Advancing Pharmaceutical Intermediates: Scalable Copper-Catalyzed Synthesis of Tri-substituted Alkenyl Borates
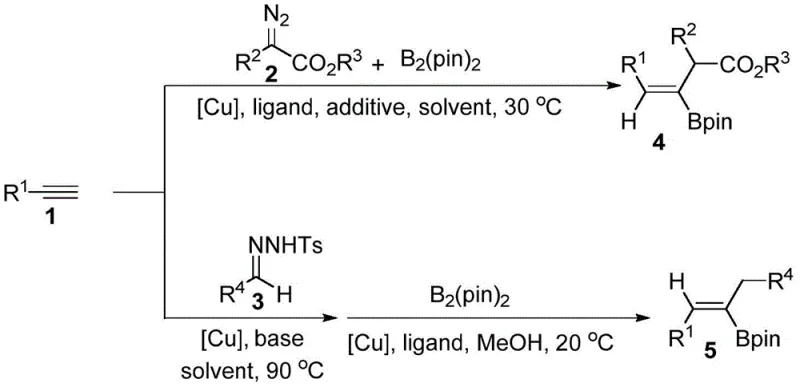
The landscape of organic synthesis for complex pharmaceutical intermediates is continually evolving, driven by the need for more efficient, selective, and cost-effective methodologies. A significant breakthrough in this domain is documented in patent CN113173939B, which discloses a robust method for synthesizing tri-substituted alkenyl borates through a copper-catalyzed three-component reaction. This technology represents a paradigm shift from traditional multi-step sequences, offering a streamlined one-pot approach that directly functionalizes terminal alkynes with high stereoselectivity. By leveraging earth-abundant copper catalysts rather than expensive precious metals, this process addresses critical economic and sustainability concerns in modern chemical manufacturing. The ability to selectively produce either (Z)- or (E)-isomers simply by altering the carbene precursor and ligand system provides unparalleled flexibility for medicinal chemists designing complex molecular architectures. As a leading player in the fine chemical sector, understanding and adopting such innovations is crucial for maintaining a competitive edge in the global supply chain.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of trisubstituted alkenyl boronates has been fraught with challenges related to regioselectivity and stereocontrol. Conventional methods often rely on the metal-catalyzed borylation of pre-formed allenes or alkynes, which frequently suffer from poor selectivity or require harsh reaction conditions that limit substrate tolerance. Furthermore, many existing protocols necessitate the isolation of reactive intermediates, such as allenes, prior to the boronation step. This multi-step approach not only increases the overall processing time and labor costs but also leads to significant material loss during purification stages. The reliance on precious metal catalysts like palladium or rhodium in some traditional cross-coupling reactions further exacerbates cost issues, particularly when scaling to industrial levels where catalyst recovery becomes a logistical burden. Additionally, achieving high stereoselectivity for specific geometric isomers often requires complex chiral auxiliaries or specialized reagents that are not commercially viable for large-scale production.
The Novel Approach
The methodology outlined in patent CN113173939B overcomes these historical bottlenecks by introducing a direct, one-pot three-component coupling strategy. This novel approach utilizes terminal alkynes, bis(pinacolato)diboron, and either diazo compounds or N-sulfonylhydrazones as carbene precursors to generate the desired alkenyl boronates in a single operational sequence. The reaction proceeds through an active allene intermediate generated in situ, which is immediately trapped by the boron reagent under copper catalysis. This eliminates the need for isolating unstable intermediates, thereby simplifying the workflow and improving overall atom economy. Crucially, the system offers precise control over stereochemistry; using diazo compounds yields (Z)-trisubstituted alkenyl borates, while N-sulfonylhydrazones facilitate the formation of (E)-isomers. This level of control, combined with the use of inexpensive copper catalysts and mild reaction temperatures, positions this technology as a superior alternative for the efficient manufacturing of high-value organoboron intermediates.
Mechanistic Insights into Copper-Catalyzed Stereoselective Boronation
The mechanistic pathway of this transformation is a testament to the sophistication of modern copper catalysis. The reaction initiates with the activation of the terminal alkyne by the copper catalyst, followed by the insertion of the carbene species derived from the diazo compound or N-sulfonylhydrazone. This step generates a reactive copper-allenyl intermediate, which is the pivotal species determining the subsequent stereochemical outcome. In the presence of a bidentate nitrogen ligand like 2,2'-bipyridine, the coordination environment favors a specific geometry that leads to the syn-addition of the boron moiety, resulting in the (Z)-configured product. Conversely, when bulky phosphine ligands such as Xantphos are employed with N-sulfonylhydrazones, the steric and electronic properties of the catalyst system direct the reaction towards the thermodynamically stable (E)-isomer. This ligand-dependent divergence allows chemists to tune the reaction outcome without changing the core catalytic metal, offering a versatile platform for diverse synthetic applications.
Impurity control is inherently managed by the high selectivity of the copper catalyst system. The use of molecular sieves as additives plays a critical role in scavenging trace moisture, which could otherwise hydrolyze the sensitive boron reagents or deactivate the catalyst. Furthermore, the mild reaction conditions (30°C for diazo pathways and 20-90°C for hydrazone pathways) minimize thermal degradation of substrates and products, ensuring a cleaner reaction profile. The broad substrate scope demonstrated in the patent, encompassing various substituted phenyl rings, heterocycles, and alkyl groups, indicates that the catalytic cycle is robust against electronic and steric variations. This resilience reduces the formation of side products associated with competing reaction pathways, such as homocoupling of alkynes or decomposition of diazo species, thereby simplifying downstream purification and enhancing the purity of the final pharmaceutical intermediate.
