Advanced Synthesis of 7-Methoxy-1-Naphthylacetonitrile: A Cost-Effective Route for Agomelatine Intermediates
Advanced Synthesis of 7-Methoxy-1-Naphthylacetonitrile: A Cost-Effective Route for Agomelatine Intermediates
The pharmaceutical industry continuously seeks robust, scalable, and economically viable synthetic routes for key active pharmaceutical ingredient (API) intermediates. A recent technological breakthrough detailed in patent CN113527139A introduces a superior methodology for synthesizing 7-methoxy-1-naphthylacetonitrile, a critical precursor for the antidepressant agomelatine. This innovation addresses long-standing challenges in organic synthesis by replacing hazardous reagents and expensive catalysts with a mild, three-step sequence involving condensation, halogenation, and dehydrohalogenation. By shifting away from traditional oxidative aromatization methods that rely on costly quinones or precious metals, this process offers a compelling value proposition for manufacturers aiming to optimize their supply chains. The technical significance of this patent lies not only in its chemical elegance but also in its potential to redefine the cost structure and safety profile of producing high-purity pharmaceutical intermediates on a commercial scale.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
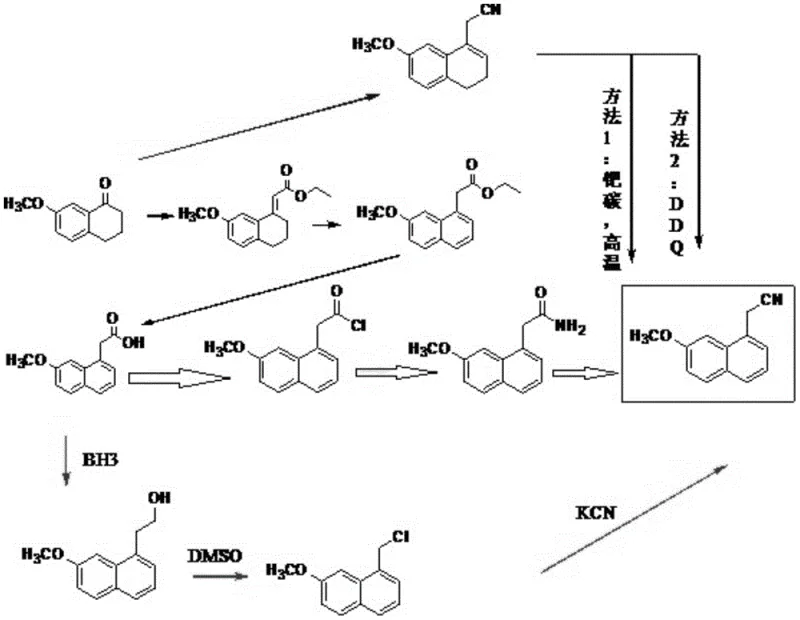
Historically, the synthesis of 7-methoxy-1-naphthylacetonitrile has been plagued by significant operational and economic hurdles that hinder efficient large-scale production. As illustrated in the comparative reaction schemes below, prior art methods often necessitate the use of extremely hazardous reagents such as butyl lithium, which poses severe safety risks due to its pyrophoric nature and requires specialized handling infrastructure. Furthermore, other established routes depend heavily on 2,3-dichloro-5,6-dicyano-1,4-benzoquinone (DDQ) for the crucial dehydrogenation step; while effective, DDQ is prohibitively expensive and generates substantial quantities of toxic phenolic by-products that complicate waste management and increase environmental compliance costs. Additionally, methods utilizing palladium on carbon for high-temperature dehydrogenation introduce the risk of catalyst poisoning and require rigorous metal removal steps to meet stringent pharmaceutical purity standards. These legacy processes collectively result in elongated production timelines, inflated raw material expenditures, and complex operational protocols that are ill-suited for modern, lean manufacturing environments.
The Novel Approach
In stark contrast to these cumbersome legacy techniques, the novel approach disclosed in CN113527139A streamlines the synthetic pathway into a highly efficient sequence that prioritizes safety and cost-effectiveness without compromising yield. The core innovation involves a strategic halogenation-dehydrohalogenation strategy that bypasses the need for direct oxidative aromatization. Instead of forcing aromatization through harsh oxidation, the process first introduces a halogen atom at the 4-position of the dihydronaphthalene ring using mild halogenating agents like N-bromosuccinimide (NBS). This intermediate is then subjected to a simple base-mediated elimination reaction to achieve the desired aromatic system. This mechanistic shift allows the reaction to proceed under remarkably mild conditions, typically between 5°C and 40°C, eliminating the need for high-temperature reactors or high-pressure equipment. By utilizing readily available and inexpensive reagents such as cyanoacetic acid and common bases like sodium hydroxide, this method drastically simplifies the procurement landscape and reduces the overall cost of goods sold (COGS) for the final intermediate.
