Advanced Solvent-Free Synthesis of Acyl Ferrocene Schiff Bases for Commercial Scale-up
Introduction to Next-Generation Ferrocene Derivative Manufacturing
The chemical industry is currently witnessing a paradigm shift towards greener, more efficient synthetic methodologies, particularly in the realm of organometallic intermediates. Patent CN103880892A introduces a groundbreaking approach to synthesizing acyl ferrocene hydrazinodithioformate Schiff bases, a class of compounds renowned for their potential applications in pharmaceuticals and functional materials. Unlike traditional methods that rely heavily on liquid-phase reactions with substantial solvent loads, this innovation leverages a solid-phase, solvent-free technique that drastically simplifies the operational workflow. The core structure of these valuable intermediates, characterized by the robust ferrocenyl moiety linked to a hydrazinodithioformate group via an imine bond, is depicted below, highlighting the versatility where R can be hydrogen or a methyl group and R' represents variable alkyl or aryl substituents.
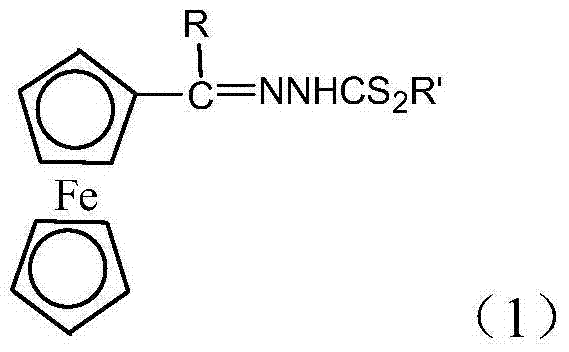
This technological advancement addresses critical pain points in modern chemical manufacturing, specifically the need for reduced environmental footprint and enhanced process safety. By utilizing p-toluenesulfonic acid (PTSA) as a solid acid catalyst, the process achieves reaction yields exceeding 97% under mild thermal conditions, typically between 75°C and 80°C. For R&D directors and procurement specialists seeking a reliable pharma intermediate supplier, this method offers a compelling value proposition: it transforms a complex, multi-step purification challenge into a streamlined grinding and heating operation. The elimination of bulk solvents during the reaction phase not only cuts down on volatile organic compound (VOC) emissions but also removes the energy-intensive step of solvent distillation and recovery, positioning this technology as a leader in sustainable fine chemical synthesis.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Schiff bases containing ferrocenyl groups has been dominated by liquid-phase protocols that utilize ethanol or similar organic solvents as the reaction medium. These traditional approaches suffer from inherent inefficiencies, including prolonged reaction times that can extend over many hours or even days to reach completion. The reliance on liquid solvents necessitates complex downstream processing, involving extensive washing, filtration, and often multiple recrystallization steps to remove residual solvent and unreacted starting materials. Furthermore, the use of liquid acid catalysts in these conventional methods can lead to equipment corrosion and generates significant amounts of acidic wastewater, posing severe environmental compliance challenges for manufacturing facilities. The overall yield in these liquid-phase processes is often inconsistent and frequently lower than desired, impacting the cost-effectiveness of producing high-purity OLED material or pharmaceutical precursors on a commercial scale.
The Novel Approach
In stark contrast, the novel solid-phase solvent-free method described in the patent data revolutionizes the production landscape by mechanizing the reaction through mechanical grinding. This approach utilizes the mechanical force generated in a mortar or similar grinding apparatus to intimately mix the reactants—acylferrocene, hydrazinodithioformate, and the PTSA catalyst—at the molecular level without the need for a liquid medium. The reaction is subsequently driven by mild heating at 75-80°C for a short duration of 1 to 1.5 hours, which is significantly faster than conventional heating methods. This transition to a solvent-free environment not only accelerates the reaction kinetics but also simplifies the post-reaction workup to a simple washing and filtration step. For supply chain heads concerned with the commercial scale-up of complex polymer additives or intermediates, this simplicity translates directly into reduced operational complexity and lower capital expenditure on specialized solvent-handling infrastructure.
