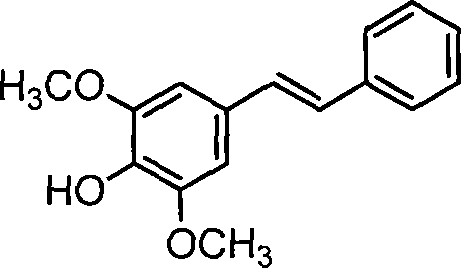
Advanced Solvent-Free Synthesis of (E)-4-Hydroxy-3,5-Dimethoxystilbene for Commercial Scale-Up
The pharmaceutical and fine chemical industries are constantly seeking more efficient pathways to synthesize bioactive stilbene derivatives, which serve as critical scaffolds for drug development and agricultural applications. Patent CN101225027A introduces a groundbreaking methodology for the preparation of (E)-4-hydroxy-3,5-dimethoxystilbene, a compound of significant interest due to its structural similarity to resveratrol and other polyhydroxystilbenes known for their antioxidant and antitumor properties. This patent details a novel one-pot reaction that utilizes Lewis acid catalysis promoted by metal zinc powder, operating under solvent-free conditions at room temperature to achieve yields exceeding 70%. The strategic elimination of organic solvents and protection groups represents a paradigm shift in green chemistry, addressing both environmental concerns and production costs simultaneously. For R&D directors and procurement specialists, this technology offers a robust alternative to traditional multi-step syntheses that often suffer from low efficiency and high waste generation. The ability to directly convert the carbonyl group of 4-hydroxy-3,5-dimethoxybenzaldehyde into a carbon-carbon double bond with high stereoselectivity is a major technical breakthrough.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of polyhydroxystilbene compounds has been plagued by complex multi-step procedures that hinder industrial scalability and economic viability. Traditional routes, such as those described by Manas and Pleixats or Erik and Merritt, typically rely on Wittig or Wittig-Horner reactions which necessitate the use of protecting groups for phenolic hydroxyls to prevent side reactions. These protection and deprotection steps not only extend the reaction timeline but also introduce additional reagents and solvents that increase the overall cost of goods sold and environmental footprint. Furthermore, methods involving Heck reactions or Perkin condensations often struggle with poor stereoselectivity, yielding mixtures of cis and trans isomers that require energy-intensive purification or isomerization processes to isolate the desired bioactive trans-form. The reliance on large volumes of organic solvents in these conventional methods poses significant safety hazards and waste disposal challenges, making them less attractive for modern sustainable manufacturing initiatives. Consequently, the low overall yields and high operational complexity of these legacy methods have limited the widespread commercial availability of high-purity stilbene intermediates.
The Novel Approach
In stark contrast to these cumbersome legacy protocols, the method disclosed in CN101225027A streamlines the synthesis into a single, efficient step that bypasses the need for functional group protection entirely. By employing activated zinc powder in conjunction with a Lewis acid such as trimethylchlorosilane, the reaction facilitates the direct coupling of 4-hydroxy-3,5-dimethoxybenzaldehyde with benzyl bromide under mild conditions. This solvent-free approach not only simplifies the workup procedure by eliminating solvent removal steps but also drastically reduces the volume of chemical waste generated per kilogram of product. The reaction proceeds at room temperature, which significantly lowers energy consumption compared to processes requiring reflux or cryogenic conditions. Moreover, the inherent chemoselectivity of this zinc-promoted system ensures that the phenolic hydroxyl groups remain intact throughout the transformation, preserving the structural integrity required for downstream biological activity. This novel approach effectively resolves the bottlenecks of low yield and poor stereoselectivity, providing a scalable solution that aligns with the principles of green chemistry and cost-effective manufacturing.
