Revolutionizing N-(2-quinoline)-2-ketone Production: A Mild, Metal-Free Commercial Strategy
Revolutionizing N-(2-quinoline)-2-ketone Production: A Mild, Metal-Free Commercial Strategy
The landscape of heterocyclic chemistry is undergoing a significant transformation driven by the urgent need for greener, more efficient synthetic pathways. A pivotal development in this domain is detailed in Chinese Patent CN112028873A, which discloses a robust method for preparing N-(2-quinoline)-2-ketone compounds under exceptionally mild conditions. This technology represents a departure from the energy-intensive and metal-dependent protocols that have historically dominated the synthesis of these biologically active scaffolds. By leveraging an innovative Atherton-Todd reaction sequence coupled with cross-dehydrogenative coupling, this process achieves high yields at room temperature, effectively resolving long-standing issues related to high thermal requirements and difficult raw material preparation. For global procurement and R&D teams, this patent offers a compelling blueprint for optimizing the supply chain of critical pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of N-(2-quinoline)-2-pyridone and quinoxalinone derivatives has been fraught with significant technical and economic hurdles. Traditional methodologies predominantly rely on copper-catalyzed nucleophilic substitution reactions between 2-haloquinolines and pyridones or quinoxalinones. These legacy processes typically demand harsh reaction temperatures reaching up to 150°C and extended reaction durations of approximately 12 hours, leading to substantial energy consumption and reduced throughput. Furthermore, recent advancements involving rhodium or palladium catalysis, while effective, introduce the complexity of using expensive transition metals that require rigorous removal steps to meet pharmaceutical purity standards. The reliance on such precious metals not only inflates the cost of goods sold but also introduces potential toxicity risks associated with residual metal impurities in the final active pharmaceutical ingredients.
The Novel Approach
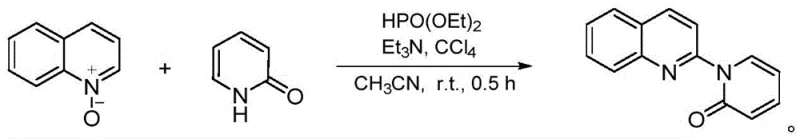
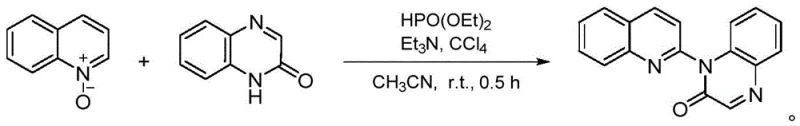
In stark contrast to these cumbersome legacy techniques, the novel approach outlined in the patent utilizes a metal-free strategy that operates efficiently at room temperature. This method employs quinoline-N-oxides and 2-hydroxypyridines (or 2-hydroxyquinoxalines) as starting materials, activated by diethyl phosphite and carbon tetrachloride in the presence of triethylamine. The reaction proceeds rapidly, often completing within just 0.5 hours, which drastically enhances process efficiency and equipment utilization rates. By eliminating the need for high heat and precious metal catalysts, this route simplifies the operational parameters and significantly lowers the barrier for commercial scale-up. The ability to generate the target N-(2-quinoline)-2-ketone structures under such benign conditions marks a substantial leap forward in process chemistry.
Mechanistic Insights into Atherton-Todd Mediated Cross-Dehydrogenative Coupling
The core of this technological breakthrough lies in the elegant orchestration of the Atherton-Todd reaction to facilitate a cross-dehydrogenative coupling. Mechanistically, the process begins with the in-situ generation of diethyl phosphoryl chloride, a highly reactive electrophile formed from the interaction of diethyl phosphite and carbon tetrachloride under the basic influence of triethylamine. This transient species then undergoes a nucleophilic substitution with the quinoline-N-oxide, activating the heterocyclic ring for subsequent coupling. This activation step is critical as it bypasses the need for pre-functionalized halogenated substrates, which are often costly and difficult to synthesize. The resulting intermediate is then poised for a direct coupling with the 2-hydroxypyridine or 2-hydroxyquinoxaline nucleophile.
Following the initial activation, the system proceeds through a cross-dehydrogenative coupling phase driven by the alkaline environment provided by the excess triethylamine. This step involves the direct formation of a carbon-nitrogen bond between the quinoline moiety and the pyridone or quinoxalinone ring, releasing small molecule byproducts that are easily removed during workup. The beauty of this mechanism is its atom economy and the avoidance of stoichiometric oxidants or metal catalysts that often complicate purification. By generating the reactive phosphoryl species in situ, the process also mitigates safety risks associated with handling toxic diethyl phosphoryl chloride directly. This mechanistic pathway ensures a clean reaction profile with minimal side products, facilitating easier downstream purification via standard silica gel chromatography.