Mechanistic Insights into the Halogenation-Dehydrohalogenation Sequence
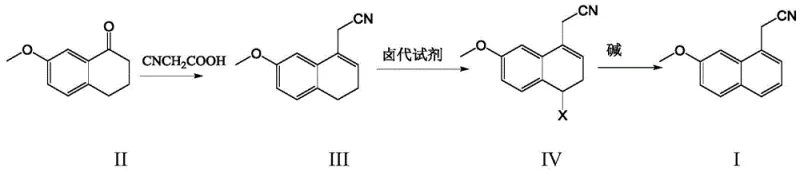
The chemical ingenuity of this process is best understood by examining the specific transformation of the tetralone derivative into the fully aromatic naphthylacetonitrile. The synthesis begins with a condensation reaction between 7-methoxytetrahydro-1-naphthalenone and cyanoacetic acid, catalyzed by a dual system of heptanoic acid and benzylamine. This step constructs the carbon framework necessary for the nitrile functionality while maintaining the partially saturated ring system. The pivotal moment in this synthesis occurs during the subsequent halogenation step, where the dihydro-intermediate reacts with an N-halosuccinimide to form a 4-halo-3,4-dihydro derivative. This specific regioselective halogenation activates the molecule for the final elimination step. Unlike direct dehydrogenation which requires strong oxidants to remove hydrogen atoms, this pathway leverages the leaving group ability of the halogen atom. When treated with a base such as sodium hydroxide in an alcoholic solvent, the molecule undergoes dehydrohalogenation, spontaneously ejecting the halogen and a proton to form the stable aromatic naphthalene ring system. This mechanism is thermodynamically favorable and kinetically accessible under ambient conditions, representing a significant departure from the high-energy barriers associated with traditional catalytic dehydrogenation.
From an impurity control perspective, this mechanistic route offers distinct advantages for ensuring high product quality. Traditional oxidative methods often generate complex mixtures of over-oxidized by-products or quinone adducts that are structurally similar to the target molecule and difficult to separate. In the halogenation-elimination pathway, the primary by-products are succinimide derivatives and inorganic salts, which are highly polar and water-soluble. This difference in physicochemical properties facilitates easy removal during the aqueous workup and crystallization stages described in the patent examples. For instance, the patent reports achieving HPLC purity greater than 99.9% simply through crystallization and water slurry washing, demonstrating the inherent cleanliness of the reaction. The avoidance of transition metal catalysts like palladium also eliminates the risk of heavy metal contamination, a critical parameter for regulatory compliance in pharmaceutical manufacturing. Consequently, this route not only simplifies the chemical transformation but also inherently builds quality into the process, reducing the burden on downstream purification units.
How to Synthesize 7-Methoxy-1-Naphthylacetonitrile Efficiently
Implementing this synthesis requires careful attention to reaction stoichiometry and temperature control to maximize the benefits of the novel pathway. The process is designed to be telescoped where possible, with the crude product from the initial condensation step being suitable for direct use in the subsequent halogenation without rigorous purification, thereby saving time and solvent. The halogenation step is particularly sensitive to molar ratios, with the patent suggesting a preferred ratio of substrate to halogenating agent between 1:1.2 and 1:1.5 to ensure complete conversion while minimizing excess reagent waste. The final elimination step is robust, tolerating various bases including sodium hydroxide, potassium hydroxide, or ammonia water, providing flexibility for process engineers to optimize based on available infrastructure. For a detailed breakdown of the specific operational parameters, solvent choices, and workup procedures required to replicate this high-yielding process, please refer to the standardized synthesis guide below.
- Condense 7-methoxytetrahydro-1-naphthalenone with cyanoacetic acid using heptanoic acid and benzylamine catalysts in toluene under reflux.
- Perform a halogenation reaction on the resulting nitrile intermediate using N-halosuccinimide (e.g., NBS) at mild temperatures (5-40°C).
