Advanced Manufacturing of o-Aminophenylpropanol: A Cost-Effective Route for Global Pharmaceutical Supply Chains
The global demand for high-quality heterocyclic intermediates continues to surge, driven by the expanding pharmaceutical and agrochemical sectors. Within this landscape, o-aminophenylpropanol stands out as a critical building block for synthesizing biologically active compounds, including potential drug candidates and functional materials. However, traditional manufacturing pathways have long been hindered by prohibitive costs and complex safety requirements. A breakthrough detailed in patent CN100357256C introduces a robust, five-step synthetic methodology that fundamentally reshapes the production economics of this valuable molecule. By shifting the starting material from expensive specialty alcohols to commodity-grade o-nitrotoluene, this innovation addresses the core pain points of supply chain volatility and equipment intensity. For R&D directors and procurement leaders, understanding this technological pivot is essential for securing a competitive edge in the sourcing of complex organic intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of o-aminophenylpropanol has relied heavily on direct catalytic hydrogenation strategies. As illustrated in the reaction scheme below, the conventional approach typically involves the reduction of o-nitrocinnamyl alcohol using hydrogen gas over a Raney-Nickel catalyst. While conceptually straightforward as a single-step transformation, this method presents severe logistical and economic barriers for commercial scale-up. The primary bottleneck is the raw material itself; o-nitrocinnamyl alcohol is a high-value, low-volume specialty chemical that commands a premium price in the global market, drastically inflating the cost of goods sold (COGS). Furthermore, the reliance on high-pressure hydrogenation necessitates the use of specialized autoclaves and rigorous safety protocols to manage explosive gas mixtures. This requirement not only increases capital expenditure (CAPEX) for manufacturing facilities but also introduces significant operational risks and maintenance overheads. Consequently, this legacy route limits supply flexibility and makes the final product vulnerable to upstream price fluctuations in the niche cinnamyl alcohol market.
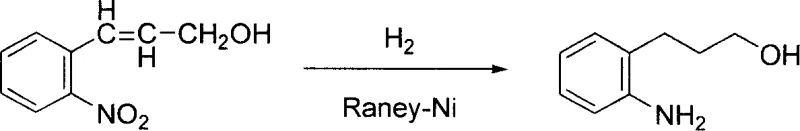
The Novel Approach
In stark contrast, the novel methodology disclosed in the patent data circumvents these limitations through a cleverly designed multi-step sequence starting from o-nitrotoluene. This starting material is a ubiquitous commodity chemical, widely produced for the dye and pesticide industries, ensuring a stable and low-cost supply base. The new route replaces the dangerous high-pressure hydrogenation with a series of controlled liquid-phase reactions, including radical bromination, nucleophilic substitution, and selective reductions. By breaking the synthesis down into five distinct, manageable steps, the process eliminates the need for specialized high-pressure equipment, allowing production to occur in standard glass-lined or stainless steel reactors found in most fine chemical plants. This transition from a high-risk, high-cost single step to a modular, ambient-pressure workflow represents a paradigm shift in process chemistry. It empowers manufacturers to achieve a total yield ranging from 56% to 75% while maintaining exceptional control over reaction parameters, ultimately delivering a more reliable and cost-efficient supply of the target intermediate.
Mechanistic Insights into the Five-Step Synthetic Cascade
The core of this technological advancement lies in the precise orchestration of five chemical transformations that build the carbon skeleton and install the necessary functional groups with high fidelity. The journey begins with a radical bromination of o-nitrotoluene using N-bromosuccinimide (NBS) and benzoyl peroxide (BPO) in carbon tetrachloride. This step selectively functionalizes the benzylic position to form o-nitrobenzyl bromide, a highly reactive electrophile crucial for chain extension. Following isolation, the intermediate undergoes a nucleophilic substitution with dimethyl malonate in the presence of potassium tert-butoxide. This carbon-carbon bond-forming reaction extends the side chain, installing the three-carbon backbone required for the final propanol structure. The resulting diester is then subjected to acidic hydrolysis and thermal decarboxylation. This critical step removes the ester masking groups and one carboxyl unit, yielding o-nitrophenylpropionic acid. The ability to isolate this acid intermediate allows for rigorous purification via acid-base extraction, effectively scrubbing away non-acidic impurities before proceeding to the reduction phases.
