Advanced Metal-Free Synthesis of Chiral Pyrazolone Intermediates for Commercial Scale-Up
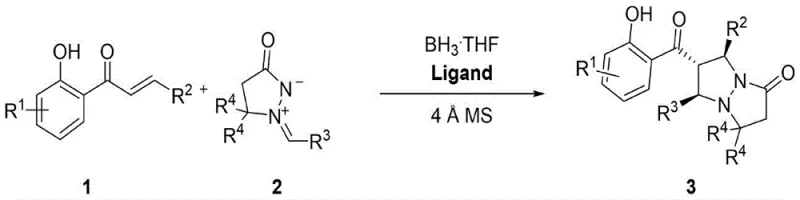
The pharmaceutical and fine chemical industries are constantly seeking more efficient, sustainable, and cost-effective routes to synthesize complex chiral heterocycles, which serve as critical scaffolds in drug discovery. Patent CN115286635A introduces a groundbreaking methodology for the synthesis of optically active cyclic pyrazolone compounds via an asymmetric 1,3-dipolar cycloaddition reaction. This technology represents a significant leap forward in organic synthesis, specifically addressing the limitations of traditional transition-metal-catalyzed processes. By utilizing 2'-hydroxy-α,β-unsaturated ketone compounds and N,N'-cyclomethanimine imines as raw materials, the invention achieves high-yield production of chiral pyrazolone derivatives in the presence of chiral binaphthol ligands, borane tetrahydrofuran, and molecular sieves. The strategic shift away from heavy metals towards an organoboron catalytic system not only enhances the environmental profile of the synthesis but also streamlines the purification workflow, making it an ideal candidate for reliable pharmaceutical intermediate supplier operations aiming for high-purity outputs.
Pyrazolone derivatives are ubiquitous in medicinal chemistry, forming the core structure of well-known antipyretic and analgesic drugs such as aminopyrine and phenylbutazone. Furthermore, their fused cyclic variants exhibit remarkable biological activities, including antipyramidal and antibacterial properties, making them highly valuable targets for new drug development. However, accessing these structures with precise stereochemical control has historically been challenging. The background art cited in the patent reveals that previous successful syntheses relied heavily on chiral nickel or rhodium complexes. While effective, these transition metal catalysts pose significant drawbacks for large-scale manufacturing, including high costs, potential toxicity, and the stringent regulatory requirements for residual metal limits in final API products. The novel approach disclosed in CN115286635A circumvents these issues entirely by employing a metal-free catalytic system that delivers exceptional enantioselectivity and diastereoselectivity under mild reaction conditions.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of chiral pyrazolopyrazolone frameworks has depended on the use of precious transition metal catalysts, which introduce multiple bottlenecks in the supply chain and production economics. As noted in the prior art, methods developed by research groups such as Suga et al. and Kang utilized chiral nickel and rhodium complexes respectively to catalyze the cycloaddition of unsaturated carbonyls with imines. While these academic precedents demonstrated feasibility, they are ill-suited for cost reduction in pharmaceutical intermediate manufacturing due to the exorbitant price of rhodium and the toxicity concerns associated with nickel. Furthermore, the removal of trace transition metals from the final product requires additional purification steps, such as specialized scavenging resins or extensive recrystallization, which drastically reduces overall process efficiency and increases waste generation. These factors collectively hinder the commercial scale-up of complex pharmaceutical intermediates, creating a demand for alternative catalytic systems that are both economically viable and environmentally benign.
The Novel Approach
The invention described in patent CN115286635A offers a transformative solution by replacing transition metals with a borane-based catalytic system supported by chiral organic ligands. This method utilizes easily accessible 2'-hydroxy-α,β-unsaturated ketones and N,N'-cyclomethanimine imines, reacting them in the presence of chiral binaphthol ligands (such as the iodine-substituted L4 ligand), borane tetrahydrofuran (BH3·THF), and 4Å molecular sieves. The reaction proceeds with remarkable efficiency, often achieving isolated yields exceeding 96% and enantiomeric excess (ee) values greater than 99%. The use of mild solvents like methyl tert-butyl ether (MTBE) and moderate temperatures (25°C to 60°C) further underscores the practicality of this route. By eliminating the need for expensive heavy metals, this novel approach not only simplifies the post-reaction workup but also ensures that the resulting high-purity OLED material or pharmaceutical precursor meets stringent quality specifications without the burden of metal contamination.
Mechanistic Insights into Borane-Catalyzed Asymmetric 1,3-Dipolar Cycloaddition
The core of this technological advancement lies in the unique activation mode provided by the borane-ligand complex. In this catalytic cycle, the borane tetrahydrofuran acts as a Lewis acid, coordinating with the carbonyl oxygen of the 2'-hydroxy-α,β-unsaturated ketone to increase its electrophilicity. Simultaneously, the chiral binaphthol ligand creates a sterically defined environment that directs the approach of the nucleophilic N,N'-cyclomethanimine dipole. This dual activation strategy ensures that the cycloaddition occurs with high regioselectivity and stereocontrol. The molecular sieves play a crucial role in sequestering trace water, which could otherwise deactivate the borane catalyst or hydrolyze the sensitive imine substrate. The result is a highly concerted reaction pathway that constructs two new stereocenters and a fused ring system in a single operational step. This level of atomic economy and stereochemical precision is critical for reducing lead time for high-purity pharmaceutical intermediates, as it minimizes the need for downstream resolution or correction of stereoisomers.
