Advanced Manufacturing of 5,6-Epoxy-5,6-Dihydro-Beta-Ionol for Global Flavor and Pharma Markets
The chemical landscape for producing high-value terpene derivatives has evolved significantly with the introduction of patent CN101787003A, which details a robust preparation method for 5,6-epoxy-5,6-dihydro-beta-ionol. This specific aromatic terpene alcohol serves as a critical metabolite and degradation product found in various tobaccos and plants, acting as a pivotal intermediate for the flavor, fragrance, and pharmaceutical industries. The disclosed technology represents a paradigm shift from traditional oxidative pathways to a highly efficient reductive strategy, leveraging the synergistic effects of sodium borohydride and cerium chloride. By addressing the historical challenges of low conversion rates and product instability, this innovation provides a scalable solution for generating high-purity intermediates essential for complex aroma chemical synthesis.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 5,6-epoxy-5,6-dihydro-beta-ionol relied heavily on the epoxidation of beta-ionol using meta-chloroperbenzoic acid (mCPBA) in the presence of sodium bicarbonate and dichloromethane. This conventional oxidative route is fraught with significant inefficiencies, primarily characterized by a disappointingly low yield of approximately 60.97%, which severely impacts the economic viability of large-scale production. Furthermore, the intermediates involved in this oxidative pathway are notoriously unstable and prone to polymerization, leading to complex impurity profiles that necessitate rigorous and costly purification steps. The reliance on peracids also introduces safety hazards related to handling explosive oxidants, while the extended reaction times required to achieve even modest conversions create bottlenecks in manufacturing schedules, ultimately driving up the cost per kilogram for downstream users in the tobacco and fragrance sectors.
The Novel Approach
In stark contrast, the novel approach outlined in the patent utilizes a Luche-type reduction strategy, starting from 5,6-epoxy-5,6-dihydro-beta-ionone as the raw material. This reductive pathway employs a combination of sodium borohydride and cerium chloride heptahydrate (or anhydrous) in anhydrous methanol to selectively reduce the carbonyl group while preserving the sensitive epoxy and olefinic functionalities. This method dramatically improves the process efficiency, achieving yields ranging from 87.8% to 91.84%, which represents a substantial leap forward in material throughput compared to the legacy epoxidation methods. The reaction conditions are remarkably mild, proceeding effectively at room temperature or in an ice-water bath, which not only enhances operational safety but also simplifies the thermal management requirements for industrial reactors, thereby facilitating a smoother transition from laboratory bench to commercial plant.
Mechanistic Insights into CeCl3-Mediated Selective Reduction
The core of this technological breakthrough lies in the precise mechanistic role played by cerium chloride during the reduction process. In the absence of lanthanide salts, sodium borohydride typically reduces alpha,beta-unsaturated ketones via 1,4-addition, resulting in the saturation of the double bond rather than the desired alcohol formation. However, the presence of Ce(III) ions coordinates strongly with the carbonyl oxygen atom, increasing the electrophilicity of the carbonyl carbon and simultaneously blocking the conjugate addition pathway. This coordination creates a transient chelate that directs the hydride attack exclusively to the 1,2-position, ensuring the formation of the allylic alcohol without compromising the integrity of the adjacent carbon-carbon double bond or the epoxide ring.
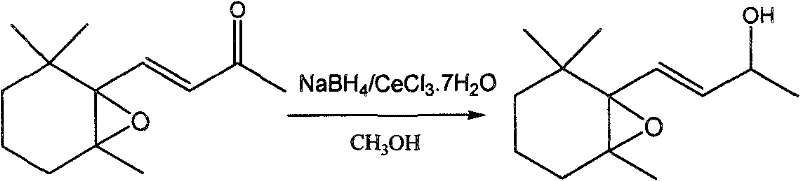
Furthermore, this mechanistic specificity is crucial for maintaining the stereochemical and structural fidelity required for high-end fragrance applications. The reaction proceeds through a six-membered transition state where the cerium ion bridges the carbonyl oxygen and the incoming hydride source, effectively lowering the activation energy for the 1,2-reduction. This controlled environment minimizes the formation of side products such as saturated alcohols or ring-opened byproducts, which are common pitfalls in non-catalyzed reductions of epoxy-enones. The result is a crude product with a GC content often exceeding 90%, significantly reducing the burden on downstream purification units and allowing for the isolation of colorless, high-purity 5,6-epoxy-5,6-dihydro-beta-ionol with purity levels greater than 95.00% after simple silica gel chromatography.
How to Synthesize 5,6-Epoxy-5,6-Dihydro-Beta-Ionol Efficiently
The synthesis protocol described in the patent offers a straightforward and reproducible pathway for manufacturing this valuable intermediate, suitable for both pilot-scale validation and full commercial deployment. The process begins with the dissolution of the ketone precursor and cerium chloride in methanol, followed by the controlled addition of the reducing agent to manage exothermicity. Detailed standardized operating procedures regarding stoichiometry, addition rates, and workup parameters are critical for maximizing yield and ensuring batch-to-batch consistency. For comprehensive technical guidance on executing this synthesis, please refer to the step-by-step instructions provided below.
