industry continuously seeks robust synthetic routes for non-steroidal anti-inflammatory drugs (NSAIDs) that balance high purity with economic viability. A pivotal advancement in this domain is documented in Chinese Patent CN102786429A, which discloses a novel synthesis method for Tolfenamic Acid, a critical active pharmaceutical ingredient used extensively in both human and veterinary medicine for treating pain and inflammation. This patent addresses the longstanding inefficiencies of traditional manufacturing by introducing a streamlined process that utilizes methyl isobutyl ketone (MIBK) as a superior reaction medium. The structural integrity of the target molecule, 2-[(3-chloro-2-methylphenyl)amino]benzoic acid, relies on the precise formation of a diarylamine bond, a transformation that has historically been plagued by low conversion rates and difficult purification protocols. 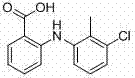 By shifting away from high-boiling polar aprotic solvents, this technology offers a pathway to significantly enhance the throughput and environmental profile of NSAID intermediate production, making it a highly attractive option for reliable pharmaceutical intermediate suppliers aiming to optimize their portfolio.
By shifting away from high-boiling polar aprotic solvents, this technology offers a pathway to significantly enhance the throughput and environmental profile of NSAID intermediate production, making it a highly attractive option for reliable pharmaceutical intermediate suppliers aiming to optimize their portfolio.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to the innovations described in CN102786429A, the prevailing industrial standard for synthesizing Tolfenamic Acid relied heavily on N,N-dimethylformamide (DMF) as the primary solvent. While DMF is a potent solvent for many organic transformations, its application in this specific Ullmann-type condensation presents severe operational bottlenecks. The process typically involves heating o-chlorobenzoic acid and 3-chloro-2-methylaniline in DMF, followed by the addition of a base like potassium carbonate. The critical failure point of this legacy route lies in the post-reaction workup; because DMF has a high boiling point and is miscible with water, it cannot be easily removed by simple washing or phase separation. Consequently, manufacturers are forced to evaporate the solvent to dryness under vacuum, a highly energy-intensive step that often leads to thermal degradation of the product and the co-precipitation of inorganic salts and side products. This results in a crude product of very low purity, necessitating multiple, laborious recrystallization steps—often involving the formation and subsequent acidification of sodium salts—to achieve acceptable quality. Historical data indicates that these cumbersome procedures cap the overall yield at a dismal level of approximately 38%, representing a massive loss of raw materials and a significant burden on waste management systems due to the toxicity of DMF residues.
The Novel Approach
The methodology introduced in the patent fundamentally re-engineers the reaction environment to overcome these thermodynamic and kinetic barriers. By substituting DMF with methyl isobutyl ketone (MIBK), the process leverages a solvent with a lower boiling point and, crucially, limited water solubility. This physical property change allows for a dramatic simplification of the isolation procedure. Instead of energy-heavy distillation, the reaction mixture can be treated directly with water to effect a liquid-liquid extraction. The product, existing as a salt in the basic aqueous phase or precipitating upon acidification, is cleanly separated from organic impurities and the catalyst residue which remain in the organic phase or are filtered off. 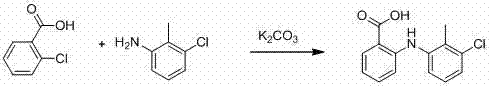 Furthermore, the patent describes a sequential addition protocol where the acid and base are pre-reacted before the introduction of the amine and copper catalyst. This controlled addition minimizes side reactions and ensures optimal activation of the nucleophile. The result is a robust process that consistently delivers yields ranging from 71% to 82% across various examples, more than doubling the efficiency of the conventional DMF route while drastically reducing the complexity of the downstream purification train.
Furthermore, the patent describes a sequential addition protocol where the acid and base are pre-reacted before the introduction of the amine and copper catalyst. This controlled addition minimizes side reactions and ensures optimal activation of the nucleophile. The result is a robust process that consistently delivers yields ranging from 71% to 82% across various examples, more than doubling the efficiency of the conventional DMF route while drastically reducing the complexity of the downstream purification train.
