Advanced Benzophenone-Catalyzed Cyclopropanation for High-Purity Fragrance Intermediates Manufacturing
The landscape of fine chemical synthesis, particularly within the fragrance and pharmaceutical sectors, is undergoing a significant paradigm shift towards metal-free catalytic systems. Patent CN108484347B introduces a groundbreaking preparation method for the cyclopropanation of terminal isoprene compounds, addressing critical pain points associated with traditional noble metal catalysis. This innovation replaces expensive and toxic palladium or zinc-copper systems with an organocatalytic approach utilizing benzophenone in an ether solvent. For R&D directors and procurement specialists, this represents a dual victory: the elimination of heavy metal residues which simplifies regulatory compliance, and a drastic reduction in raw material costs. The technology enables the efficient transformation of readily available terpenes like farnesene, myrcene, and ocimene into high-value cyclopropane derivatives with yields consistently exceeding 95 percent. By leveraging this novel pathway, manufacturers can achieve superior purity profiles essential for sensitive applications in perfumery and drug synthesis, while simultaneously streamlining the supply chain through simplified downstream processing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the cyclopropanation of terminal isoprene units has relied heavily on transition metal catalysts, creating substantial bottlenecks in both cost and quality control. As illustrated in prior art such as WO2017/046122a1, the industry standard often involves Palladium (II) catalysts like Pd(acac)2 or Pd(OAc)2 used in conjunction with NMK reaction solutions. 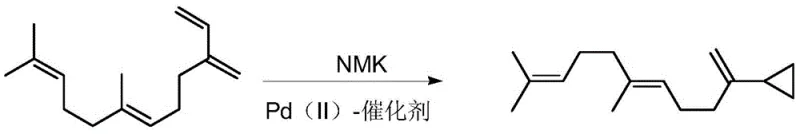 This legacy approach suffers from severe drawbacks, primarily the necessity for a two-phase system due to the insolubility of the catalytic species in organic solvents, which necessitates the addition of alkaline aqueous solutions. Furthermore, the removal of residual palladium is notoriously difficult and expensive, often requiring specialized scavenging resins or multiple recrystallization steps to meet the stringent parts-per-million (ppm) limits imposed by global safety regulations. Alternative methods utilizing zinc-copper couples, while cheaper in terms of catalyst cost, introduce different risks related to heavy metal pollution and difficult post-treatment protocols, ultimately compromising the environmental footprint and operational efficiency of the manufacturing process.
This legacy approach suffers from severe drawbacks, primarily the necessity for a two-phase system due to the insolubility of the catalytic species in organic solvents, which necessitates the addition of alkaline aqueous solutions. Furthermore, the removal of residual palladium is notoriously difficult and expensive, often requiring specialized scavenging resins or multiple recrystallization steps to meet the stringent parts-per-million (ppm) limits imposed by global safety regulations. Alternative methods utilizing zinc-copper couples, while cheaper in terms of catalyst cost, introduce different risks related to heavy metal pollution and difficult post-treatment protocols, ultimately compromising the environmental footprint and operational efficiency of the manufacturing process.
The Novel Approach
The methodology disclosed in CN108484347B fundamentally reengineers this transformation by employing benzophenone as a soluble organic catalyst within a homogeneous ether solvent system. This shift from heterogeneous metal catalysis to homogeneous organocatalysis resolves the solubility issues inherent in previous methods, allowing for uniform dispersion of the catalyst and improved contact with the substrate. The reaction proceeds under mild conditions, typically below 35°C, which preserves the integrity of sensitive functional groups often found in complex terpene structures. By avoiding the use of noble metals entirely, the process inherently guarantees a product free from metal ion contamination, a critical quality attribute for fragrance intermediates where odor profiles can be ruined by trace metallic impurities. Additionally, the simplicity of the workup—merely requiring an acid quench followed by standard washing and distillation—drastically reduces the operational complexity and time-to-market for these high-value intermediates.
