Advanced Manufacturing of Entrectinib Intermediates via Novel Grignard Cyclization Technology
The pharmaceutical landscape for oncology treatments continues to evolve rapidly, with kinase inhibitors like Entrectinib playing a pivotal role in targeting ALK, ROS1, and NTRK gene fusions. Central to the supply chain of this critical medication is the efficient production of its key building block, 3-amino-5-(3,5-difluorobenzyl)-1H-indazole. Recent intellectual property developments, specifically patent CN113801062B, have unveiled a transformative synthetic methodology that addresses long-standing bottlenecks in manufacturing this high-value pharmaceutical intermediate. This technical insight report analyzes the strategic implications of this novel Grignard-based route, contrasting it against legacy technologies to highlight opportunities for cost optimization and supply chain resilience. For R&D directors and procurement strategists, understanding these mechanistic shifts is essential for securing a reliable pharmaceutical intermediate supplier capable of delivering high-purity materials at a competitive margin.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of 3-amino-5-(3,5-difluorobenzyl)-1H-indazole has relied heavily on palladium-catalyzed cross-coupling reactions, specifically the Suzuki-Miyaura protocol. As illustrated in the prior art referenced within the patent background, this conventional pathway necessitates the coupling of 1-bromomethyl-3,5-difluorobenzene with 3-cyano-4-fluorophenylboronic acid. This approach presents severe logistical and economic challenges for large-scale operations. Firstly, the reliance on 3-cyano-4-fluorophenylboronic acid introduces a significant cost burden, as functionalized boronic acids are inherently more expensive than their halogenated counterparts due to complex synthesis requirements. Secondly, the use of 1-bromomethyl-3,5-difluorobenzene poses substantial occupational health risks; it is a potent lachrymator that causes severe irritation to the skin and respiratory tract, demanding rigorous and costly containment engineering controls. Finally, the dependence on palladium catalysts not only inflates raw material costs but also necessitates extensive downstream purification steps to meet stringent heavy metal limits required for API manufacturing.
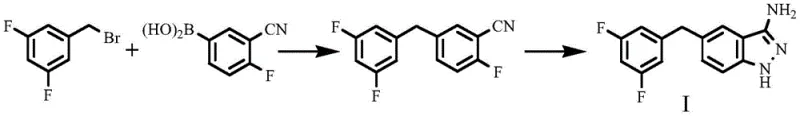
The Novel Approach
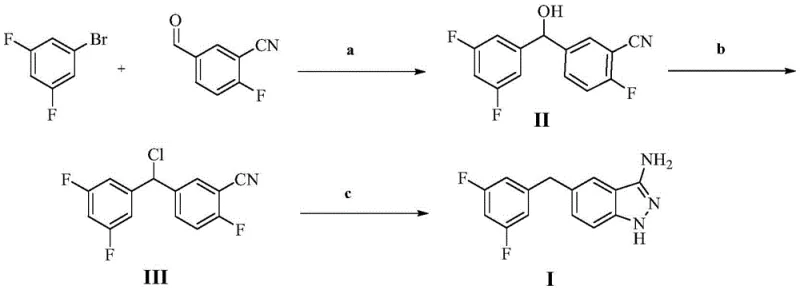
In stark contrast, the methodology disclosed in patent CN113801062B represents a paradigm shift towards atom-economical and operationally simpler chemistry. This innovative route bypasses the need for precious metal catalysis entirely by leveraging classical Grignard chemistry followed by a unique cyclization-reduction sequence. The process initiates with the formation of a Grignard reagent from the significantly cheaper and safer 1-bromo-3,5-difluorobenzene. This organometallic species undergoes nucleophilic addition to 2-fluoro-5-formylbenzonitrile, constructing the carbon-carbon bond framework efficiently. The subsequent steps involve a straightforward halogenation of the benzylic alcohol followed by a remarkable tandem reaction with hydrazine hydrate. This new strategy not only drastically reduces the cost of goods sold (COGS) by eliminating expensive catalysts and ligands but also simplifies the purification profile, making it an ideal candidate for cost reduction in API manufacturing where margin compression is a constant pressure.
Mechanistic Insights into Grignard Addition and Tandem Cyclization
The core of this technological advancement lies in the precise control of reaction conditions across three distinct stages, ensuring high selectivity and yield. The initial step involves the generation of the Grignard reagent in tetrahydrofuran (THF), preferred for its ability to stabilize the organomagnesium species. The patent specifies a reaction temperature range of -20°C to 60°C, with an optimal addition temperature of -10°C for the nucleophilic attack on the aldehyde carbonyl of 2-fluoro-5-formylbenzonitrile. This low-temperature control is critical for minimizing side reactions such as Wurtz-type coupling or over-addition, ensuring the formation of the secondary alcohol intermediate (Formula II) with high fidelity. The use of THF is particularly advantageous from a process chemistry perspective, as it allows the crude reaction mixture to be potentially telescoped into the next step without rigorous isolation, thereby saving time and solvent volume.
The final transformation is the most mechanistically intriguing aspect of this patent, offering a streamlined path to the indazole core. Instead of a multi-step sequence involving separate cyclization and reduction phases, the process utilizes hydrazine hydrate to effect both transformations simultaneously. In this step, the benzylic chloride (Formula III) reacts with hydrazine to form the indazole ring via nucleophilic aromatic substitution and condensation. Concurrently, the hydrazine acts as a reducing agent to cleave the benzylic carbon-chlorine bond, effectively dehalogenating the molecule to yield the final methylene-linked structure (Formula I). This metal-free reductive cyclization is a rare and valuable transformation in heterocyclic chemistry, eliminating the need for hydrogenation equipment or stoichiometric metal hydrides, which significantly enhances the safety and scalability of the commercial scale-up of complex pharmaceutical intermediates.
