Advancing Pharmaceutical Intermediate Production with Efficient Copper-Catalyzed Dihydropyrazole Synthesis
Advancing Pharmaceutical Intermediate Production with Efficient Copper-Catalyzed Dihydropyrazole Synthesis
The landscape of pharmaceutical intermediate manufacturing is constantly evolving, driven by the need for more efficient, cost-effective, and environmentally benign synthetic routes. A significant breakthrough in this domain is detailed in patent CN109988114B, which discloses a robust preparation method for polysubstituted 4,5-dihydropyrazole compounds. These heterocyclic scaffolds are not merely academic curiosities; they serve as the core structural backbone for a vast array of biologically active natural products and functional molecules with applications spanning medicine, biology, and agrochemicals. As illustrated in the diverse biological profiles shown below, these compounds exhibit potent activities ranging from antibacterial and antimalarial properties to COX-2 inhibition and anti-inflammatory effects, underscoring their critical value in drug discovery pipelines.
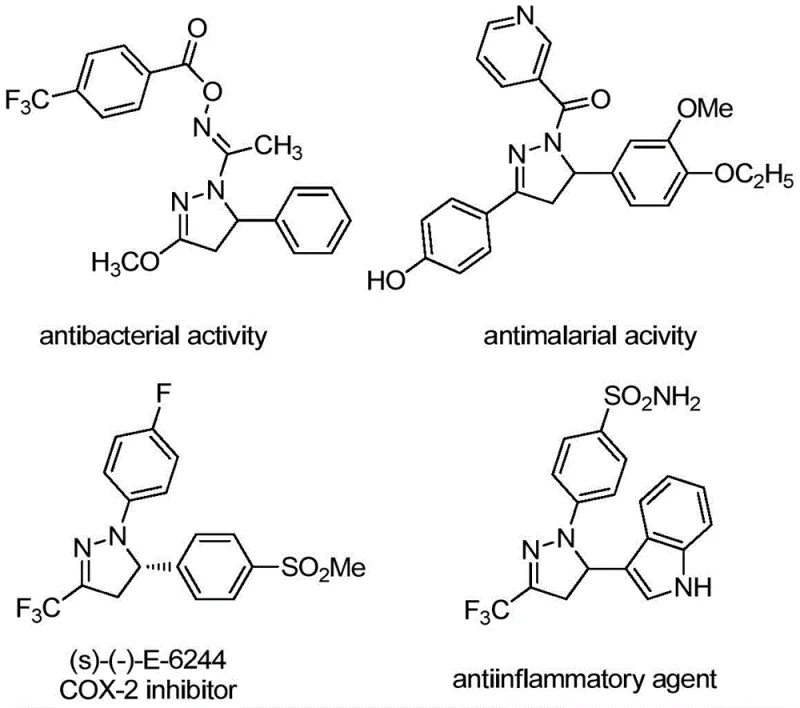
For R&D directors and process chemists, the ability to access these privileged structures efficiently is paramount. The disclosed method leverages a divalent copper salt and pivalic acid promoter system to facilitate the cyclization of sulfur ylides and benzenesulfonyl hydrazones. This approach represents a paradigm shift from traditional methodologies that often rely on precious metal catalysts or苛刻 reaction conditions. By utilizing readily available starting materials and operating under relatively mild thermal conditions without the strict requirement for anhydrous or oxygen-free environments, this technology offers a compelling solution for the reliable pharmaceutical intermediate supplier seeking to optimize their production capabilities and reduce lead time for high-purity intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 4,5-dihydropyrazoles has been fraught with challenges that hinder large-scale adoption and economic viability. Literature reports typically describe methods involving the reaction of α,β-unsaturated ketones with hydrazine hydrate, or base-catalyzed reactions of propargyl alcohols with hydrazines. More advanced techniques have employed gold-catalyzed one-pot syntheses using alkynes, aldehydes, or ketones. However, these conventional pathways suffer from significant drawbacks that impact both the bottom line and operational safety. For instance, many of these routes necessitate the pre-synthesis of complex reaction substrates, which inherently increases the number of synthetic steps and lowers the overall atom economy and reaction efficiency. Furthermore, the reliance on expensive alkyne precursors or precious metal catalysts like gold creates a substantial cost barrier, making cost reduction in API manufacturing difficult to achieve. Additionally, some protocols require stringent exclusion of moisture and oxygen, demanding specialized equipment and increasing the risk of batch failure due to environmental exposure.