How to Synthesize Tri-substituted Alkenyl Borates Efficiently
Implementing this synthesis route requires careful attention to reaction parameters to maximize yield and selectivity. The process is divided into two distinct protocols depending on the desired stereochemistry. For (Z)-isomers, the reaction is conducted under an inert argon atmosphere using DMF as the solvent at a mild 30°C. For (E)-isomers, a two-stage temperature protocol is utilized, starting at 90°C in dioxane to generate the intermediate, followed by cooling to 20°C for the boronation step. The choice of ligand is paramount, with 2,2'-bipyridine being optimal for the former and Xantphos for the latter. Detailed standardized operating procedures regarding stoichiometry, addition rates, and workup protocols are essential for reproducibility. The following guide outlines the critical steps for executing this transformation effectively in a laboratory or pilot plant setting.
- For (Z)-isomers: React terminal alkyne, B2(pin)2, and diazo compound with CuI catalyst and 2,2'-bipyridine ligand in DMF at 30°C for 18 hours under argon.
- For (E)-isomers: React terminal alkyne and N-sulfonylhydrazone with CuI and base in dioxane at 90°C, then add B2(pin)2, ligand (Xantphos), and methanol at 20°C.
- Purify the crude reaction mixture via column chromatography using ethyl acetate and petroleum ether to isolate the high-purity alkenyl boronate product.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, this copper-catalyzed technology offers transformative benefits that directly impact the bottom line. The shift from precious metal catalysts to copper significantly reduces raw material costs, as copper salts are orders of magnitude cheaper than palladium or rhodium complexes. Moreover, the one-pot nature of the reaction consolidates multiple synthetic steps into a single vessel, drastically reducing solvent consumption, energy usage, and labor hours associated with intermediate isolation. This consolidation translates to a substantially simplified manufacturing process that is easier to validate and regulate, which is a key consideration for GMP production of API intermediates. The use of common, commercially available solvents like DMF and dioxane further ensures supply chain stability, avoiding reliance on exotic or restricted reagents that might face sourcing bottlenecks.
- Cost Reduction in Manufacturing: The elimination of expensive precious metal catalysts and the reduction of unit operations lead to significant cost savings. By avoiding the isolation of unstable allene intermediates, the process minimizes material loss and waste generation. The high atom economy of the three-component coupling ensures that a greater proportion of raw materials end up in the final product, optimizing resource utilization. Additionally, the mild reaction conditions reduce energy consumption for heating and cooling, contributing to lower utility costs per kilogram of product produced.
- Enhanced Supply Chain Reliability: The starting materials for this reaction, including terminal alkynes, bis(pinacolato)diboron, and diazo compounds, are widely available from multiple global suppliers. This diversity in sourcing mitigates the risk of supply disruptions that can occur with single-source specialty reagents. The robustness of the reaction across a wide range of substrates means that the same process platform can be adapted for various derivatives without extensive re-optimization, allowing for rapid response to changing market demands for different API intermediates.
- Scalability and Environmental Compliance: The process is inherently scalable due to its simple operational requirements and lack of hazardous high-pressure or high-temperature steps. The use of copper, a less toxic metal compared to many alternatives, simplifies waste treatment and disposal protocols, aiding in compliance with increasingly stringent environmental regulations. The high selectivity of the reaction reduces the burden on purification systems, lowering the volume of chromatographic solvents required and minimizing the environmental footprint of the manufacturing facility.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this copper-catalyzed synthesis method. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation. Understanding these nuances is vital for R&D teams evaluating the feasibility of this route for their specific projects and for procurement officers assessing the long-term viability of the supply chain.
Q: What is the primary advantage of this copper-catalyzed method over traditional boronation techniques?
A: The primary advantage is the ability to achieve high stereoselectivity (Z or E) in a one-pot three-component reaction, eliminating the need for pre-synthesized allene intermediates and reducing overall process complexity.
Q: Can this method be scaled for commercial production of API intermediates?
A: Yes, the method utilizes readily available raw materials, mild reaction temperatures (30°C or 20-90°C), and common solvents like DMF and dioxane, making it highly suitable for commercial scale-up.
Q: How is stereoselectivity controlled in this synthesis?
A: Stereoselectivity is controlled by the choice of carbene precursor and ligand; diazo compounds with bipyridine ligands yield (Z)-isomers, while N-sulfonylhydrazones with Xantphos ligands yield (E)-isomers.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tri-substituted Alkenyl Borate Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of advanced synthetic methodologies like the one described in patent CN113173939B for the development of next-generation pharmaceuticals. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory processes can be seamlessly translated into robust industrial operations. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of high-purity alkenyl boronates meets the exacting standards required by global regulatory bodies. Our commitment to quality and consistency makes us a trusted partner for companies seeking to secure their supply of critical organic intermediates.
We invite you to collaborate with us to leverage this cutting-edge technology for your specific drug development programs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your project's unique requirements. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise in copper-catalyzed transformations can accelerate your timeline to market while optimizing your manufacturing costs.