How to Synthesize 3-Amino-5-(3,5-difluorobenzyl)-1H-indazole Efficiently
Implementing this novel synthetic route requires strict adherence to the optimized parameters outlined in the patent to ensure reproducibility and safety. The process is designed to be robust, utilizing common industrial solvents and reagents that are readily available in the global chemical supply chain. The following guide summarizes the critical operational windows defined in the intellectual property, serving as a foundational reference for process engineers aiming to replicate or adapt this technology for pilot plant trials. For detailed standard operating procedures and specific batch records, please refer to the technical documentation provided below.
- Prepare a Grignard reagent from 1-bromo-3,5-difluorobenzene and magnesium in THF, then perform nucleophilic addition to 2-fluoro-5-formylbenzonitrile at -10°C to obtain the benzyl alcohol intermediate (Formula II).
- Conduct a nucleophilic substitution on Formula II using thionyl chloride in dichloromethane at room temperature to convert the hydroxyl group into a chloride, yielding Formula III.
- React Formula III with hydrazine hydrate in THF at 60°C to effect simultaneous indazole ring cyclization and reductive dehalogenation, yielding the final target compound (Formula I).
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this Grignard-based methodology offers tangible strategic benefits beyond mere technical novelty. The shift away from palladium catalysis and specialized boronic acids directly translates to a more resilient and cost-effective supply chain. By utilizing commodity chemicals like bromobenzene derivatives and aldehydes, manufacturers can mitigate the risk of price volatility associated with specialized fine chemical building blocks. Furthermore, the elimination of heavy metal catalysts removes a major bottleneck in the quality control workflow, as extensive testing and purification to remove trace metals are no longer required, accelerating the release of batches for downstream API synthesis.
- Cost Reduction in Manufacturing: The economic argument for this new route is compelling, driven primarily by the substitution of high-cost inputs with commodity reagents. By removing the requirement for palladium catalysts, which are subject to significant market fluctuation and require expensive recovery systems, the overall variable cost of production is substantially lowered. Additionally, the avoidance of 3-cyano-4-fluorophenylboronic acid, a premium-priced starting material, further drives down the raw material bill. The simplified workup procedures, particularly the potential for telescoping steps in THF, reduce solvent consumption and labor hours, contributing to significant operational expenditure savings without compromising yield.
- Enhanced Supply Chain Reliability: From a sourcing perspective, this method relies on 1-bromo-3,5-difluorobenzene and 2-fluoro-5-formylbenzonitrile, which are produced by multiple suppliers globally, ensuring a diversified and secure supply base. Unlike the conventional route which depends on specific, potentially single-source boronic acids, this approach leverages widely available halogenated aromatics. Moreover, by eliminating the use of the hazardous lachrymator 1-bromomethyl-3,5-difluorobenzene, the process reduces regulatory compliance burdens and transportation restrictions, facilitating smoother logistics and reducing lead time for high-purity pharmaceutical intermediates.
- Scalability and Environmental Compliance: The environmental profile of this synthesis is markedly improved, aligning with modern green chemistry principles and increasingly strict environmental regulations. The absence of heavy metals simplifies waste stream treatment, reducing the cost and complexity of effluent management. The reaction conditions are mild, typically proceeding at or near ambient temperatures for the halogenation step and moderate heating for the cyclization, which lowers energy consumption. This inherent safety and environmental friendliness make the process highly scalable, allowing for seamless transition from kilogram-scale R&D batches to multi-ton commercial production with minimal engineering modifications.
Frequently Asked Questions (FAQ)
To assist technical decision-makers in evaluating the feasibility of this technology, we have compiled answers to common inquiries regarding the synthesis and application of this intermediate. These responses are derived directly from the experimental data and beneficial effects reported in the patent literature, providing a transparent view of the process capabilities. Understanding these technical nuances is crucial for assessing the fit of this route within your existing manufacturing infrastructure.
Q: What are the primary advantages of this new synthesis route over the conventional Suzuki coupling method?
A: The novel route eliminates the need for expensive palladium catalysts and costly boronic acid raw materials. Furthermore, it avoids the use of 1-bromomethyl-3,5-difluorobenzene, a hazardous lachrymator, thereby improving operational safety and reducing environmental impact.
Q: How does the process achieve indazole ring formation without metal catalysts?
A: The process utilizes a unique one-pot transformation where hydrazine hydrate acts as both the cyclization agent and the reducing agent. This allows for the simultaneous construction of the indazole core and the removal of the benzylic halogen without requiring transition metals.
Q: Is this method suitable for large-scale commercial production?
A: Yes, the method employs cheap, readily available raw materials and mild reaction conditions. The use of THF as a solvent in the first step allows for telescoping without intermediate purification, significantly simplifying the workflow for industrial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Amino-5-(3,5-difluorobenzyl)-1H-indazole Supplier
As the demand for next-generation kinase inhibitors continues to surge, securing a manufacturing partner with deep technical expertise in complex heterocyclic synthesis is paramount. NINGBO INNO PHARMCHEM stands at the forefront of this sector, possessing extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped to handle the specific requirements of Grignard chemistry and hydrazine-mediated reactions with the utmost safety and precision. We maintain stringent purity specifications and operate rigorous QC labs to ensure that every batch of 3-amino-5-(3,5-difluorobenzyl)-1H-indazole meets the exacting standards required for oncology drug development.
We invite global pharmaceutical partners to collaborate with us to leverage this innovative synthetic route for your supply chain. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out today to obtain specific COA data and comprehensive route feasibility assessments, ensuring that your project moves forward with the most efficient and economically viable chemistry available in the market.