Mechanistic Insights into Zinc-Promoted Lewis Acid Catalysis
The core of this technological advancement lies in the unique mechanistic pathway enabled by the synergy between activated zinc powder and the Lewis acid catalyst. The zinc powder acts as a single-electron reductant, facilitating the formation of organozinc intermediates that are crucial for the carbon-carbon bond formation without the need for pre-formed phosphonium salts typical of Wittig chemistry. The Lewis acid, specifically trimethylchlorosilane, plays a pivotal role in activating the carbonyl group of the aldehyde substrate, making it more susceptible to nucleophilic attack while simultaneously stabilizing the transition state. This activation allows the reaction to proceed smoothly at room temperature, avoiding the thermal degradation of sensitive functional groups that can occur under harsher conditions. The mechanism ensures that the newly formed carbon-carbon double bond adopts the thermodynamically more stable trans-configuration, resulting in high stereoselectivity without the need for post-reaction isomerization. Understanding this mechanistic nuance is vital for R&D teams aiming to replicate or optimize the process for specific scale-up requirements, as the activation state of the zinc powder is a critical parameter for success.
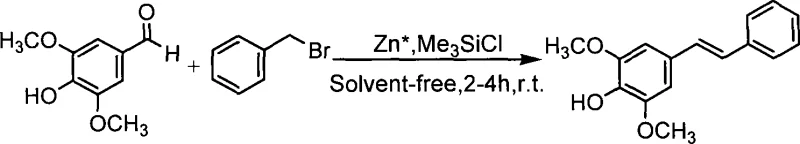
Furthermore, the impurity profile of the final product is significantly improved due to the high chemoselectivity of this catalytic system. In traditional methods, side reactions involving the protecting groups or incomplete deprotection often lead to complex impurity spectra that are difficult to separate, compromising the purity required for pharmaceutical applications. In this zinc-promoted protocol, the absence of protecting groups eliminates a major source of potential byproducts, while the mild reaction conditions minimize the formation of thermal degradation products. The direct conversion of the carbonyl group ensures that the functional groups on the substrate are fully retained and incorporated into the addition product, maintaining the desired substitution pattern on the aromatic rings. This clean reaction profile simplifies the downstream purification process, often allowing for high-purity isolation through standard column chromatography or crystallization techniques. For quality control teams, this translates to a more consistent product with a well-defined impurity spectrum, facilitating easier regulatory compliance and batch release.
How to Synthesize (E)-4-Hydroxy-3,5-Dimethoxystilbene Efficiently
Implementing this synthesis route requires careful attention to the activation of the zinc powder and the stoichiometric balance of the reagents to ensure optimal yield and selectivity. The process begins with the activation of commercial zinc powder using dilute hydrochloric acid to remove surface oxides, followed by thorough washing with water and organic solvents to eliminate inorganic salts and moisture. Once activated, the zinc powder is combined with 4-hydroxy-3,5-dimethoxybenzaldehyde in a dry reactor under an inert nitrogen atmosphere to prevent oxidation. The detailed standardized synthesis steps, including specific molar ratios and addition rates for the Lewis acid and benzyl bromide, are outlined in the guide below to ensure reproducibility and safety during scale-up operations.
- Activate zinc powder by washing with dilute hydrochloric acid and organic solvents, followed by vacuum drying.
- React 4-hydroxy-3,5-dimethoxybenzaldehyde with activated zinc powder and benzyl bromide under nitrogen protection.
- Add trimethylchlorosilane gradually, stir at room temperature for 2-4 hours, and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this solvent-free zinc-catalyzed methodology offers substantial advantages that directly impact the bottom line and supply chain resilience for chemical manufacturers. The elimination of organic solvents not only reduces the raw material costs associated with purchasing and recovering these volatile compounds but also significantly lowers the expenses related to waste disposal and environmental compliance. By operating at room temperature, the process drastically reduces energy consumption compared to traditional heating or cooling-intensive reactions, leading to lower utility costs per unit of production. These operational efficiencies translate into a more competitive cost structure, allowing suppliers to offer high-purity intermediates at more attractive price points without compromising on quality or margin. Additionally, the simplified workflow reduces the manpower and time required for each batch, enhancing overall production throughput and responsiveness to market demand fluctuations.