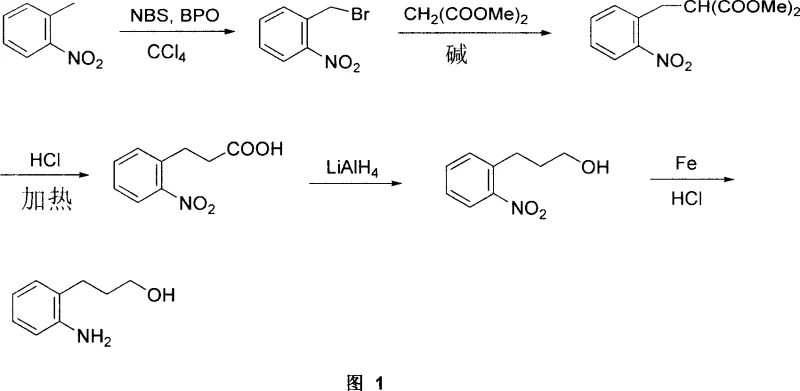
The final phase of the synthesis involves two sequential reduction events that define the product's functionality. First, the carboxylic acid moiety of o-nitrophenylpropionic acid is reduced to a primary alcohol using lithium aluminum hydride (LiAlH4) in tetrahydrofuran. This powerful reducing agent ensures complete conversion of the acid to the corresponding alcohol without affecting the aromatic nitro group under controlled conditions. The subsequent step targets the nitro group, utilizing a classic Bechamp-style reduction with iron powder and hydrochloric acid in an ethanol solvent system. This heterogeneous reduction is highly selective for the nitro-to-amine transformation, avoiding over-reduction of the aromatic ring. From an impurity control perspective, the process leverages the amphiprotic nature of the intermediates. For instance, during the isolation of o-nitrophenylpropionic acid, the mixture is basified to form a water-soluble carboxylate salt, allowing organic-soluble impurities to be washed away. Re-acidification then precipitates or extracts the pure acid. Similarly, the final amine product can be purified by exploiting its solubility in acidic aqueous phases, ensuring that the final high-purity o-aminophenylpropanol meets the stringent specifications demanded by pharmaceutical customers.
How to Synthesize o-Aminophenylpropanol Efficiently
Implementing this synthesis route requires careful attention to stoichiometry and temperature control across the five stages to maximize the cumulative yield. The process is designed to be operationally simple, relying on standard workup procedures such as filtration, extraction, and rotary evaporation rather than complex chromatographic separations, which are impractical at scale. The initial bromination sets the tone for the entire sequence, requiring precise molar ratios of NBS to avoid poly-bromination. Subsequent steps utilize common laboratory and industrial solvents like THF and ethyl acetate, facilitating solvent recovery and recycling. For a comprehensive breakdown of the specific reaction conditions, reagent quantities, and isolation protocols validated in the patent examples, please refer to the standardized technical guide below.
- Perform radical bromination of o-nitrotoluene using NBS and BPO in carbon tetrachloride to generate o-nitrobenzyl bromide.
- Execute nucleophilic substitution with dimethyl malonate and potassium tert-butoxide in THF to extend the carbon chain.
- Conduct acid-catalyzed hydrolysis and decarboxylation to form o-nitrophenylpropionic acid, followed by purification via acid-base extraction.
- Reduce the carboxylic acid group to an alcohol using lithium aluminum hydride (LiAlH4) under inert atmosphere.
- Complete the synthesis by reducing the nitro group to an amine using iron powder and hydrochloric acid in ethanol.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the shift to this five-step synthesis offers tangible strategic benefits that extend beyond simple chemistry. The most immediate impact is seen in the drastic reduction of raw material costs. By anchoring the synthesis on o-nitrotoluene, a bulk chemical with a mature and competitive global market, manufacturers insulate themselves from the price volatility associated with specialty starting materials like o-nitrocinnamyl alcohol. This stability translates directly into more predictable pricing models for long-term supply agreements. Furthermore, the elimination of high-pressure hydrogenation equipment removes a significant barrier to entry for contract manufacturing organizations (CMOs). Facilities that lack high-pressure autoclaves can now bid on production contracts for this intermediate, increasing the pool of qualified suppliers and enhancing supply chain resilience against regional disruptions or capacity bottlenecks.