Furthermore, the impurity profile of this reaction is exceptionally clean due to the high diastereoselectivity observed. The patent data indicates diastereomeric ratios (dr) often exceeding 30:1 and up to >99:1 under optimized conditions. This high selectivity means that the formation of unwanted diastereomers is suppressed at the source, rather than being removed later. For R&D directors, this implies a more predictable impurity spectrum, facilitating faster regulatory filing and validation. The robustness of the catalyst system is also evident in its tolerance to various functional groups on the aromatic rings of the substrates, including halogens, alkyl groups, and electron-withdrawing trifluoromethyl groups. This substrate versatility allows for the rapid generation of diverse compound libraries, accelerating the drug discovery process while maintaining a consistent and reliable synthetic platform.
How to Synthesize Chiral Pyrazolone Efficiently
The operational simplicity of this synthesis makes it highly attractive for process chemists looking to implement new routes quickly. The standard procedure involves charging a reaction vessel with the chiral ligand, borane source, and molecular sieves under an inert nitrogen atmosphere, followed by the addition of the ketone substrate in anhydrous MTBE. After a brief pre-stirring period to form the active catalytic species, the imine reactant is introduced, and the mixture is allowed to stir at ambient or slightly elevated temperatures. The reaction progress is easily monitored by thin-layer chromatography (TLC), and upon completion, the product is isolated via standard silica gel chromatography. For detailed standardized synthesis steps and specific molar ratios, please refer to the guide below.
- Prepare the reaction mixture by combining 2'-hydroxy-α,β-unsaturated ketone, N,N'-cyclomethanimine, chiral ligand (e.g., L4), and BH3·THF in anhydrous MTBE with 4Å molecular sieves under inert atmosphere.
- Stir the reaction mixture at mild temperatures ranging from 25°C to 60°C for 12 to 24 hours until the starting imine is fully consumed as monitored by TLC.
- Remove the solvent under reduced pressure and purify the crude residue via flash silica gel column chromatography to isolate the high-purity chiral pyrazolone product.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this metal-free synthesis route offers profound strategic benefits that extend beyond mere chemical elegance. The elimination of precious transition metals like rhodium and nickel directly translates to significant cost savings in raw material procurement, shielding the supply chain from the volatility of the precious metals market. Moreover, the simplified purification process reduces the consumption of solvents and stationary phases, lowering the overall cost of goods sold (COGS). The ability to recycle the chiral ligands, as highlighted in the patent's beneficial effects, further enhances the economic viability of the process, allowing for substantial cost savings over long production runs. This efficiency is crucial for maintaining competitive pricing in the global market for fine chemical intermediates.
- Cost Reduction in Manufacturing: The replacement of expensive transition metal catalysts with organoboron reagents and recoverable organic ligands fundamentally alters the cost structure of production. By removing the need for costly metal scavengers and extensive metal testing, manufacturers can achieve drastic simplification of the downstream processing workflow. This qualitative improvement in process efficiency leads to substantial cost savings, making the production of complex chiral scaffolds more economically accessible for generic drug manufacturers and specialty chemical producers alike.
- Enhanced Supply Chain Reliability: The reliance on readily available starting materials such as substituted chalcones and simple imines ensures a robust and resilient supply chain. Unlike specialized metal catalysts which may face supply constraints or long lead times, the reagents for this process are commodity chemicals available from multiple global vendors. This diversification of the supply base mitigates the risk of production stoppages and ensures consistent delivery schedules, which is vital for meeting the just-in-time demands of modern pharmaceutical manufacturing.
- Scalability and Environmental Compliance: The mild reaction conditions and the use of greener solvents like MTBE facilitate easy scale-up from laboratory to pilot and commercial plant scales without the need for specialized high-pressure or cryogenic equipment. Additionally, the absence of heavy metals simplifies waste treatment and disposal, ensuring compliance with increasingly stringent environmental regulations. This environmental compatibility not only reduces regulatory risk but also aligns with the sustainability goals of major multinational corporations, enhancing the marketability of the final products.
Frequently Asked Questions (FAQ)
To assist technical decision-makers in evaluating this technology, we have compiled answers to common questions regarding the implementation and benefits of this synthesis method. These insights are derived directly from the technical specifications and experimental data provided in the patent documentation, ensuring accuracy and relevance for process development teams.
Q: What are the advantages of this borane-catalyzed method over traditional transition metal catalysis?
A: Unlike previous methods requiring expensive and toxic nickel or rhodium catalysts, this protocol utilizes organoboron catalysis with chiral ligands. This eliminates the need for rigorous heavy metal removal steps, significantly simplifying downstream processing and ensuring higher product purity suitable for pharmaceutical applications.
Q: Is this synthesis method scalable for industrial production?
A: Yes, the reaction operates under mild conditions (25-60°C) using readily available solvents like MTBE. The patent demonstrates high yields (up to 99%) and excellent stereoselectivity across a broad substrate scope, indicating robust feasibility for commercial scale-up from kilogram to tonnage levels.
Q: Can the chiral ligands be recovered and reused?
A: The patent explicitly highlights that the chiral ligands employed in this asymmetric cycloaddition are recyclable. This recoverability contributes to substantial cost reductions in manufacturing by minimizing the consumption of high-value chiral auxiliaries.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Pyrazolone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the synthetic methodologies disclosed in CN115286635A for the production of high-value chiral intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive consistent quality regardless of order size. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including chiral HPLC analysis to guarantee the high enantiomeric excess required for advanced pharmaceutical applications. We are committed to leveraging this metal-free technology to deliver superior products that meet the evolving needs of the global healthcare industry.
We invite procurement leaders and R&D directors to collaborate with us to explore how this innovative synthesis route can optimize your supply chain. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific project requirements. We encourage you to reach out today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that drive efficiency and innovation in your drug development pipeline.