Mechanistic Insights into Solid-Phase Acid-Catalyzed Condensation
The underlying chemical mechanism of this synthesis involves a classic condensation reaction between the carbonyl group of the acylferrocene and the primary amino group of the hydrazinodithioformate, facilitated by the Brønsted acidity of p-toluenesulfonic acid. In the solid state, the grinding process serves a dual purpose: it reduces particle size to increase the surface area for contact and provides the activation energy necessary to initiate the nucleophilic attack. The PTSA catalyst protonates the carbonyl oxygen, making the carbonyl carbon more electrophilic and thus more susceptible to attack by the nitrogen lone pair of the hydrazine derivative. This mechanistic pathway is highly efficient because the absence of solvent molecules prevents the solvation of ions that might otherwise stabilize intermediates and slow down the reaction rate. The result is a rapid formation of the imine (C=N) bond, which is the defining characteristic of the Schiff base structure, as clearly illustrated in the reaction scheme below.
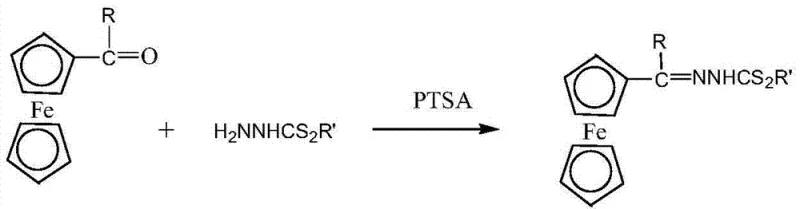
From an impurity control perspective, the solid-phase nature of this reaction offers distinct advantages over solution chemistry. In liquid-phase synthesis, side reactions such as hydrolysis or oligomerization can occur due to the mobility of species in the solvent. However, in the constrained environment of the solid lattice during grinding and heating, the reactants are held in close proximity in a specific stoichiometric ratio (2:2:1~1.2), which favors the desired bimolecular condensation over competing pathways. The high selectivity of the PTSA catalyst further ensures that the ferrocenyl ring system remains intact, preserving the aromaticity and electrochemical properties that make these derivatives valuable. This precise control over the reaction environment leads to the reported yields of over 97%, minimizing the formation of by-products and reducing the burden on purification units, which is a critical factor for maintaining high-purity specifications in electronic chemical manufacturing.
How to Synthesize Acyl Ferrocene Hydrazinodithioformate Schiff Base Efficiently
The implementation of this synthesis route requires careful attention to the physical state of the reactants and the efficiency of the mechanical mixing process. The patent outlines a straightforward protocol that begins with the dry grinding of the three key components in a mortar, ensuring a homogeneous mixture before thermal treatment. This initial mechanical activation is crucial for breaking down crystal lattices and initiating the solid-state diffusion required for the reaction to proceed. Following the grinding step, the mixture is subjected to a controlled thermal incubation, after which the crude product is isolated through a simple washing procedure with aqueous ethanol. The detailed standardized synthesis steps, including specific molar ratios and temperature profiles for different R and R' substituents, are provided in the guide below to assist technical teams in replicating this high-yield process.
- Grind equimolar amounts of hydrazinodithioformate, acylferrocene, and p-toluenesulfonic acid catalyst in a dry mortar at room temperature for 5-15 minutes.
- Transfer the mixture to an oven and maintain the temperature between 75-80°C for 1 to 1.5 hours to complete the condensation reaction.
- Cool the mixture, wash with 90-95% ethanol, filter, and recrystallize the filter cake using absolute ethanol to obtain the pure product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain strategists, the adoption of this solvent-free technology presents a multitude of tangible benefits that directly impact the bottom line and operational resilience. The most significant advantage lies in the drastic reduction of raw material costs associated with organic solvents; by eliminating the need for bulk ethanol during the reaction phase, manufacturers can achieve substantial cost savings in both material procurement and waste disposal. Additionally, the simplified equipment requirements—essentially needing only grinding machinery and standard ovens rather than complex reactors with reflux condensers and solvent recovery columns—lower the barrier to entry for production and reduce maintenance overheads. This lean manufacturing approach enhances supply chain reliability by minimizing the dependencies on volatile solvent markets and reducing the risk of production stoppages due to equipment failure or regulatory restrictions on VOC emissions.
