Revolutionizing Pharmaceutical Intermediate Synthesis via Blue-Light Photocatalysis and C-C Bond Cleavage
Introduction to Breakthrough Photocatalytic C-C Bond Cleavage
The landscape of synthetic organic chemistry is undergoing a significant transformation driven by the need for more sustainable and efficient methodologies, particularly in the realm of pharmaceutical intermediate production. Patent CN115745836A introduces a pioneering approach that leverages visible-light photoredox catalysis to induce the reaction of alkyl neopentylbenzenes with various capture reagents. This technology addresses a long-standing challenge in the field: the activation and cleavage of unactivated carbon-carbon bonds in tertiary alkyl systems without the need for extreme thermal conditions or hazardous reagents. By utilizing acridinium salt photocatalysts under blue light irradiation, this method facilitates the generation of transient alkyl radicals and carbocations, which are subsequently trapped to form valuable functionalized products. This innovation not only expands the toolbox available to medicinal chemists but also offers a pathway to more cost-effective and environmentally benign manufacturing processes for complex organic molecules.
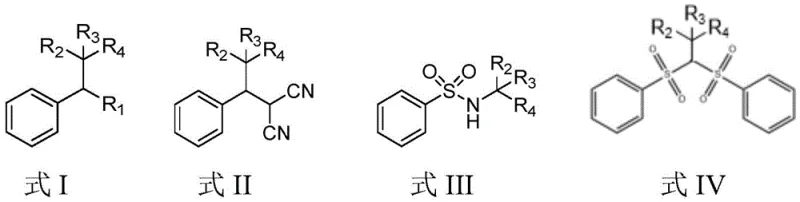
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the functionalization of unactivated alkyl chains, especially those involving tertiary carbon centers, has been fraught with difficulties. Conventional methods often rely on pre-functionalized substrates, such as alkyl halides or redox-active esters, which require additional synthetic steps to prepare, thereby increasing the overall cost and waste generation. Furthermore, thermal radical generation methods frequently necessitate high temperatures and toxic initiators, posing significant safety risks and limiting the scope of compatible functional groups. The high oxidation potential of pure alkyl backbones has historically made the direct generation of alkyl radicals from simple precursors a primary challenge in photoredox catalysis. These limitations restrict the structural diversity accessible to researchers and complicate the supply chain for high-purity intermediates needed in drug discovery and development.
The Novel Approach
In stark contrast, the novel approach detailed in the patent utilizes a photocatalyst-induced strategy that bypasses these traditional hurdles. By employing alkyl neopentylbenzenes as direct substrates, the method achieves carbon-carbon bond cleavage through a single-electron transfer mechanism initiated by blue light. This process eliminates the need for pre-installed leaving groups, streamlining the synthetic route significantly. The use of mild reaction conditions, specifically room temperature irradiation in nitroalkane solvents, ensures compatibility with a wide range of sensitive functional groups. This shift from thermal to photochemical activation represents a paradigm shift in how chemists approach C-C bond activation, offering a safer, more controllable, and atom-economical alternative that aligns perfectly with the principles of green chemistry and modern process intensification.
Mechanistic Insights into Acridinium Salt Photocatalysis
The core of this transformative technology lies in the unique properties of the acridinium salt photocatalyst, which acts as a potent single-electron oxidant upon excitation. When irradiated with blue light, the photocatalyst enters an excited state capable of oxidizing the electron-rich aromatic ring of the alkyl neopentylbenzene substrate. This initial oxidation triggers a cascade of events leading to the fragmentation of the adjacent carbon-carbon bond. The resulting intermediate species, comprising both alkyl radicals and carbocations, are highly reactive and can be efficiently intercepted by various nucleophilic capture reagents. This dual-capture capability allows for the synthesis of diverse product classes, including dinitriles and sulfonamides, from a single starting material platform. The precise control over the radical generation process minimizes side reactions and enhances the selectivity of the transformation, which is critical for producing high-purity compounds required in regulated industries.
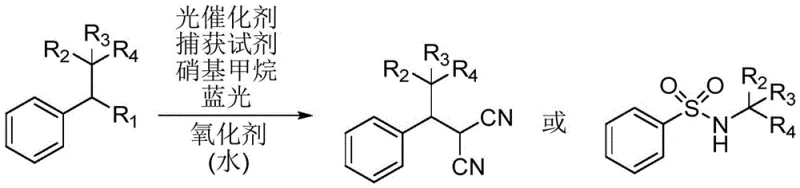
Furthermore, the synergistic effect of the oxidant and water additives plays a crucial role in maintaining the catalytic cycle and improving overall efficiency. The oxidant, such as ammonium persulfate, helps regenerate the ground state of the photocatalyst, ensuring turnover and sustained reactivity throughout the process. Water, acting as a co-solvent or additive, appears to facilitate the stabilization of charged intermediates or assist in proton transfer steps, thereby boosting the yield of the desired capture products. The specific structure of the acridinium catalyst, often featuring bulky substituents to prevent deactivation, ensures robust performance even in complex reaction matrices. Understanding these mechanistic nuances is essential for optimizing the process for commercial scale-up, as it allows engineers to fine-tune parameters like light intensity, reagent stoichiometry, and mixing efficiency to maximize throughput while maintaining product quality.
