Advanced Copper-Catalyzed Synthesis of Conjugated Enynes for Scalable Pharmaceutical Intermediate Production
Introduction to Next-Generation Conjugated Enyne Synthesis
The landscape of fine chemical synthesis is undergoing a significant transformation, driven by the urgent need for more sustainable and cost-effective methodologies. Patent CN110423217B introduces a groundbreaking preparation method for conjugated enyne compounds, a class of molecules that serves as critical building blocks in the development of natural products, functional materials, and advanced pharmaceutical intermediates. This innovation addresses long-standing challenges in organic synthesis by replacing expensive and scarce noble metal catalysts with abundant and inexpensive copper-based systems. The technology enables the efficient conversion of terminal alkynes and chloroacetamide derivatives into high-value (E)-3-benzyl-5-phenyl-N-(quinolin-8-yl)pent-4-ynamide structures under remarkably mild conditions. For R&D directors and procurement strategists, this represents a pivotal shift towards greener chemistry that does not compromise on yield or purity, offering a robust solution for the commercial scale-up of complex pharmaceutical intermediates.
Traditionally, the construction of carbon-carbon triple bonds adjacent to double bonds has relied on methodologies that are operationally demanding and economically burdensome. The novel approach detailed in this patent leverages a dual-alkyne strategy mediated by a cheap metal catalyst and a specific directing group, achieving high stereoselectivity and yield. By operating at temperatures as low as 35°C and utilizing readily available organic solvents, this process drastically reduces energy consumption and safety risks associated with high-pressure or high-temperature reactions. This technical breakthrough positions the technology as a cornerstone for cost reduction in fine chemical manufacturing, appealing directly to supply chain leaders looking to optimize their production pipelines for active pharmaceutical ingredients (APIs) and specialty chemicals.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of conjugated enynes has been dominated by methods such as the Wittig reaction and dehydration of α-hydroxyalkynes, both of which present severe limitations for industrial application. The Wittig reaction, while classic, necessitates the use of highly air-sensitive organolithium reagents like n-BuLi or t-BuLi to generate phosphorus ylides, requiring stringent anhydrous and anaerobic conditions that are difficult and costly to maintain on a large scale. Furthermore, the dehydration of α-hydroxyalkynes often generates water as a byproduct, mandating the use of molecular sieves or pre-dried solvents to drive the equilibrium, which adds significant complexity and waste to the process. Transition metal-catalyzed cross-couplings using palladium, rhodium, or iridium have offered improvements but introduce the prohibitive cost of precious metals and the challenge of removing trace metal residues to meet pharmaceutical purity standards.
The Novel Approach
In stark contrast, the methodology disclosed in CN110423217B utilizes a copper-catalyzed system that operates under ambient pressure and mild thermal conditions, effectively bypassing the hazards of organolithium chemistry. By employing 2-chloro-N-(quinolin-8-yl)acetamide as a key precursor, the reaction achieves high efficiency without the need for expensive noble metals, thereby eliminating the downstream burden of heavy metal scavenging. The process tolerates a wide range of functional groups, including electron-rich and electron-deficient aryl substituents, as well as heterocycles, demonstrating superior versatility compared to traditional routes. This shift not only enhances the reliability of the pharmaceutical intermediate supplier network by diversifying the chemical toolbox but also ensures that the production of high-purity OLED material precursors and drug candidates can be achieved with substantially lower environmental impact and operational cost.
Mechanistic Insights into Copper-Catalyzed Alkyne Coupling
The core of this innovation lies in the unique reactivity of the copper catalyst in conjunction with the 8-aminoquinoline directing group, which facilitates a sequential coupling mechanism. The reaction initiates with the activation of the terminal alkyne by the copper species, forming a copper-acetylide intermediate that undergoes nucleophilic substitution with the chloroacetamide derivative. This step is crucial as it installs the first alkyne unit while maintaining the integrity of the amide bond, setting the stage for the subsequent coupling. The presence of the quinoline moiety acts as a bidentate ligand, stabilizing the metal center and enhancing the regioselectivity of the transformation, which is essential for obtaining the desired (E)-isomer exclusively. This mechanistic pathway avoids the formation of unwanted byproducts commonly seen in non-directed couplings, ensuring a cleaner reaction profile that simplifies purification.