- Execute dehydrohalogenation using a base such as sodium hydroxide in alcohol solvent to achieve aromatization and obtain the final product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route translates into tangible strategic advantages that extend beyond mere chemical yield. The primary driver of value is the substantial reduction in raw material costs achieved by eliminating the dependency on premium-priced reagents. By removing DDQ and palladium catalysts from the bill of materials, manufacturers can insulate their production costs from the volatility of the precious metals market and the supply constraints often associated with specialized oxidants. Furthermore, the use of commodity chemicals like N-bromosuccinimide and sodium hydroxide ensures a stable and diversified supply base, reducing the risk of production stoppages due to single-source supplier issues. The simplified operational profile also意味着 lower utility consumption, as the process does not require energy-intensive high-temperature heating or cryogenic cooling for pyrophoric reagents, contributing to a lower carbon footprint and reduced operational expenditure.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the substitution of high-cost inputs with low-cost alternatives. Traditional routes relying on DDQ incur significant expenses not only for the reagent itself but also for the disposal of the resulting toxic waste streams. By switching to a halogenation-elimination sequence, the process avoids these hidden costs entirely. Additionally, the high selectivity of the reaction minimizes the formation of difficult-to-remove impurities, which reduces the volume of solvents and adsorbents needed for purification. This efficiency gain directly improves the gross margin of the final intermediate, making it a more competitive offering in the global marketplace for agomelatine precursors.
- Enhanced Supply Chain Reliability: Supply chain resilience is significantly bolstered by the use of widely available starting materials. Reagents such as cyanoacetic acid, toluene, and N-halosuccinimides are produced by numerous chemical manufacturers globally, preventing bottlenecks that can occur with niche catalysts. The mild reaction conditions also reduce the wear and tear on reactor vessels and ancillary equipment, leading to higher asset utilization rates and fewer unplanned maintenance downtimes. This reliability ensures consistent delivery schedules to downstream API manufacturers, a critical factor in maintaining long-term contractual relationships in the pharmaceutical sector.
- Scalability and Environmental Compliance: Scaling this process from laboratory to commercial production is straightforward due to the absence of hazardous unit operations. The elimination of butyl lithium removes the need for specialized inert atmosphere handling systems and fire suppression infrastructure, lowering the capital expenditure required for plant retrofitting. Moreover, the environmental profile is markedly improved; the aqueous waste streams generated are easier to treat compared to the phenolic waste from DDQ oxidation. This alignment with green chemistry principles facilitates smoother regulatory approvals and supports corporate sustainability goals, which are increasingly important criteria for selection by major multinational pharmaceutical companies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation, offering clarity on the practical aspects of adopting this route. Understanding these details is essential for technical teams evaluating the feasibility of integrating this process into existing manufacturing lines or for procurement teams assessing the quality assurances associated with the new method.
Q: How does this new method improve upon traditional DDQ oxidation routes?
A: Traditional methods often rely on 2,3-dichloro-5,6-dicyano-1,4-benzoquinone (DDQ) for dehydrogenation, which is expensive and generates significant phenolic pollution. This novel patent utilizes a halogenation-dehydrohalogenation sequence that eliminates the need for DDQ, significantly reducing raw material costs and environmental waste treatment burdens.
Q: What are the safety advantages over butyl lithium-based synthesis?
A: Previous synthetic pathways described in literature utilized butyl lithium, a pyrophoric and highly dangerous reagent unsuitable for large-scale industrial production. The disclosed method operates under mild conditions (5-40°C) using stable reagents like N-bromosuccinimide and sodium hydroxide, drastically improving operational safety and scalability.
Q: What purity levels can be achieved with this halogenation route?
A: The patent data demonstrates exceptional purity control, with specific examples achieving HPLC purity greater than 99.9% after simple crystallization and washing steps. This high purity is critical for pharmaceutical intermediates intended for agomelatine synthesis, minimizing downstream purification requirements.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 7-Methoxy-1-Naphthylacetonitrile Supplier
As the demand for efficient antidepressant therapies grows, the need for a secure and high-quality supply of key intermediates like 7-methoxy-1-naphthylacetonitrile becomes paramount. NINGBO INNO PHARMCHEM stands at the forefront of this sector, leveraging advanced synthetic methodologies such as the one described in CN113527139A to deliver superior products. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the volumetric requirements of global supply chains without compromising on quality. We operate with stringent purity specifications and utilize rigorous QC labs to verify that every batch meets the exacting standards required for pharmaceutical synthesis, providing our partners with total confidence in material consistency.
We invite you to collaborate with us to optimize your supply chain for agomelatine production. Our technical experts are ready to provide a Customized Cost-Saving Analysis tailored to your specific volume needs, demonstrating how our optimized synthesis route can reduce your overall procurement costs. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us demonstrate how our commitment to innovation translates into reliable value for your organization.