- Dissolve 5,6-epoxy-5,6-dihydro-beta-ionone and cerium chloride in anhydrous methanol under ice-water bath or room temperature conditions.
- Slowly add sodium borohydride to the solution within 5 minutes while stirring, then continue stirring for 5 to 10 minutes to complete the selective reduction.
- Quench the reaction with dilute hydrochloric acid, extract with ether, dry over magnesium sulfate, and purify the crude product via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this Luche reduction methodology presents a compelling value proposition centered on yield optimization and process robustness. The dramatic increase in reaction yield from roughly 60% to over 90% directly translates to a substantial reduction in raw material consumption per unit of finished product, effectively lowering the variable cost of goods sold without requiring capital-intensive equipment upgrades. This efficiency gain is particularly impactful for high-volume applications in the tobacco and flavor industries, where margin pressures often dictate the selection of synthetic routes. Moreover, the elimination of hazardous peracids and the use of common, inexpensive reagents like sodium borohydride and methanol simplify the procurement of starting materials, reducing exposure to volatile specialty chemical markets and ensuring a more stable supply base.
- Cost Reduction in Manufacturing: The shift to a high-yield reductive pathway eliminates the significant material losses associated with the traditional epoxidation route, leading to profound cost savings in raw material acquisition. By avoiding the use of expensive and potentially hazardous oxidizing agents like m-chloroperbenzoic acid, manufacturers can drastically reduce waste disposal costs and safety compliance expenditures. The simplified reaction profile also means shorter cycle times and higher reactor utilization rates, allowing facilities to produce more tonnage with existing infrastructure, thereby driving down the fixed cost allocation per kilogram of the final fragrance intermediate.
- Enhanced Supply Chain Reliability: Utilizing widely available commodity chemicals such as methanol and sodium borohydride mitigates the risk of supply disruptions often caused by reliance on niche oxidants. The robustness of the reaction conditions, which tolerate both room temperature and ice-bath cooling, provides operational flexibility that ensures consistent production output regardless of minor fluctuations in utility availability. This reliability is critical for maintaining continuous supply lines to major multinational clients in the flavor and pharma sectors, where interruptions can have cascading effects on downstream formulation schedules and product launches.
- Scalability and Environmental Compliance: The aqueous workup and standard solvent extraction techniques employed in this process are inherently scalable, posing fewer engineering challenges during the transition from laboratory to multi-ton production compared to sensitive oxidation reactions. The reduction in hazardous waste generation, specifically the avoidance of chlorinated benzoic acid byproducts, aligns with increasingly stringent environmental regulations and corporate sustainability goals. This greener profile not only facilitates easier permitting for new production lines but also enhances the marketability of the final product to eco-conscious brands seeking sustainable sourcing options for their fragrance and tobacco additives.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of 5,6-epoxy-5,6-dihydro-beta-ionol, derived directly from the patented methodology. These insights are intended to clarify the operational benefits and quality standards associated with this advanced synthesis route. Understanding these details is essential for stakeholders evaluating the feasibility of integrating this intermediate into their supply chains.
Q: What is the primary advantage of the Luche reduction method over traditional epoxidation for this compound?
A: The Luche reduction method utilizing NaBH4 and CeCl3 offers a significantly higher yield (87.8% to 91.84%) compared to the traditional m-chloroperbenzoic acid epoxidation route, which typically yields only around 60.97%. Additionally, the reduction pathway avoids the stability issues associated with polymerization often seen in direct epoxidation processes.
Q: How does the use of Cerium Chloride improve the selectivity of the reaction?
A: Cerium chloride acts as a Lewis acid that coordinates with the carbonyl oxygen, enhancing the electrophilicity of the carbonyl carbon. This coordination facilitates the 1,2-addition of the hydride from sodium borohydride, ensuring selective reduction of the ketone to the alcohol without affecting the conjugated double bond or the sensitive epoxy ring.
Q: What purity levels can be achieved with this synthesis protocol?
A: Following the standard workup and silica gel column purification described in the patent, the final product consistently achieves a purity greater than 95.00%, with optimized examples demonstrating purity levels exceeding 98.00%, making it suitable for high-end fragrance and pharmaceutical applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5,6-Epoxy-5,6-Dihydro-Beta-Ionol Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-purity intermediates in the development of premium fragrances and pharmaceutical formulations. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the sophisticated Luche reduction chemistry described in patent CN101787003A is executed with precision and consistency. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of 5,6-epoxy-5,6-dihydro-beta-ionol meets the exacting standards required by global regulatory bodies and end-user applications.
We invite you to collaborate with our technical team to explore how this optimized synthesis route can enhance your product portfolio and reduce overall manufacturing costs. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and detailed route feasibility assessments to support your R&D and sourcing decisions, ensuring a seamless integration of this high-value intermediate into your supply chain.