- Cost Reduction in Manufacturing: The elimination of solvent usage during the reaction phase fundamentally alters the cost structure of producing these ferrocene derivatives. Without the need to purchase, store, and recover large volumes of ethanol, the operational expenditure (OPEX) is significantly lowered. Furthermore, the high reaction yield of over 97% means that raw material utilization is maximized, reducing the cost per kilogram of the final active intermediate. The ability to reuse the solid acid catalyst also contributes to long-term cost efficiency, as it minimizes the consumption of catalytic reagents and reduces the volume of hazardous waste requiring neutralization and disposal.
- Enhanced Supply Chain Reliability: The simplicity of the process parameters, which operate at mild temperatures and atmospheric pressure, ensures a robust and stable production workflow. This reliability is crucial for meeting tight delivery schedules for high-purity agrochemical intermediates or pharmaceutical precursors. Since the process does not rely on complex distillation towers or cryogenic cooling systems, the risk of unplanned downtime is minimized. Moreover, the raw materials—acylferrocenes and hydrazinodithioformates—are commercially available and stable, ensuring a continuous supply feedstock that is not subject to the same logistical volatility as specialized solvents or sensitive reagents.
- Scalability and Environmental Compliance: Scaling this process from laboratory to industrial production is inherently safer and more straightforward due to the absence of flammable solvent vapors during the reaction. This aligns perfectly with increasingly stringent global environmental regulations regarding VOC emissions and hazardous waste generation. The 'green' nature of this synthesis, characterized by atom economy and waste reduction, positions manufacturers favorably for sustainability certifications and audits. The ease of scale-up allows for flexible production capacities, enabling suppliers to respond rapidly to fluctuating market demands without the need for massive capital investment in new infrastructure.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of acyl ferrocene hydrazinodithioformate Schiff bases. These answers are derived directly from the patented methodology and are intended to provide clarity on the feasibility and advantages of this solid-state synthesis route. Understanding these details is essential for technical teams evaluating the integration of this technology into their existing manufacturing portfolios or for procurement officers assessing the quality and consistency of the supply.
Q: What are the advantages of the solid-phase method over traditional liquid-phase synthesis for Schiff bases?
A: The solid-phase method eliminates the need for large volumes of organic solvents like ethanol during the reaction phase, significantly reducing waste generation and solvent recovery costs. It also shortens reaction times and simplifies post-treatment, leading to higher overall yields exceeding 97%.
Q: Can this synthesis method be scaled for industrial production?
A: Yes, the process utilizes simple mechanical grinding and standard heating equipment (ovens), avoiding high-pressure or complex reactor requirements. This low equipment demand makes it highly suitable for commercial scale-up with minimal capital expenditure.
Q: What is the role of p-toluenesulfonic acid (PTSA) in this reaction?
A: PTSA acts as a solid acid catalyst that accelerates the condensation between the carbonyl group of the acylferrocene and the amino group of the hydrazine derivative. Its solid nature allows for easy mixing in the solvent-free environment and avoids the corrosion issues associated with liquid mineral acids.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Acyl Ferrocene Hydrazinodithioformate Schiff Base Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this solvent-free synthesis technology in delivering high-value ferrocene derivatives to the global market. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the benefits of this innovative method are fully realized at an industrial level. Our state-of-the-art facilities are equipped to handle solid-phase reactions with precision, adhering to stringent purity specifications and rigorous QC labs to guarantee that every batch meets the exacting standards required for pharmaceutical and electronic applications. We are committed to leveraging our technical expertise to optimize this green chemistry route for our clients, delivering products that are not only chemically superior but also economically and environmentally sustainable.
We invite you to collaborate with us to explore how this advanced synthesis method can enhance your supply chain efficiency and product quality. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and application needs. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us demonstrate how our commitment to innovation can drive value for your organization.
