Advanced Palladium-Catalyzed Cross-Coupling For Commercial Scale-Up Of Complex Pharmaceutical Intermediates
Advanced Palladium-Catalyzed Cross-Coupling For Commercial Scale-Up Of Complex Pharmaceutical Intermediates
Introduction: Technological Breakthroughs In Indole Synthesis
The pharmaceutical industry continuously demands more efficient and scalable routes for synthesizing complex heterocyclic scaffolds, particularly 2,3-disubstituted indoles which serve as critical cores for numerous therapeutic agents including HCV polymerase inhibitors and anti-inflammatory drugs. Patent CN1930124A discloses a highly optimized process for preparing these compounds via palladium-catalyzed cross-coupling, addressing longstanding challenges in regioselectivity and byproduct formation that have historically plagued industrial production. This technology represents a significant leap forward for any reliable pharmaceutical intermediate supplier seeking to enhance their portfolio with high-value active pharmaceutical ingredient precursors. By leveraging specific ligand and solvent combinations, the disclosed method achieves superior control over the reaction pathway, minimizing the formation of undesired 2H-indole isomers that complicate downstream purification. The implications for large-scale manufacturing are profound, offering a robust framework for producing high-purity indole derivatives that meet stringent regulatory standards for clinical applications. As we analyze the technical nuances of this patent, it becomes clear that this methodology provides a competitive edge in the global supply chain for specialty chemicals.
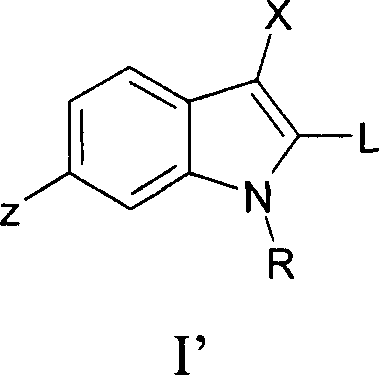
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional methods for constructing the indole core often rely on classical cyclization strategies or early-generation cross-coupling techniques that suffer from significant substrate limitations and inconsistent yields. Conventional Negishi or Suzuki couplings frequently require extensive screening of reaction variables to find optimal conditions for each specific substrate, leading to prolonged development timelines and increased resource consumption during process research. A major technical hurdle in these legacy processes is the tendency to form substantial amounts of dehalogenated byproducts or rearranged 2H-indole species, which drastically reduces the overall mass balance and complicates isolation procedures. Furthermore, the reliance on单一 solvent systems like pure THF often fails to suppress side reactions effectively, resulting in product-to-byproduct ratios that are commercially unacceptable for high-purity API manufacturing. These inefficiencies translate directly into higher production costs and longer lead times, creating bottlenecks for procurement managers who need consistent supply volumes. The inability to broadly apply a single set of conditions across a range of substrates means that every new analog requires a bespoke optimization campaign, hindering rapid scale-up capabilities.
The Novel Approach
The innovative process described in the patent data overcomes these historical barriers through a meticulously engineered catalytic system that utilizes a specific combination of palladium sources, phosphine ligands, and mixed solvent environments. By employing a mixture of N-methyl-2-pyrrolidone (NMP) and tetrahydrofuran (THF) in a precise 2:1 ratio, the reaction environment is tuned to stabilize the active catalytic species while simultaneously suppressing the formation of the thermodynamic 2H-indole byproduct. This novel approach allows for the use of milder reaction temperatures ranging from 70°C to 90°C, which enhances operational safety and reduces energy consumption compared to high-temperature reflux conditions often required in older methodologies. The selection of ligands such as triphenylphosphine or tricyclohexylphosphine is critical, as they modulate the electronic properties of the palladium center to facilitate oxidative addition and reductive elimination steps with high fidelity. Consequently, this method delivers a streamlined pathway that is not only chemically superior but also economically viable for industrial implementation. The result is a versatile platform technology capable of handling diverse substitution patterns without sacrificing yield or purity.