How to Synthesize N-(2-quinoline)-2-pyridone Efficiently
Implementing this synthesis route requires precise control over reagent stoichiometry and mixing conditions to maximize the yield of the desired N-(2-quinoline)-2-pyridone or quinoxalinone derivatives. The protocol dictates dispersing the hydroxyl-containing heterocycles and quinoline-N-oxide in acetonitrile, followed by the sequential addition of the activator system. Maintaining the reaction at ambient temperature (0-45°C) is crucial to preserving the mild nature of the process while ensuring complete conversion within the short 0.5-hour window. Detailed standard operating procedures regarding specific molar ratios, such as the 10:6 ratio of triethylamine to substrate, are essential for reproducibility. For a comprehensive, step-by-step technical guide on executing this synthesis, please refer to the standardized protocol section below.
- Disperse 2-hydroxypyridine and quinoline-N-oxide in acetonitrile solvent within a reaction vessel.
- Add diethyl phosphite activator and carbon tetrachloride, followed by triethylamine to initiate the Atherton-Todd reaction.
- Stir the mixture at room temperature for 0.5 hours, then quench with water, extract with ethyl acetate, and purify via silica gel chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this mild synthesis methodology offers profound advantages for procurement managers and supply chain directors seeking to optimize their sourcing strategies for complex heterocyclic intermediates. The most immediate benefit is the drastic simplification of the manufacturing process, which translates directly into reduced operational expenditures. By removing the dependency on expensive transition metal catalysts like rhodium and palladium, manufacturers can achieve significant cost reduction in pharmaceutical intermediates manufacturing without compromising on quality or yield. Furthermore, the elimination of these metals removes the need for specialized scavenging resins and extensive purification steps, streamlining the production workflow and reducing waste generation.
- Cost Reduction in Manufacturing: The economic impact of switching to this metal-free protocol is substantial, primarily driven by the removal of high-cost catalytic systems and the reduction in energy consumption. Traditional methods requiring heating to 150°C incur heavy utility costs, whereas this room-temperature process operates with minimal energy input. Additionally, the use of commodity chemicals like diethyl phosphite and triethylamine, rather than bespoke halogenated precursors, ensures a stable and low-cost raw material supply base. This shift allows for a more competitive pricing structure for the final N-(2-quinoline)-2-ketone products, enhancing margin potential for downstream drug developers.
- Enhanced Supply Chain Reliability: Supply chain resilience is significantly bolstered by the use of widely available, non-specialized reagents in this synthesis. Unlike proprietary catalysts that may face supply bottlenecks or long lead times, the key components of this reaction are bulk chemicals with robust global availability. The shortened reaction time of 0.5 hours compared to the conventional 12 hours also dramatically increases production capacity, allowing suppliers to respond more agilely to fluctuating market demands. This efficiency gain reduces lead time for high-purity pharmaceutical intermediates, ensuring that project timelines for drug development programs are met without delay.
- Scalability and Environmental Compliance: The environmental profile of this process aligns perfectly with modern green chemistry principles, facilitating easier regulatory approval and environmental compliance. The absence of heavy metal waste streams simplifies effluent treatment and reduces the environmental footprint of the manufacturing site. Moreover, the mild conditions and simple workup procedure involving aqueous quenching and extraction make the process highly amenable to commercial scale-up from kilogram to multi-ton scales. This scalability ensures a continuous and reliable supply of complex polymer additives or pharmaceutical building blocks, mitigating the risk of production stoppages due to process complexity.
Frequently Asked Questions (FAQ)
To further clarify the technical and commercial implications of this patented technology, we have compiled a set of frequently asked questions based on the specific details found in the patent documentation. These inquiries address common concerns regarding reaction safety, scalability, and product purity that are critical for decision-makers evaluating new synthetic routes. Understanding these nuances is essential for assessing the feasibility of integrating this method into existing production pipelines. The answers provided below are derived directly from the experimental data and beneficial effects described in the source intellectual property.
Q: What are the primary advantages of this new synthesis method over traditional copper-catalyzed routes?
A: The primary advantages include operation at room temperature (0-45°C) versus the harsh 150°C required for traditional methods, significantly shorter reaction times (0.5 hours vs 12 hours), and the complete elimination of expensive transition metal catalysts like copper, rhodium, or palladium.
Q: How does the in-situ generation of diethyl phosphoryl chloride improve safety?
A: By generating the reactive diethyl phosphoryl chloride intermediate in situ via the Atherton-Todd reaction using diethyl phosphite and carbon tetrachloride, the process avoids the direct handling and storage of this toxic substance, thereby enhancing operational safety for laboratory and plant personnel.
Q: Is this method scalable for commercial production of pharmaceutical intermediates?
A: Yes, the method utilizes readily available commercial reagents and mild conditions that are highly conducive to scale-up. The absence of sensitive transition metal catalysts simplifies downstream processing and impurity control, making it ideal for large-scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-(2-quinoline)-2-pyridone Supplier
As the global demand for high-quality heterocyclic building blocks continues to rise, partnering with an experienced CDMO partner is essential for translating laboratory innovations into commercial reality. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and precision. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of N-(2-quinoline)-2-ketone compounds adheres to the highest industry standards. We are committed to delivering value through technical excellence and operational reliability.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis route can be tailored to your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the potential economic benefits of adopting this metal-free methodology for your supply chain. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring that your next project is built on a foundation of scientific rigor and commercial viability.