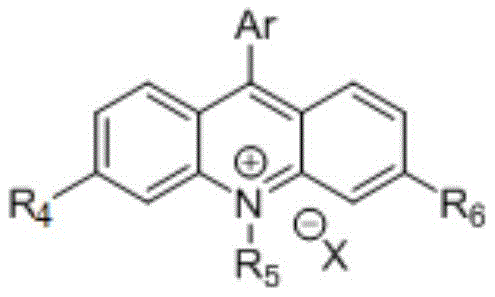
How to Synthesize Alkyl Neopentylbenzene Derivatives Efficiently
Implementing this photocatalytic method in a laboratory or pilot plant setting requires careful attention to the reaction setup and reagent quality to ensure reproducible results. The process begins with the preparation of a reaction mixture containing the alkyl neopentylbenzene substrate, a chosen capture reagent like benzylidenemalononitrile, and the acridinium photocatalyst dissolved in a nitroalkane solvent. The addition of an oxidant and a controlled amount of water is critical for driving the reaction to completion. Once the mixture is degassed and placed under a blue light source, the transformation proceeds smoothly, typically monitored by thin-layer chromatography to determine the endpoint. The detailed standardized synthesis steps see the guide below.
- Prepare the reaction mixture by combining the alkyl neopentylbenzene substrate, capture reagent (such as benzylidenemalononitrile), acridinium salt photocatalyst, and oxidant in a nitroalkane solvent with water.
- Degas the reaction vessel and backfill with inert gas, then irradiate the mixture with a 450nm blue light source to initiate the photoredox catalytic cycle.
- Monitor the reaction progress via TLC, and upon completion, concentrate the mixture and purify the target product using flash column chromatography on silica gel.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this photocatalytic technology offers substantial strategic benefits beyond mere technical novelty. The ability to utilize unactivated, readily available alkyl neopentylbenzenes as starting materials significantly reduces the dependency on expensive, pre-functionalized building blocks that often suffer from supply volatility. By simplifying the synthetic sequence and eliminating multiple protection-deprotection steps or precursor syntheses, the overall manufacturing cost is drastically reduced. This streamlined approach translates directly into improved margin potential for downstream products and greater flexibility in pricing strategies for contract manufacturing organizations. Moreover, the mild reaction conditions reduce the energy consumption associated with heating and cooling, contributing to a lower carbon footprint and aligning with corporate sustainability goals.
- Cost Reduction in Manufacturing: The elimination of transition metal catalysts and the use of inexpensive organic photocatalysts remove the need for costly heavy metal removal steps, which are often a bottleneck in API production. This simplification of the purification workflow leads to significant savings in both time and resources, allowing for a more lean manufacturing operation. Additionally, the high atom economy of the reaction ensures that a larger proportion of the starting material ends up in the final product, minimizing waste disposal costs and maximizing raw material utilization efficiency.
- Enhanced Supply Chain Reliability: Sourcing unactivated hydrocarbon feedstocks is generally more stable and less prone to geopolitical disruptions compared to specialized radical precursors. This robustness in raw material availability ensures consistent production schedules and reduces the risk of delays caused by supplier shortages. The scalability of the photochemical process, supported by the increasing availability of industrial-grade LED reactors, further strengthens supply chain resilience, enabling manufacturers to respond quickly to fluctuating market demands without compromising on quality or lead times.
- Scalability and Environmental Compliance: The process operates under ambient pressure and temperature, reducing the engineering controls required for high-pressure or high-temperature reactors. This inherent safety profile simplifies regulatory compliance and lowers the barrier for scaling up from gram to ton quantities. The use of nitroalkane solvents, while requiring careful handling, is well-established in the industry, and the overall reduction in hazardous by-products simplifies waste treatment protocols, making the process more environmentally compliant and easier to permit in strict regulatory jurisdictions.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this photocatalytic C-C bond cleavage technology. These insights are derived directly from the patent specifications and are intended to clarify the operational feasibility and strategic value of the method for potential partners and licensees. Understanding these details is crucial for evaluating the fit of this technology within existing production portfolios and R&D pipelines.
Q: What is the primary advantage of this photocatalytic method over traditional C-C bond cleavage?
A: This method allows for the direct generation of alkyl radicals and carbocations from unactivated tertiary alkyl neopentylbenzenes under mild visible light conditions, eliminating the need for harsh thermal conditions or pre-functionalized radical precursors.
Q: Which catalyst system is preferred for this transformation?
A: The patent specifies the use of acridinium salt photocatalysts, particularly Mes-Acr-PhBF4, which provides superior single-electron oxidation potential to drive the carbon-carbon bond breakage efficiently.
Q: Is this process suitable for large-scale manufacturing of pharmaceutical intermediates?
A: Yes, the process utilizes commercially available reagents, standard blue LED light sources, and common solvents like nitromethane, making it highly adaptable for commercial scale-up with simplified operational procedures.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alkyl Neopentylbenzene Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced photocatalytic methods in reshaping the production of high-value chemical intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory discoveries are seamlessly translated into robust industrial processes. Our commitment to quality is underscored by our stringent purity specifications and rigorous QC labs, which guarantee that every batch of alkyl neopentylbenzene derivatives meets the exacting standards required by the global pharmaceutical and fine chemical industries. We are equipped to handle the specific nuances of photochemical reactions, providing a safe and efficient environment for your most challenging synthesis projects.
We invite you to collaborate with us to leverage this cutting-edge technology for your next project. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific molecule. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our expertise can accelerate your development timeline and optimize your manufacturing costs. Let us be your trusted partner in navigating the complexities of modern synthetic chemistry.