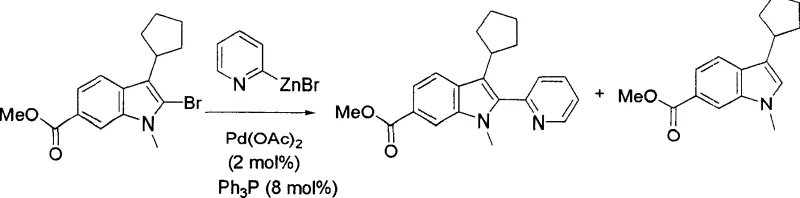
Mechanistic Insights into Pd-Catalyzed Cross-Coupling
Understanding the mechanistic underpinnings of this transformation is essential for R&D directors focused on impurity control and process robustness. The catalytic cycle initiates with the oxidative addition of the palladium(0) species into the carbon-halogen bond of the 2-haloindole substrate, a step that is highly sensitive to the steric and electronic environment provided by the ligand sphere. The use of bulky phosphine ligands helps to prevent the aggregation of palladium into inactive black metal precipitates, thereby maintaining a high concentration of active catalyst throughout the reaction duration. Subsequent transmetallation with the organozinc reagent occurs rapidly in the polar aprotic medium, transferring the aryl or heteroaryl group to the palladium center with high efficiency. The final reductive elimination step releases the desired 2,3-disubstituted indole product and regenerates the palladium(0) catalyst to continue the cycle. Crucially, the solvent system plays a dual role by solubilizing the zinc salts and stabilizing the transition states, which kinetically favors the formation of the 1H-indole tautomer over the 2H-isomer. This level of mechanistic control ensures that the impurity profile remains manageable even at large batch sizes.
Impurity control is further enhanced by the specific choice of base and workup conditions which are designed to quench reactive intermediates before they can degrade the final product. The patent data highlights that maintaining strict temperature control during the addition of reagents prevents exothermic runaways that could lead to decomposition or polymerization of sensitive functional groups. Additionally, the hydrolysis step utilized to convert ester intermediates to carboxylic acids is performed under controlled alkaline conditions to avoid racemization or hydrolysis of other labile moieties within the molecule. This attention to detail in the reaction engineering ensures that the final API intermediate meets the rigorous purity specifications required by global regulatory bodies. For technical teams, this means a reduced burden on analytical resources and a smoother technology transfer from pilot plant to commercial manufacturing suites. The predictability of the reaction outcome allows for tighter specification setting and more reliable quality assurance protocols.
How to Synthesize 2,3-Disubstituted Indoles Efficiently
Implementing this synthesis route requires adherence to precise operational parameters to replicate the high yields and selectivity reported in the intellectual property documentation. The process begins with the in situ generation of the organozinc nucleophile, which must be handled under inert atmosphere to prevent oxidation and moisture contamination that would deactivate the reagent. Following the formation of the zinc species, the palladium catalyst is introduced along with the indole substrate in the pre-mixed solvent system, initiating the cross-coupling event. Detailed standardized synthesis steps are provided in the guide below to ensure reproducibility across different manufacturing sites. Operators must monitor the reaction progress via HPLC to determine the exact endpoint, ensuring complete consumption of the starting halide while avoiding over-reaction that could generate secondary impurities. The subsequent workup involves careful pH adjustments and phase separations to maximize recovery of the organic product while removing metal residues to acceptable levels. This structured approach minimizes variability and ensures that every batch produced conforms to the expected quality attributes.
- Preparation of Organozinc Reagent: React 2-bromopyridine with isopropylmagnesium chloride in THF at 35-45°C, followed by transmetallation with ZnBr2 at temperatures below 65°C to ensure stability.
- Catalytic Cross-Coupling Reaction: Combine the organozinc species with the 2-bromoindole substrate in a NMP:THF (2:1) solvent system using Pd(OAc)2 and PPh3 ligands at 70-90°C.
- Workup and Purification: Quench the reaction with saturated ammonium chloride, extract with isopropyl acetate, and perform hydrolysis using sodium hydroxide in 1-propanol to isolate the final carboxylic acid derivative.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this technology offers compelling advantages that directly address the pain points of procurement managers and supply chain heads responsible for cost containment and continuity of supply. The ability to utilize aryl chlorides instead of the more expensive bromo or iodo analogs represents a significant opportunity for raw material cost optimization without compromising reaction efficiency. This flexibility in starting material selection allows sourcing teams to leverage broader supplier bases and negotiate better pricing terms, thereby reducing the overall cost of goods sold for the final pharmaceutical intermediate. Furthermore, the improved selectivity of the reaction reduces the load on downstream purification units, leading to substantial cost savings in chromatography resins and solvent consumption during the isolation phase. The robust nature of the process also enhances supply chain reliability by minimizing the risk of batch failures due to sensitivity to minor variations in reagent quality or environmental conditions. These factors combine to create a more resilient supply network capable of meeting the demanding delivery schedules of multinational pharmaceutical clients.