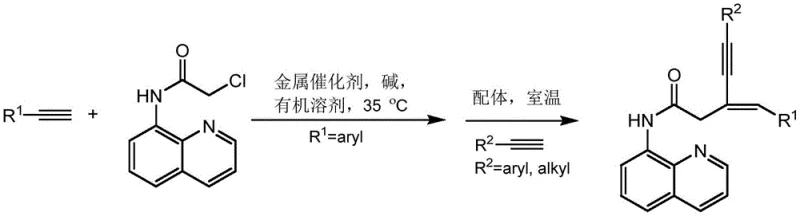
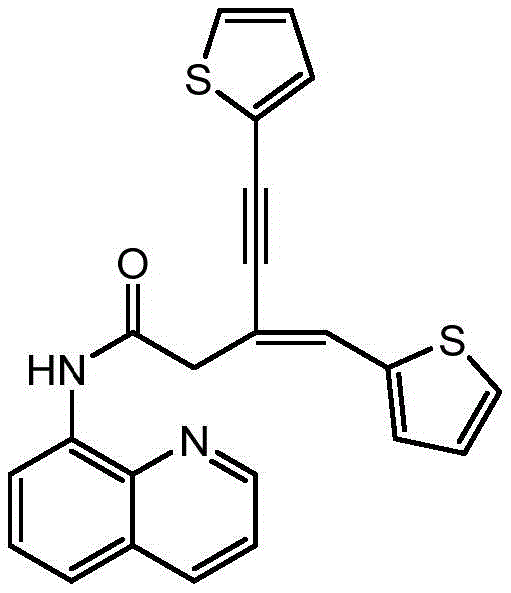
Following the initial coupling, the introduction of a second terminal alkyne in the presence of a nitrogen-based ligand, such as phenanthroline, triggers the final C-C bond formation. This second step proceeds smoothly at room temperature, highlighting the high catalytic activity of the system even under mild conditions. The ligand plays a pivotal role in modulating the electronic properties of the copper center, facilitating the oxidative addition and reductive elimination steps required for the coupling. The result is a highly efficient construction of the 1,3-conjugated enyne skeleton with excellent stereocontrol. As demonstrated in the patent examples, this mechanism is robust enough to accommodate diverse substrates, including thiophene derivatives, as shown below, proving its utility for synthesizing complex heterocyclic scaffolds found in modern medicinal chemistry.
Impurity control is inherently built into this catalytic cycle due to the high specificity of the copper-quinoline interaction. Unlike radical-based processes that can lead to polymerization or oligomerization of alkynes, this coordinated mechanism ensures that the reaction proceeds through defined intermediates. The use of mild bases like cesium carbonate further minimizes side reactions such as hydrolysis or elimination that could degrade the product quality. For R&D teams, this means that the impurity profile of the final API intermediate is significantly cleaner, reducing the number of recrystallization steps needed to achieve regulatory compliance. The ability to tune the reaction by selecting specific copper salts (e.g., CuI, CuCl) and ligands allows for fine-tuning the process to maximize yield and minimize trace metal contamination, a critical factor for reducing lead time for high-purity pharmaceutical intermediates.
How to Synthesize (E)-3-benzyl-5-phenyl-N-(quinolin-8-yl)pent-4-ynamide Efficiently
The practical implementation of this synthesis route is designed for ease of operation, making it accessible for both laboratory-scale optimization and pilot-plant production. The process begins with the preparation of the reaction mixture under an inert atmosphere to prevent oxidation of the copper catalyst, followed by the controlled addition of reagents to manage exotherms. Detailed standard operating procedures (SOPs) derived from the patent data ensure reproducibility and safety, guiding chemists through the precise molar ratios and timing required for optimal results. The following guide outlines the critical stages of the synthesis, emphasizing the parameters that drive high yield and selectivity.
- Combine 2-chloro-N-(quinolin-8-yl)acetamide, copper catalyst (e.g., CuI), and base (e.g., Cs2CO3) in an organic solvent under nitrogen atmosphere.