- Cost Reduction in Manufacturing: The removal of protection and deprotection steps fundamentally alters the cost equation by reducing the number of unit operations and the quantity of reagents consumed per kilogram of product. Without the need for expensive protecting group reagents and the solvents required to handle them, the direct material costs are significantly lowered, creating a leaner manufacturing process. The high yield reported in the patent, exceeding 70%, further amplifies these savings by maximizing the output from a fixed amount of starting material, reducing the effective cost of goods sold. This efficiency allows for a more robust pricing strategy in the competitive pharmaceutical intermediate market, where margin pressure is often high. The qualitative reduction in process complexity also minimizes the risk of batch failures, ensuring more consistent financial performance.
- Enhanced Supply Chain Reliability: The use of readily available and inexpensive reagents such as zinc powder and benzyl bromide ensures a stable supply chain that is less susceptible to the volatility often seen with specialized catalysts or exotic reagents. The solvent-free nature of the reaction reduces the logistical burden and safety risks associated with transporting and storing large volumes of flammable organic solvents, simplifying warehouse management and regulatory compliance. This simplicity enhances the reliability of supply, as the production process is less dependent on complex infrastructure or specialized equipment that could become bottlenecks. For supply chain heads, this means a more predictable lead time and a reduced risk of production stoppages due to reagent shortages or safety incidents. The robustness of the chemistry supports continuous or large-batch production schedules, ensuring consistent availability for downstream customers.
- Scalability and Environmental Compliance: The green chemistry attributes of this process, specifically the absence of solvent waste and the use of non-toxic zinc, align perfectly with increasingly stringent environmental regulations and corporate sustainability goals. Scaling up a solvent-free reaction is inherently safer and easier than managing large volumes of volatile organic compounds, reducing the capital expenditure required for explosion-proof facilities and solvent recovery systems. The reduced environmental footprint simplifies the permitting process for new production lines and minimizes the risk of regulatory fines or shutdowns related to emissions. This environmental compliance is not just a regulatory necessity but a market differentiator that appeals to eco-conscious pharmaceutical partners. The ease of scale-up ensures that the transition from laboratory grams to commercial tons can be achieved with minimal process re-engineering, accelerating time-to-market.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this novel synthesis technology. These answers are derived directly from the technical specifications and advantageous effects detailed in the patent documentation, providing clarity for stakeholders evaluating this method for adoption. Understanding these nuances is essential for making informed decisions about process integration and supplier selection in the competitive fine chemical landscape.
Q: What are the primary advantages of the zinc-promoted method over traditional Wittig reactions?
A: The zinc-promoted method eliminates the need for protecting groups and organic solvents, significantly reducing waste and operational complexity compared to multi-step Wittig or Heck protocols.
Q: How does this process ensure high stereoselectivity for the trans-isomer?
A: The reaction mechanism inherently favors the formation of the trans-configuration carbon-carbon double bond, achieving high stereoselectivity without requiring post-reaction isomerization steps.
Q: Is this synthesis method suitable for large-scale industrial production?
A: Yes, the solvent-free nature and mild room temperature conditions make it highly scalable, reducing energy consumption and safety risks associated with volatile organic compounds.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (E)-4-Hydroxy-3,5-Dimethoxystilbene Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and sustainable synthesis routes in the development of next-generation pharmaceutical intermediates. Our team of expert chemists has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods like the one described in CN101225027A can be successfully translated into robust industrial processes. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of (E)-4-hydroxy-3,5-dimethoxystilbene meets the exacting standards required for drug substance manufacturing. Our commitment to technical excellence allows us to navigate the complexities of solvent-free chemistry and zinc-promoted reactions, delivering consistent quality that supports your R&D and commercialization timelines.
We invite you to collaborate with us to leverage this advanced technology for your specific project needs, ensuring a competitive edge in your supply chain. Please contact our technical procurement team to request a Customized Cost-Saving Analysis that quantifies the potential economic benefits of switching to this greener synthesis route for your operations. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can support your long-term strategic goals. Let us partner with you to bring high-quality, cost-effective stilbene intermediates to the market efficiently.