Mechanistic Insights into Copper-Catalyzed Ullmann Condensation
The core chemical transformation in this synthesis is a copper-catalyzed nucleophilic aromatic substitution, often referred to as an Ullmann condensation, which forms the C-N bond between the benzoic acid derivative and the aniline. In the MIBK system, the initial step involves the deprotonation of o-chlorobenzoic acid by an alkali metal hydroxide, such as sodium hydroxide or potassium hydroxide, generating a carboxylate salt that is soluble in the reaction medium. Upon the addition of the copper catalyst—selected from cuprous bromide, cuprous chloride, copper powder, or copper acetate—and the acid-binding agent, the catalytic cycle initiates. The copper species likely undergoes oxidative addition into the carbon-chlorine bond of the o-chlorobenzoic acid derivative, forming an organocopper intermediate. Simultaneously, the 3-chloro-2-methylaniline coordinates with the metal center. The presence of the acid-binding agent, such as sodium acetate or sodium bicarbonate, is critical for scavenging the hydrogen chloride generated during the coupling, thereby driving the equilibrium forward and preventing the protonation of the amine nucleophile which would render it inactive. The subsequent reductive elimination step releases the Tolfenamic Acid product and regenerates the active copper catalyst. This mechanism is highly sensitive to the reaction temperature, with the patent specifying a range from 90°C to reflux, ensuring sufficient energy to overcome the activation barrier of the C-Cl bond cleavage without promoting excessive decomposition.
Impurity control in this system is intrinsically linked to the choice of solvent and the workup strategy. In traditional DMF processes, the inability to wash away inorganic byproducts leads to 'salt trapping' within the crystal lattice of the product. In contrast, the MIBK-water biphasic system acts as a continuous purification engine. During the extraction phase, water-soluble impurities, including excess base, acid-binding agent salts, and polar side products, are partitioned into the aqueous layer, while the organic layer retains non-polar impurities. When the aqueous phase containing the product salt is acidified with hydrochloric acid, the Tolfenamic Acid precipitates in a highly pure form. The final recrystallization from an ethanol/water mixture further refines the crystal habit and removes trace organic contaminants. This multi-stage purification, facilitated by the solvent properties, ensures that the final API meets stringent purity specifications required for regulatory approval, minimizing the risk of genotoxic impurities or heavy metal residues that often plague copper-catalyzed reactions.
How to Synthesize Tolfenamic Acid Efficiently
Implementing this synthesis route requires precise adherence to the stoichiometric ratios and thermal profiles outlined in the patent to maximize the economic benefits. The process begins with the formation of the carboxylate salt in MIBK, followed by the careful introduction of the amine and catalyst system under reflux conditions. The reaction time is flexible, ranging from 3 to 12 hours depending on the specific catalyst loading and temperature, allowing process engineers to tune the cycle time for their specific reactor capabilities. Following the reaction, the phase separation and acidification steps must be controlled to ensure complete precipitation of the product. For a detailed breakdown of the specific reagent quantities, molar ratios, and operational parameters validated in the patent examples, please refer to the standardized synthesis guide below.
- Preparation of Reaction Mixture: Dissolve o-chlorobenzoic acid and an alkali metal hydroxide (such as sodium hydroxide) in methyl isobutyl ketone (MIBK) and heat to initiate the reaction.
- Catalytic Coupling: Introduce 3-chloro-2-methylaniline, an acid-binding agent (e.g., sodium acetate), and a copper catalyst to the mixture, then maintain heating at reflux temperatures to facilitate C-N bond formation.
- Workup and Purification: Cool the reaction, perform aqueous extraction to separate phases, acidify the aqueous layer to precipitate the crude product, and finally recrystallize using an ethanol/water system to obtain refined Tolfenamic Acid.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition from the legacy DMF process to this MIBK-based technology represents a strategic opportunity to de-risk the supply of Tolfenamic Acid while simultaneously driving down the cost of goods sold (COGS). The primary driver of cost reduction in pharmaceutical intermediate manufacturing is often the yield and the complexity of the workup. By eliminating the need for high-vacuum solvent stripping and multiple salt-formation cycles, this new method significantly reduces utility consumption (steam and electricity) and labor hours per batch. The qualitative shift from a 38% yield process to one consistently achieving over 80% yield effectively doubles the output capacity of existing infrastructure without requiring capital expenditure on new reactors. This substantial increase in throughput directly translates to lower unit costs, allowing suppliers to offer more competitive pricing structures to downstream API manufacturers. Furthermore, the reduction in solvent usage and the switch to a less toxic solvent system lowers the environmental compliance burden, reducing waste disposal fees and mitigating regulatory risks associated with Class 2 solvents like DMF.
