Advancing Pharmaceutical Intermediate Manufacturing with Efficient Copper-Catalyzed Isoquinolinone Synthesis
The pharmaceutical and fine chemical industries are constantly seeking robust, scalable, and economically viable pathways to access complex heterocyclic scaffolds that serve as critical building blocks for bioactive molecules. Patent CN110028448B introduces a transformative preparation method for 3-hydroxy-2,3-dihydroisoquinoline-1,4-dione compounds, a class of structures renowned for their potent anti-inflammatory, analgesic, and antitumor properties. This innovation addresses long-standing challenges in organic synthesis by replacing expensive and hazardous reagents with a benign, copper-catalyzed oxidative system. By leveraging molecular oxygen or air as the terminal oxidant, this technology not only aligns with green chemistry principles but also offers a streamlined route for the commercial scale-up of complex pharmaceutical intermediates. The structural versatility of this approach allows for the efficient generation of diverse derivatives, making it an invaluable asset for R&D teams focused on drug discovery and process optimization.
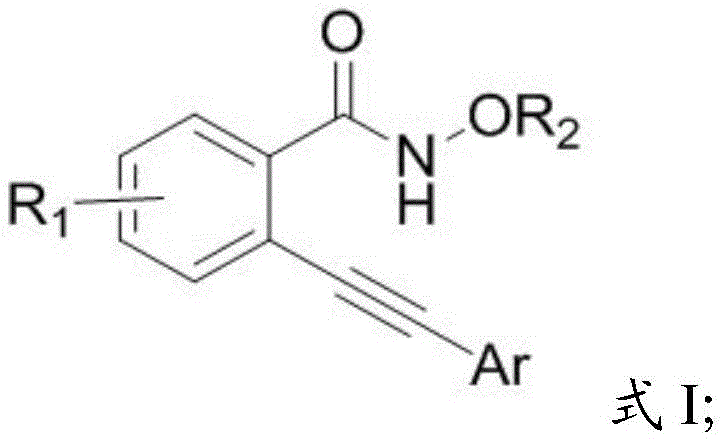
The core of this invention lies in the conversion of N-alkoxy-2-alkynylbenzamides, represented by Formula I, into the corresponding 3-hydroxy-2,3-dihydroisoquinoline-1,4-diones, shown as Formula II. This transformation is achieved through a concise one-step cyclization that avoids the use of strong acids and precious metal catalysts. For procurement managers and supply chain directors, the implications are profound: the shift from noble metals to base metals drastically alters the cost structure of production while simplifying the regulatory burden associated with heavy metal residues in final API products. This patent provides a clear pathway for cost reduction in pharmaceutical intermediate manufacturing by utilizing readily available copper salts and common organic solvents, ensuring a reliable supply chain for high-value heterocyclic compounds.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 3-hydroxy-2,3-dihydroisoquinoline-1,4-dione derivatives has relied on methodologies that present significant operational and economic hurdles for large-scale production. One prominent prior art method utilizes hypervalent iodine reagents, such as PIFA (phenyliodine(III) bis(trifluoroacetate)), to mediate the cyclization of o-(1-alkynyl)benzamides. While effective on a laboratory scale, this approach requires stoichiometric amounts (typically 2.0 equivalents) of expensive iodine(III) species, which generates substantial waste and inflates raw material costs prohibitively for industrial applications. Furthermore, alternative routes employing palladium acetate catalysts often necessitate harsh acidic conditions to facilitate the reaction. These strong acids impose severe constraints on reactor metallurgy, requiring specialized corrosion-resistant equipment that increases capital expenditure. Additionally, the use of palladium introduces the risk of toxic metal contamination, mandating rigorous and costly purification steps to meet stringent pharmaceutical purity specifications, thereby extending production lead times and complicating the overall process workflow.