The Novel Approach
In stark contrast to these legacy methods, the novel approach outlined in the patent data utilizes a direct and efficient coupling strategy that bypasses the need for pre-functionalized, expensive substrates. The core innovation lies in the use of sulfur ylides and benzenesulfonyl hydrazones as direct building blocks, catalyzed by an inexpensive divalent copper salt system. As depicted in the general reaction scheme below, this method streamlines the synthetic pathway into a single pot operation that is both operationally simple and economically superior. The elimination of precious metals and the tolerance for ambient atmospheric conditions mean that the reaction does not need to be carried out under anhydrous and oxygen-free conditions, which is a massive advantage for facility throughput. This simplicity allows for easier operation and post-processing, directly addressing the pain points of procurement managers who are tasked with sourcing cost-effective raw materials and minimizing waste generation in the production of complex pharmaceutical intermediates.
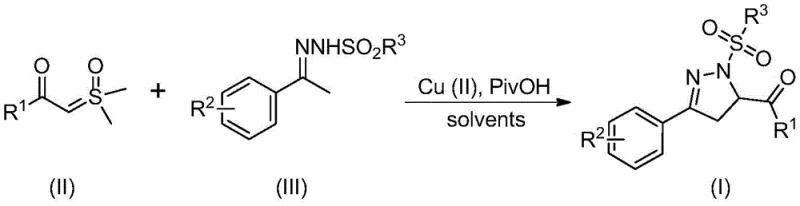
Mechanistic Insights into Cu(II)-Catalyzed Cyclization
Understanding the mechanistic underpinnings of this transformation is crucial for R&D teams aiming to replicate and optimize the process for specific target molecules. The proposed mechanism suggests a sophisticated interplay between the copper catalyst and the organic substrates. Initially, the divalent copper salt acts as a Lewis acid or oxidant to promote the dehydrogenation of the benzenesulfonyl hydrazone, generating a reactive 1,2-azadiene intermediate. Concurrently, the sulfur ylide undergoes decomposition, losing a molecule of dimethyl sulfoxide to form a highly reactive carbene intermediate. This carbene species then attacks the double bond of the azadiene intermediate in a Michael-type addition. Following this key carbon-carbon bond formation, the copper catalyst facilitates an intramolecular C-N bond formation, closing the five-membered ring to yield the final 4,5-dihydropyrazole structure. The presence of pivalic acid is believed to act as a crucial promoter, potentially stabilizing intermediates or accelerating the dehydrogenation step, thereby enhancing the overall reaction rate and yield.
From an impurity control perspective, this mechanism offers distinct advantages over radical-based or high-energy pathways. The concerted nature of the copper-catalyzed cycle tends to favor the formation of the desired regioisomer, minimizing the generation of side products that are difficult to separate. The use of specific substituents on the aryl rings, such as electron-donating methoxy groups or electron-withdrawing halogens, can be tuned to influence the electronic density of the intermediates, further refining the selectivity. For example, the patent data indicates that substrates with R1 groups like phenyl or 4-bromophenyl, and R2 groups like hydrogen or methyl, result in high yields, suggesting that the electronic environment is well-tolerated. This predictability is vital for ensuring high-purity pharmaceutical intermediates, as it reduces the burden on downstream purification processes like column chromatography, ultimately leading to a cleaner impurity profile and a more robust quality control framework for the final active ingredient.
How to Synthesize Polysubstituted 4,5-Dihydropyrazole Efficiently
Implementing this synthesis route in a laboratory or pilot plant setting requires adherence to specific parameters to maximize yield and reproducibility. The protocol involves combining the divalent copper salt, pivalic acid, sulfur ylide, and benzenesulfonyl hydrazone in a suitable organic solvent. The choice of solvent is critical; while various solvents can dissolve the reactants, aprotic solvents like toluene have been identified as optimal for promoting the reaction efficiency. The reaction mixture is then heated to a temperature range of 100~110℃ and maintained for a duration of 3 to 6 hours. This thermal energy is sufficient to drive the formation of the reactive intermediates and the subsequent cyclization without causing excessive decomposition. Upon completion, the workup procedure is straightforward, involving filtration to remove insoluble catalyst residues, followed by silica gel mixing and purification via column chromatography. The detailed standardized synthesis steps for this process are provided in the guide below.
- Combine divalent copper salt, pivalic acid, sulfur ylide, and benzenesulfonyl hydrazone in an organic solvent such as toluene.
- Heat the reaction mixture to 100-110°C and maintain stirring for 3 to 6 hours to ensure complete conversion.
- Perform post-processing including filtration and silica gel mixing, followed by column chromatography purification to isolate the target compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel synthetic methodology offers tangible strategic benefits that extend beyond mere chemical curiosity. The primary advantage lies in the drastic simplification of the supply chain for raw materials. Unlike traditional methods that might require custom-synthesized alkynes or sensitive organometallic reagents, this process utilizes sulfur ylides and benzenesulfonyl hydrazones, which are either commercially available or can be prepared from widely abundant precursors like aryl ketones and sulfonyl hydrazides. This accessibility ensures a stable supply of starting materials, reducing the risk of production delays caused by raw material shortages. Furthermore, the elimination of the need for specialized anhydrous or oxygen-free reaction conditions means that standard reactor vessels can be used, lowering capital expenditure requirements and simplifying facility maintenance. This operational flexibility translates directly into enhanced supply chain reliability and reduced lead time for high-purity intermediates.