- Cost Reduction in Manufacturing: The elimination of the solvent evaporation step is a major economic win. In the old process, removing high-boiling DMF requires significant energy input and extended equipment occupancy time. By switching to MIBK, which allows for aqueous extraction, the cycle time per batch is drastically shortened. This increased asset turnover means that the same production facility can produce significantly more tons of product annually. Additionally, the higher yield implies that for every ton of raw material purchased, nearly double the amount of finished product is generated compared to the old method, effectively halving the raw material cost contribution per kilogram of final API. The simplified purification also reduces the consumption of recrystallization solvents and acids, further compounding the savings.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by the complexity of synthesis; processes with low yields and difficult purifications are prone to batch failures and delays. The robustness of the MIBK method, with its forgiving workup and high conversion rates, ensures a more predictable production schedule. The raw materials required—o-chlorobenzoic acid, 3-chloro-2-methylaniline, and common copper salts—are commodity chemicals with stable global supply chains, reducing the risk of raw material shortages. The ability to produce high-purity material in fewer steps also reduces the lead time for high-purity pharmaceutical intermediates, enabling faster response to market demand spikes for Tolfenamic Acid formulations in both human and veterinary sectors.
- Scalability and Environmental Compliance: Scaling chemical processes often amplifies safety and environmental issues. The legacy DMF route poses significant occupational health risks due to the reproductive toxicity of DMF and the potential for thermal runaway during vacuum distillation. The new method operates at atmospheric pressure during the workup and uses a solvent with a more favorable safety profile. This makes the commercial scale-up of complex pharmaceutical intermediates much safer and easier to permit. The reduction in hazardous waste generation aligns with modern green chemistry principles, helping pharmaceutical companies meet their sustainability goals and reducing the long-term liability associated with hazardous waste disposal.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and technical disclosures within patent CN102786429A, providing a factual basis for evaluating the feasibility of this route for your specific manufacturing needs. Understanding these details is crucial for R&D teams assessing technology transfer and procurement teams negotiating supply agreements.
Q: How does the MIBK solvent system improve yield compared to traditional DMF methods?
A: The patented MIBK system eliminates the need for evaporating high-boiling toxic solvents like DMF. By utilizing a biphasic extraction workup, impurities are effectively removed before crystallization, boosting yields from approximately 38% in older methods to over 80% in this optimized process.
Q: What specific catalysts are effective for this Tolfenamic Acid synthesis?
A: The patent specifies that various copper sources are effective, including cuprous bromide, cuprous chloride, copper powder, and copper acetate. These catalysts facilitate the critical Ullmann-type coupling between the benzoic acid derivative and the aniline.
Q: Is this synthesis method suitable for large-scale industrial production?
A: Yes, the method is explicitly designed for industrial scalability. It uses readily available reagents, avoids complex vacuum distillation steps required for DMF removal, and employs standard extraction and recrystallization techniques that are easily managed in multi-ton reactors.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tolfenamic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize that the adoption of advanced synthetic methodologies like the one described in CN102786429A is essential for maintaining competitiveness in the global pharmaceutical market. As a leading CDMO and supplier, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the laboratory efficiencies of this MIBK-based process are faithfully translated to industrial reality. Our facilities are equipped with state-of-the-art reactors capable of handling the specific thermal and mixing requirements of copper-catalyzed couplings, and our rigorous QC labs enforce stringent purity specifications to guarantee that every batch of Tolfenamic Acid meets the highest international standards. We understand that consistency is key for API manufacturers, and our process controls are designed to minimize batch-to-batch variability.
We invite you to collaborate with us to leverage this cost-effective and environmentally superior synthesis route. Our technical team is prepared to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating exactly how this technology can improve your margins. We encourage potential partners to contact our technical procurement team to request specific COA data from our pilot batches and to discuss route feasibility assessments for your broader portfolio. By partnering with NINGBO INNO PHARMCHEM, you secure not just a supplier, but a strategic ally committed to innovation, quality, and supply chain resilience in the production of critical pharmaceutical intermediates.