The Novel Approach
In stark contrast, the methodology disclosed in CN110028448B revolutionizes this synthetic landscape by employing a catalytic system based on earth-abundant copper salts under an oxidative atmosphere. This novel approach eliminates the dependency on stoichiometric hypervalent iodine oxidants and expensive palladium catalysts, replacing them with inexpensive copper sources like cupric chloride, cuprous oxide, or cuprous bromide. The reaction proceeds smoothly under mild thermal conditions, typically between 40°C and 80°C, without the need for corrosive strong acids. This gentler reaction environment not only preserves the integrity of standard stainless-steel manufacturing equipment but also significantly reduces the generation of hazardous waste streams. By enabling a direct, one-step synthesis from readily accessible N-alkoxy-2-alkynylbenzamide precursors, this method offers a streamlined, high-efficiency route that is ideally suited for the commercial scale-up of complex polymer additives and pharmaceutical intermediates alike, ensuring consistent quality and supply continuity.
Mechanistic Insights into Copper-Catalyzed Oxidative Cyclization
The mechanistic elegance of this transformation lies in the ability of the copper catalyst to activate the alkyne moiety towards nucleophilic attack by the adjacent amide oxygen or nitrogen, facilitated by the oxidative environment. Under an atmosphere of oxygen or air, the copper species likely cycles between oxidation states, promoting the formation of reactive intermediates that drive the intramolecular cyclization. This oxidative cyclization effectively constructs the isoquinoline-1,4-dione core while simultaneously installing the hydroxyl group at the 3-position. The tolerance of the catalytic system to various functional groups is a testament to its robustness; electron-withdrawing groups such as fluorine, chlorine, or bromine, as well as electron-donating groups like methoxy or methyl on the aromatic ring, are well-tolerated. Similarly, the alkyne terminus can bear diverse aryl groups, including phenyl, thienyl, and naphthyl moieties, without compromising reaction efficiency. This broad substrate scope is critical for medicinal chemists who require rapid access to structural analogs for structure-activity relationship (SAR) studies.
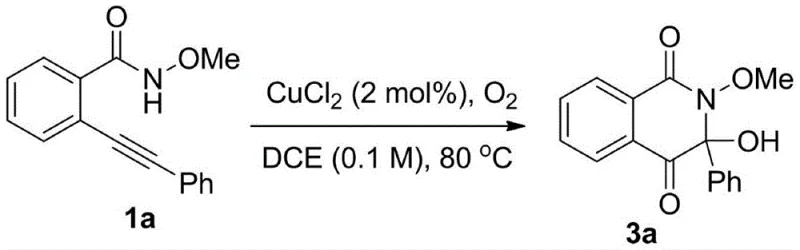
Furthermore, the impurity profile of the resulting products is significantly improved compared to acid-mediated methods. The absence of strong acids minimizes side reactions such as polymerization or decomposition of sensitive functional groups, leading to cleaner reaction mixtures. The use of molecular oxygen as the oxidant ensures that the only byproduct is water, aligning with green chemistry metrics and simplifying downstream processing. For R&D directors, understanding this mechanism highlights the potential for developing highly selective processes that minimize the formation of difficult-to-remove impurities. The ability to control the reaction outcome through simple adjustments in temperature and catalyst loading provides a high degree of process controllability, which is essential for maintaining stringent purity specifications required in the production of high-purity OLED materials or active pharmaceutical ingredients.
How to Synthesize 3-Hydroxy-2,3-dihydroisoquinoline-1,4-dione Efficiently
The practical implementation of this synthesis is designed for ease of operation, making it accessible for both laboratory research and pilot plant operations. The process begins with the preparation of the N-alkoxy-2-alkynylbenzamide starting material, which can be synthesized via standard Sonogashira coupling followed by amidation, as detailed in the patent examples. Once the substrate is secured, the core cyclization step involves mixing the substrate with a catalytic amount of copper salt in a suitable organic solvent. The reaction is then heated under an oxygen or air atmosphere, allowing the oxidative cyclization to proceed to completion. Detailed standardized synthesis steps for this procedure are provided in the guide below, ensuring reproducibility and safety during execution.
- Mix N-alkoxy-2-alkynylbenzamide substrate with a copper salt catalyst (such as CuCl2, Cu2O, or CuBr) in an organic solvent like 1,2-dichloroethane or acetonitrile.
- Conduct the synthesis reaction under an oxygen or air atmosphere at temperatures ranging from 40°C to 80°C for approximately 9 to 11 hours.