- Cost Reduction in Manufacturing: The economic advantage of this route is driven by the substitution of expensive feedstock with commodity chemicals. While the process involves five steps compared to the traditional one-step method, the aggregate cost of reagents (NBS, dimethyl malonate, LiAlH4, iron powder) is significantly lower than the premium charged for o-nitrocinnamyl alcohol. Additionally, the avoidance of high-pressure infrastructure reduces both capital depreciation costs and insurance premiums associated with hazardous operations. The simplified purification strategy, which relies on pH-dependent extraction rather than expensive column chromatography, further lowers operational expenditures (OPEX) by minimizing solvent consumption and waste disposal costs, resulting in substantial overall cost savings for the final product.
- Enhanced Supply Chain Reliability: Supply continuity is a critical metric for downstream drug manufacturers. This synthetic route enhances reliability by diversifying the raw material base. O-nitrotoluene is produced by multiple major chemical companies globally, ensuring that a shortage from a single vendor does not halt production. Moreover, the operational simplicity of the steps—reflux, stirring, and filtration—reduces the likelihood of batch failures due to equipment malfunction or operator error. The robustness of the chemistry allows for flexible scheduling and faster turnaround times between batches, enabling suppliers to respond more agilely to fluctuating market demands and urgent procurement requests without compromising on quality or delivery timelines.
- Scalability and Environmental Compliance: Scaling chemical processes often introduces unforeseen challenges, but this route is inherently scalable due to its reliance on standard unit operations. The exothermic nature of the reactions is manageable with conventional cooling systems, and the use of iron powder for nitro reduction generates iron oxide sludge that is easier to handle and dispose of compared to heavy metal catalysts like Raney Nickel. This aligns well with modern environmental, health, and safety (EHS) standards, reducing the regulatory burden on manufacturing sites. The ability to run these reactions in standard vessels facilitates a smoother transition from pilot plant to commercial tonnage production, ensuring that commercial scale-up of complex pharmaceutical intermediates can be achieved rapidly to meet growing market needs.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of o-aminophenylpropanol. These insights are derived directly from the patented methodology and are intended to clarify the feasibility and advantages of this specific synthetic route for potential partners and technical evaluators.
Q: Why is the new 5-step synthesis preferred over the traditional hydrogenation method?
A: The traditional method relies on expensive o-nitrocinnamyl alcohol and requires high-pressure hydrogenation equipment, posing safety and cost risks. The new 5-step route utilizes cheap, readily available o-nitrotoluene and operates under standard atmospheric pressure, significantly lowering capital expenditure and operational complexity.
Q: How does this process ensure high purity for pharmaceutical applications?
A: The process incorporates multiple purification stages, specifically leveraging acid-base extraction properties of intermediates like o-nitrophenylpropionic acid. By converting impurities into water-soluble salts or removing them via organic extraction at specific pH levels, the final product achieves stringent purity specifications required for API synthesis.
Q: Is this synthesis route scalable for industrial production?
A: Yes, the route is highly scalable. It avoids specialized high-pressure reactors and uses common solvents like THF, ethanol, and ethyl acetate. The total yield ranges from 56% to 75%, which is economically viable for large-scale manufacturing of fine chemical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable o-Aminophenylpropanol Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of new therapeutic agents depends on the availability of high-quality, cost-effective intermediates. Our technical team has thoroughly analyzed the synthetic pathway described in patent CN100357256C and possesses the expertise to implement this efficient five-step route at an industrial scale. We understand the critical importance of maintaining stringent purity specifications throughout the manufacturing process to prevent downstream contamination in API synthesis. Our state-of-the-art facilities are equipped with rigorous QC labs capable of monitoring every stage of the reaction, from the initial bromination to the final amine isolation. With extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, we are uniquely positioned to serve as your strategic partner for this key building block.
We invite you to collaborate with us to optimize your supply chain for o-aminophenylpropanol. Our engineering team is ready to conduct a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this alternative synthesis can reduce your overall procurement spend. We encourage you to contact our technical procurement team today to request specific COA data from our pilot batches and to discuss route feasibility assessments for your upcoming projects. Let us help you secure a sustainable and economical supply of this vital intermediate.