- Cost Reduction in Manufacturing: The economic implications of this process are profound, primarily driven by the substitution of expensive catalysts and reagents with commodity chemicals. By replacing precious metal catalysts like gold with inexpensive divalent copper salts, the direct material cost per kilogram of product is significantly lowered. Additionally, the reaction's tolerance for ambient conditions eliminates the energy and equipment costs associated with maintaining inert atmospheres, such as nitrogen purging systems or gloveboxes. The high conversion rates observed in toluene further contribute to cost efficiency by maximizing the output from each batch, reducing the amount of solvent and energy required per unit of product. These factors combine to create a manufacturing process that is inherently leaner and more cost-competitive, allowing for substantial cost savings in the production of complex pharmaceutical intermediates without compromising on quality.
- Enhanced Supply Chain Reliability: In the volatile landscape of global chemical supply, reliability is as valuable as price. This synthesis route enhances reliability by relying on a robust chemistry that is less susceptible to minor fluctuations in environmental conditions. The fact that the reaction can be easily scaled up to the gram level and beyond, as noted in the patent, indicates a high degree of process robustness. This scalability ensures that as demand for the final API grows, the intermediate supply can be ramped up quickly without the need for extensive process re-engineering. Moreover, the use of common solvents like toluene, DMSO, or DCE ensures that solvent supply chains remain stable, avoiding bottlenecks associated with exotic or regulated solvents. This stability provides a secure foundation for long-term production planning and contract fulfillment.
- Scalability and Environmental Compliance: From an environmental and regulatory standpoint, this method aligns well with modern green chemistry principles. The avoidance of heavy metal contamination (other than trace copper which is easily removed) simplifies the waste treatment process and reduces the environmental footprint of the manufacturing site. The straightforward post-processing, which involves standard filtration and chromatography, minimizes the generation of hazardous waste streams compared to more complex multi-step syntheses. The ability to scale this reaction easily means that commercial scale-up of complex pharmaceutical intermediates can be achieved with minimal risk of encountering unforeseen engineering challenges. This ease of scale-up, combined with the use of less toxic reagents relative to alternatives, positions this technology as a sustainable choice for future-proofing chemical manufacturing operations against tightening environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this copper-catalyzed synthesis technology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing a clear picture of what potential partners and licensees can expect. Understanding these nuances is essential for making informed decisions about integrating this methodology into existing production workflows or R&D pipelines. The answers reflect the practical realities of running this chemistry at scale, focusing on yield, purity, and operational feasibility.
Q: What are the primary advantages of this copper-catalyzed method over traditional gold-catalyzed routes?
A: This method eliminates the need for expensive gold catalysts and avoids harsh anhydrous or oxygen-free conditions, significantly reducing operational complexity and raw material costs while maintaining high yields.
Q: Is this synthesis protocol scalable for industrial production?
A: Yes, the patent explicitly demonstrates that the reaction can be easily scaled up from gram levels to larger quantities without losing efficiency, making it highly suitable for commercial manufacturing.
Q: What types of substituents are tolerated in this reaction system?
A: The system exhibits excellent substrate scope, tolerating various groups including halogens, alkyl, alkoxy, and aryl groups on both the sulfur ylide and the benzenesulfonyl hydrazone components.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Polysubstituted 4,5-Dihydropyrazole Supplier
As the global demand for bioactive heterocycles continues to surge, having a partner with deep technical expertise and proven manufacturing capabilities is essential. NINGBO INNO PHARMCHEM stands at the forefront of this industry, leveraging advanced catalytic technologies like the copper-mediated cyclization described herein to deliver superior results. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from the bench to the plant. We understand that consistency is key in the pharmaceutical sector; therefore, our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications to guarantee that every batch of polysubstituted 4,5-dihydropyrazole meets the highest international standards. Our commitment to quality assurance means that you can rely on us for a steady supply of critical intermediates that drive your drug development forward.
We invite you to explore how our optimized synthesis routes can transform your supply chain dynamics. By partnering with us, you gain access not just to a product, but to a comprehensive service model designed to enhance your competitive edge. We encourage you to contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. Whether you need specific COA data for regulatory filings or detailed route feasibility assessments for new analogues, our experts are ready to provide the support you need. Let us help you navigate the complexities of fine chemical manufacturing with confidence, ensuring that your projects remain on schedule and within budget while achieving the highest levels of chemical excellence.