Mechanistic Insights into Benzophenone-Catalyzed Cyclopropanation
The core of this technological advancement lies in the unique interaction between benzophenone, diazomethane, and the terminal isoprene substrate within an ether medium. Benzophenone, traditionally known as a photoinitiator, functions here as a highly effective chemical catalyst that facilitates the transfer of the methylene group from diazomethane to the electron-rich terminal double bond. The choice of ether solvents, such as tetrahydrofuran (THF), diethyl ether, or dioxane, is not arbitrary; these solvents exhibit excellent solubility for benzophenone, ensuring a true homogeneous reaction phase that maximizes catalytic turnover.  Crucially, the mechanistic pathway demonstrates remarkable regioselectivity. In substrates containing multiple olefinic bonds, such as farnesene or myrcene, the catalytic system preferentially targets the terminal isoprene moiety. This selectivity is driven by the specific electronic and steric environment created by the catalyst-solvent complex, which lowers the activation energy for the terminal double bond while leaving internal alkenes largely untouched. This precision minimizes the formation of unwanted regioisomers, such as internal cyclopropanation byproducts, thereby simplifying the purification process and boosting the overall isolated yield of the target molecule.
Crucially, the mechanistic pathway demonstrates remarkable regioselectivity. In substrates containing multiple olefinic bonds, such as farnesene or myrcene, the catalytic system preferentially targets the terminal isoprene moiety. This selectivity is driven by the specific electronic and steric environment created by the catalyst-solvent complex, which lowers the activation energy for the terminal double bond while leaving internal alkenes largely untouched. This precision minimizes the formation of unwanted regioisomers, such as internal cyclopropanation byproducts, thereby simplifying the purification process and boosting the overall isolated yield of the target molecule.
From an impurity control perspective, the absence of metal salts eliminates a major class of potential contaminants that are difficult to detect and remove. Traditional metal-catalyzed routes often generate metal-carbene complexes that can lead to unpredictable side reactions or polymerization if not strictly controlled. In contrast, the benzophenone-mediated pathway operates through a cleaner mechanism that avoids these metallacyclic intermediates. The reaction temperature is maintained at a low range, preferably between 10°C and 20°C, which further suppresses thermal decomposition of the diazomethane reagent and prevents non-selective background reactions. This tight control over reaction parameters ensures that the final product possesses a gas chromatography (GC) purity often exceeding 95 percent, meeting the rigorous specifications required for direct use in high-end fragrance formulations or as advanced pharmaceutical building blocks.
How to Synthesize Cyclopropanated Terpenes Efficiently
Implementing this synthesis route requires careful attention to reagent stoichiometry and temperature control to maximize the benefits of the benzophenone catalytic system. The process is designed to be robust and scalable, utilizing standard laboratory equipment that can be easily adapted for pilot and commercial plant operations. The following guide outlines the generalized protocol derived from the patent examples, focusing on the critical steps that ensure high yield and purity. For detailed standardized operating procedures (SOPs) tailored to specific substrates, please refer to the technical documentation provided below.
- Dissolve the terminal isoprene substrate (e.g., farnesene, myrcene) and benzophenone catalyst (3-6 wt%) in an ether solvent such as diethyl ether or THF at room temperature.
- Maintain the reaction temperature below 35°C (preferably 10-20°C) and slowly add a solution of diazomethane in ether dropwise to the mixture.
- After completion, quench the reaction with dilute hydrochloric acid, wash the organic layer with water until neutral, dry over sodium sulfate, and purify via rectification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this benzophenone-catalyzed method offers transformative economic and logistical benefits. The most immediate impact is seen in the raw material cost structure; by replacing scarce and volatile noble metals like palladium with commodity chemicals like benzophenone, the direct material cost of the catalyst is reduced by orders of magnitude. Furthermore, the elimination of expensive metal scavengers and the reduction in solvent usage during the purification phase contribute to a significantly leaner cost of goods sold (COGS). The simplified workflow also translates into reduced labor hours and faster batch cycle times, allowing manufacturing facilities to increase throughput without additional capital expenditure. This efficiency gain is crucial for maintaining competitiveness in the fast-moving fragrance and flavor markets where time-to-market is a key differentiator.