- Add terminal alkyne 1 and stir at 35°C for 24-48 hours to form the initial intermediate.
- Add ligand (e.g., phenanthroline) and terminal alkyne 2 under nitrogen, stir at room temperature for 24 hours, and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this technology offers transformative advantages that directly impact the bottom line and supply chain resilience. The replacement of precious metal catalysts with inexpensive copper salts results in a drastic reduction in raw material costs, which is particularly significant when scaling to multi-ton production volumes. Furthermore, the mild reaction conditions (35°C and room temperature) eliminate the need for specialized high-pressure reactors or extensive heating infrastructure, allowing for production in standard glass-lined or stainless steel vessels. This flexibility enhances supply chain reliability by enabling manufacturing across a broader range of facilities, reducing the risk of bottlenecks associated with specialized equipment availability.
- Cost Reduction in Manufacturing: The elimination of palladium, rhodium, or iridium catalysts removes a major cost driver from the bill of materials, while the avoidance of cryogenic conditions or air-sensitive reagents lowers utility and handling expenses. The simplified workup procedure, which relies on standard column chromatography rather than complex metal scavenging resins, further reduces processing time and consumable costs. These factors combine to create a highly economical process that improves margin potential for cost reduction in fine chemical manufacturing without sacrificing product quality.
- Enhanced Supply Chain Reliability: The starting materials, including terminal alkynes and chloroacetamides, are commodity chemicals that are readily available from multiple global suppliers, mitigating the risk of single-source dependency. The robustness of the reaction against moisture and oxygen variations (compared to organolithium methods) ensures consistent batch-to-batch quality, reducing the rate of failed batches and ensuring steady delivery schedules. This stability is crucial for maintaining continuous production lines for high-purity pharmaceutical intermediates and meeting the rigorous demands of international regulatory bodies.
- Scalability and Environmental Compliance: The use of common organic solvents like dichloromethane or toluene, which can be efficiently recovered and recycled, aligns with green chemistry principles and simplifies waste management. The absence of toxic heavy metals in the catalyst system eases the burden on wastewater treatment facilities and reduces the environmental footprint of the manufacturing process. This eco-friendly profile supports corporate sustainability goals and facilitates smoother regulatory approvals for new drug applications, positioning the technology as a future-proof solution for the commercial scale-up of complex polymer additives and fine chemicals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this copper-catalyzed synthesis method. These insights are derived directly from the experimental data and beneficial effects reported in the patent, providing clarity on how this technology can be integrated into existing workflows. Understanding these details is essential for technical teams evaluating the feasibility of adopting this route for their specific product portfolios.
Q: What are the advantages of this copper-catalyzed method over traditional Wittig reactions?
A: Unlike Wittig reactions that require air-sensitive organolithium reagents and strict anhydrous conditions, this copper-catalyzed method operates at mild temperatures (35°C) using inexpensive, stable metal catalysts, significantly simplifying operational safety and equipment requirements.
Q: Can this synthesis method tolerate heterocyclic substrates?
A: Yes, the protocol demonstrates excellent functional group tolerance, successfully accommodating heterocyclic alkynes such as thiophene derivatives with high yields (up to 93%), making it highly suitable for diverse pharmaceutical scaffold construction.
Q: Is this process scalable for industrial production?
A: The method utilizes cheap metals and mild conditions without the need for expensive noble metals like palladium or rhodium, offering a cost-effective pathway that is inherently more viable for large-scale commercial manufacturing compared to traditional precious metal catalysis.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Conjugated Enyne Compounds Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of adopting innovative synthetic routes like the one described in CN110423217B to enhance our service offerings. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab bench to manufacturing plant is seamless and efficient. We are committed to delivering stringent purity specifications through our rigorous QC labs, utilizing advanced analytical techniques to verify the structural integrity and impurity profiles of every batch of conjugated enyne compounds we produce.
We invite you to collaborate with us to leverage this cost-effective and scalable technology for your next project. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this copper-catalyzed method can optimize your budget. Please contact us today to request specific COA data and route feasibility assessments, and let us help you secure a reliable supply of high-quality intermediates for your pharmaceutical and material science applications.