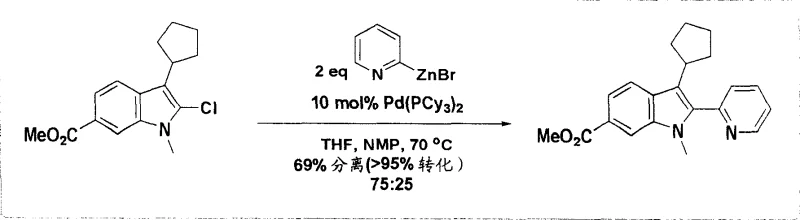
- Cost Reduction in Manufacturing: The elimination of expensive halide starting materials in favor of readily available chlorides drives down the direct material costs significantly, while the high conversion rates reduce waste disposal expenses associated with unreacted inputs. By optimizing the catalyst loading and solvent recovery loops, the process achieves a lean manufacturing profile that maximizes asset utilization and throughput capacity. This economic efficiency is critical for maintaining competitiveness in the generic drug market where margin pressure is intense. Qualitative analysis of the process flow suggests that the simplified workup procedure reduces labor hours and utility consumption per kilogram of product. Consequently, the total manufacturing cost structure is improved, allowing for more aggressive pricing strategies or higher margin retention depending on the commercial agreement.
- Enhanced Supply Chain Reliability: The use of stable and commercially abundant reagents ensures that production schedules are not disrupted by shortages of exotic or specialized chemicals. The scalability of the reaction from gram to multi-kilogram scale has been proven, providing confidence that supply volumes can be ramped up quickly to meet sudden increases in demand from downstream partners. This reliability is a key differentiator for a reliable pharmaceutical intermediate supplier operating in a volatile global market. The robustness of the chemistry against minor fluctuations in water content or oxygen levels further reduces the risk of unplanned downtime or batch rejections. Supply chain heads can therefore plan inventory levels with greater accuracy and reduce the need for excessive safety stock buffers.
- Scalability and Environmental Compliance: The process is designed with green chemistry principles in mind, utilizing solvent systems that are easier to recover and recycle compared to traditional chlorinated solvents. The reduction in heavy metal waste due to efficient catalyst usage aligns with increasingly stringent environmental regulations governing pharmaceutical manufacturing facilities. Scaling this process to multi-ton production is feasible without requiring specialized high-pressure equipment, lowering the capital expenditure barrier for expansion. This environmental and operational scalability ensures long-term sustainability of the supply source. It also facilitates smoother regulatory audits and inspections, as the process controls are well-defined and documented according to current good manufacturing practices.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this cross-coupling technology in a production environment. These answers are derived directly from the experimental data and embodiments disclosed in the patent literature to provide accurate guidance for decision-makers. Understanding these details helps stakeholders evaluate the feasibility of adopting this route for their specific product pipelines. The information covers aspects ranging from raw material compatibility to expected impurity profiles. We encourage technical teams to review these points thoroughly before initiating any technology transfer activities.
Q: How does the solvent system impact the product ratio in indole cross-coupling?
A: The patent data indicates that using a mixed solvent system of NMP and THF (2:1 ratio) significantly improves the ratio of the desired 2,3-disubstituted indole over the 2H-indole byproduct compared to using THF alone, achieving ratios up to 94:6.
Q: Can aryl chlorides be used instead of bromides for cost reduction?
A: Yes, the technology supports the use of 2-chloroindole intermediates which are typically less expensive than bromo analogs, utilizing specific ligand systems like Pd(PCy3)2 to maintain effective conversion rates.
Q: What is the scalability potential of this synthesis route?
A: The process has been demonstrated at a 25 kg scale in the patent examples, indicating robust feasibility for commercial manufacturing with established protocols for temperature control and exotherm management.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,3-Disubstituted Indoles Supplier
NINGBO INNO PHARMCHEM stands ready to leverage this advanced synthetic technology to support your drug development and commercialization goals with unparalleled expertise and capacity. As a seasoned CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from the laboratory bench to full-scale manufacturing. Our facility is equipped with state-of-the-art rigorous QC labs and analytical instruments capable of verifying stringent purity specifications for complex heterocyclic compounds. We understand the critical importance of timeline and quality in the pharmaceutical sector and have structured our operations to deliver consistent results that meet international regulatory standards. By partnering with us, you gain access to a team of chemists and engineers dedicated to optimizing every aspect of the production process for maximum efficiency and yield.
We invite you to contact our technical procurement team to discuss how we can tailor this synthesis route to your specific requirements and provide a Customized Cost-Saving Analysis for your project. Our experts are available to share specific COA data from previous campaigns and conduct detailed route feasibility assessments to identify potential risks and opportunities early in the engagement. Taking this proactive step will enable you to secure a stable supply of high-quality intermediates while optimizing your overall development budget. We look forward to collaborating with you to bring your next generation of therapeutics to market faster and more cost-effectively.