- Cost Reduction in Manufacturing: The transition to a metal-free catalytic system removes the financial burden associated with precious metal recovery and disposal. Since benzophenone is an inexpensive organic compound, the catalyst cost becomes negligible compared to traditional Pd-based systems. Moreover, the homogeneous nature of the reaction eliminates the need for complex filtration equipment or specialized resin columns for metal removal, leading to substantial savings in both equipment maintenance and consumable costs. The high selectivity of the reaction also means less waste generation, as fewer byproducts need to be separated and disposed of, further enhancing the overall economic viability of the process.
- Enhanced Supply Chain Reliability: Relying on noble metals exposes supply chains to geopolitical risks and market volatility, as seen with fluctuating palladium prices. By switching to a fully organic catalytic system, manufacturers insulate their production from these external shocks. The reagents required—benzophenone, diazomethane precursors, and ether solvents—are widely available from multiple global suppliers, ensuring a stable and continuous supply of critical inputs. This diversification of the supply base reduces the risk of production stoppages due to raw material shortages, providing a more resilient operational framework for long-term planning.
- Scalability and Environmental Compliance: The mild reaction conditions and simple aqueous workup make this process inherently safer and easier to scale from kilogram to multi-ton quantities. The absence of heavy metals simplifies wastewater treatment and reduces the regulatory burden associated with hazardous waste disposal. This aligns perfectly with modern green chemistry principles and corporate sustainability goals, making the final products more attractive to environmentally conscious customers. The ability to produce high-purity intermediates with a lower environmental footprint positions companies as leaders in sustainable manufacturing, a growing requirement in the global supply chains of major multinational corporations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel cyclopropanation technology. These answers are derived directly from the experimental data and technical specifications outlined in the patent literature, providing a reliable foundation for decision-making. Understanding these nuances is essential for R&D teams evaluating the feasibility of integrating this method into existing production lines.
Q: Why is benzophenone preferred over palladium catalysts for this cyclopropanation?
A: Benzophenone eliminates the risk of heavy metal contamination, which is critical for fragrance and pharmaceutical applications. Unlike Pd(II) systems that require complex two-phase separations and alkaline conditions, benzophenone operates in a homogeneous ether system, simplifying purification and ensuring no metal ion residues remain in the final product.
Q: What is the selectivity profile of this reaction regarding multiple double bonds?
A: The method exhibits exceptional regioselectivity for the terminal isoprene double bond. Even in substrates like farnesene or myrcene which contain internal double bonds, the reaction preferentially cyclopropanates the terminal position, minimizing byproduct formation and achieving yields exceeding 95%.
Q: Can this process be scaled for industrial production?
A: Yes, the process is highly scalable due to its mild operating conditions (temperatures below 35°C) and simple workup procedure involving standard acid quenching and distillation. The absence of expensive noble metals and the use of common ether solvents make it economically viable for large-scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Fragrance Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the shift towards metal-free synthesis is not just a technical upgrade but a strategic imperative for modern chemical manufacturing. Our team of expert chemists has extensively validated the benzophenone-catalyzed cyclopropanation pathway, confirming its potential to deliver exceptional purity and yield for complex terpene derivatives. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab bench to reactor is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of detecting trace impurities, guaranteeing that every batch meets stringent purity specifications required by the most demanding global clients.
We invite you to collaborate with us to leverage this cutting-edge technology for your specific product needs. Whether you require custom synthesis of cyclopropanated farnesene derivatives or scale-up of other terminal isoprene intermediates, our technical procurement team is ready to assist. Please contact us to request a Customized Cost-Saving Analysis tailored to your current production volumes. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how this innovative method can optimize your supply chain and enhance your product portfolio.