- Upon completion, filter the reaction mixture and purify the crude product via column chromatography to isolate the target 3-hydroxy-2,3-dihydroisoquinoline-1,4-dione compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this copper-catalyzed technology offers tangible strategic benefits that extend beyond simple yield improvements. The shift from precious metal catalysts to base metals fundamentally alters the cost dynamics of the supply chain, reducing exposure to the volatile pricing of palladium and rhodium. Moreover, the simplified workup procedures and reduced equipment corrosion risks translate into lower operational expenditures and extended asset life. This technology enables manufacturers to offer cost reduction in electronic chemical manufacturing and pharma sectors by delivering high-quality intermediates at a more competitive price point, securing a stronger market position.
- Cost Reduction in Manufacturing: The replacement of expensive palladium catalysts and stoichiometric hypervalent iodine reagents with low-cost copper salts results in a drastic reduction in raw material costs. Since copper is abundant and inexpensive compared to noble metals, the overall cost of goods sold (COGS) is significantly lowered. Additionally, the elimination of strong acids reduces the need for specialized corrosion-resistant reactors and extensive neutralization waste treatment, further driving down operational expenses. This economic efficiency allows for the production of high-purity pharmaceutical intermediates with a much healthier margin, making the final products more competitive in the global marketplace.
- Enhanced Supply Chain Reliability: The reliance on readily available and stable reagents such as cupric chloride and molecular oxygen ensures a robust and uninterrupted supply chain. Unlike hypervalent iodine compounds, which can be hazardous to transport and store, the reagents used in this process are common industrial chemicals with established global supply networks. This availability minimizes the risk of production delays caused by raw material shortages. Furthermore, the mild reaction conditions reduce the likelihood of runaway reactions or equipment failures, ensuring consistent delivery schedules and reducing lead time for high-purity specialty chemicals to reach downstream customers.
- Scalability and Environmental Compliance: The process is inherently scalable due to its simplicity and the use of benign oxidants like air or oxygen. The absence of toxic heavy metal waste streams and corrosive acid byproducts simplifies environmental compliance and waste disposal, which is increasingly critical in modern manufacturing. The straightforward workup involving filtration and column chromatography is easily adaptable to large-scale continuous flow or batch processes. This scalability ensures that the technology can meet growing market demands for reliable agrochemical intermediate supplier needs without compromising on environmental standards or safety protocols.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this copper-catalyzed synthesis. These answers are derived directly from the technical specifications and experimental data provided in the patent documentation, offering clarity on reaction conditions, substrate compatibility, and process safety. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this method into their existing production workflows.
Q: What are the primary cost advantages of this copper-catalyzed method compared to traditional palladium systems?
A: The method utilizes inexpensive copper salts instead of costly palladium catalysts or hypervalent iodine reagents, significantly reducing raw material expenses and eliminating the need for expensive heavy metal removal processes.
Q: Does this synthesis require harsh acidic conditions that could damage production equipment?
A: No, the process operates under mild conditions without strong acids, which reduces corrosion risks for reactor vessels and lowers maintenance costs for manufacturing facilities.
Q: What is the substrate scope for the N-alkoxy-2-alkynylbenzamide starting materials?
A: The method demonstrates broad applicability, tolerating various electron-withdrawing and electron-donating groups on the benzene ring, as well as different aryl substituents on the alkyne, including phenyl, thienyl, and naphthyl groups.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Hydroxy-2,3-dihydroisoquinoline-1,4-dione Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and sustainable synthetic routes in the development of next-generation therapeutics and functional materials. Our team of expert chemists has thoroughly analyzed the potential of the copper-catalyzed oxidative cyclization described in CN110028448B and is fully prepared to leverage this technology for your specific project needs. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop discovery to full-scale manufacturing is seamless. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of 3-hydroxy-2,3-dihydroisoquinoline-1,4-dione meets the highest industry standards.
We invite you to collaborate with us to optimize your supply chain and reduce manufacturing costs through this innovative chemistry. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us today to obtain specific COA data and comprehensive route feasibility assessments that demonstrate how our expertise can accelerate your project timelines and enhance your product competitiveness in the global market.
