Advanced Ionic Liquid Catalysis for Scalable Polysubstituted Benzene Production
Advanced Ionic Liquid Catalysis for Scalable Polysubstituted Benzene Production
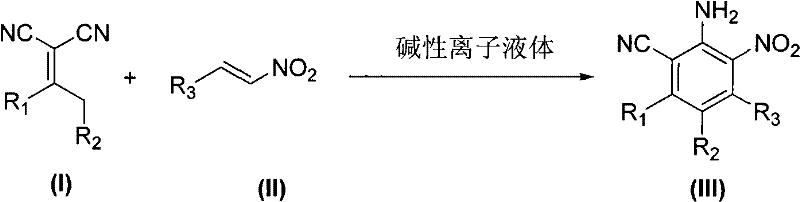
The synthesis of polysubstituted benzene compounds represents a critical challenge in modern organic chemistry, particularly for the development of high-value pharmaceutical intermediates and agrochemical active ingredients. Traditional routes often suffer from harsh conditions and poor atom economy, necessitating innovative catalytic systems. Patent CN101445470B introduces a groundbreaking methodology utilizing basic ionic liquids to facilitate the cyclization of substituted malononitriles and nitroethylenes. This technology offers a robust pathway for generating complex aromatic scaffolds under mild conditions, typically between 20~100℃, with reaction times drastically reduced to 1~10h. For R&D directors and procurement specialists seeking reliable pharmaceutical intermediate supplier partnerships, this patent data underscores a shift towards greener, more efficient manufacturing protocols that align with modern sustainability goals while maintaining high chemical fidelity.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of polysubstituted benzene rings has relied heavily on classical substitution reactions such as Friedel-Crafts alkylation or acylation, which frequently require stoichiometric amounts of Lewis acids and generate significant hazardous waste. Furthermore, prior art methods involving the condensation of malononitriles and nitroethylenes, such as those reported by Dong Xue in J. Org. Chem. 2007, utilized triethylamine followed by stoichiometric sodium ethoxide in acetonitrile. These legacy processes are plagued by excessively long reaction durations ranging from 12 to 80 hours and necessitate the use of equivalent or excess amounts of strong bases that cannot be recovered. The inability to recycle reagents in these traditional workflows results in elevated operational expenditures and complicates waste management, posing significant hurdles for cost reduction in API manufacturing at an industrial scale.
The Novel Approach
In stark contrast, the novel approach detailed in the patent leverages basic ionic liquids, specifically [Bmim]OH or [Hmim]OH, to drive the aromatization process with exceptional efficiency. This method allows for the reaction to proceed either under solvent-free conditions or in common organic solvents like ethanol, achieving completion in merely 2~6 hours under preferred conditions of 50~80℃. The versatility of this system is evident in its tolerance for a wide array of functional groups, including halogens, alkoxy groups, and heterocycles, without compromising yield. By eliminating the need for stoichiometric inorganic bases and enabling catalyst recovery, this technology provides a streamlined route for the commercial scale-up of complex aromatic scaffolds, directly addressing the pain points of throughput and environmental compliance faced by modern chemical enterprises.
Mechanistic Insights into Ionic Liquid Catalyzed Aromatization
The core of this synthetic breakthrough lies in the dual functionality of the basic ionic liquid, which acts simultaneously as a Brønsted base catalyst and a polar reaction medium. The mechanism likely initiates with the deprotonation of the active methylene group in the substituted malononitrile by the hydroxide anion of the ionic liquid, generating a nucleophilic carbanion. This species undergoes a Michael addition to the beta-carbon of the nitroethylene derivative, forming a key intermediate that subsequently cyclizes. The unique solvation properties of the ionic liquid stabilize the transition states and charged intermediates, facilitating the elimination of water or other small molecules to achieve aromatization. This stabilization effect is crucial for maintaining high regioselectivity, ensuring that the desired polysubstituted benzene isomer is formed predominantly over potential side products, which is vital for downstream purification efforts in high-purity OLED material or drug substance production.
Furthermore, the impurity profile of the reaction is significantly improved due to the mild nature of the catalytic environment. Unlike strong inorganic bases that can promote hydrolysis of sensitive nitrile groups or decomposition of labile substituents, the tunable basicity of the ionic liquid minimizes degradation pathways. The patent data indicates that the ionic liquid layer can be separated from the organic product layer simply by extraction with ethyl acetate. After washing and vacuum drying, the catalyst retains its activity for multiple cycles, with only minor make-up required to compensate for mechanical losses. This recyclability not only reduces the consumption of expensive catalytic materials but also ensures a consistent impurity profile across batches, a critical parameter for regulatory approval in the pharmaceutical sector where batch-to-batch consistency is paramount.
How to Synthesize Polysubstituted Benzene Efficiently
Implementing this synthesis route requires careful attention to the molar ratios and separation techniques to maximize the benefits of the ionic liquid system. The standard protocol recommends a 1:1 molar ratio of the malononitrile and nitroethylene precursors, with the ionic liquid loading optimized between 5~20mol% relative to the substrate. For laboratory-scale optimization or pilot runs, the reaction can be conducted in ethanol or acetonitrile to improve mixing, though solvent-free conditions are preferred for larger batches to enhance green chemistry metrics. Following the reaction, the workup involves a simple liquid-liquid extraction where the product partitions into the organic phase while the catalyst remains in the ionic phase. Detailed standardized synthesis steps see the guide below.
- Mix substituted malononitrile and nitroethylene derivatives with basic ionic liquid catalyst (e.g., [Bmim]OH) at a molar ratio of 1: 1.
- Heat the reaction mixture to 50-80°C under solvent-free conditions or in ethanol/acetonitrile for 2-6 hours.
- Extract the product with ethyl acetate, recover the ionic liquid layer for reuse, and purify the organic layer via silica gel chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a supply chain perspective, the adoption of this ionic liquid catalyzed process offers transformative advantages in terms of operational stability and resource efficiency. The primary benefit stems from the elimination of stoichiometric inorganic bases and the ability to recycle the catalyst, which fundamentally alters the cost structure of the synthesis. By removing the need for massive quantities of sodium ethoxide and the associated neutralization waste streams, manufacturers can achieve substantial cost savings in raw material procurement and waste disposal fees. Additionally, the shorter reaction times directly translate to increased reactor turnover rates, allowing facilities to produce more batches within the same timeframe without requiring capital investment in new equipment, thereby enhancing overall asset utilization.
- Cost Reduction in Manufacturing: The economic impact of this technology is driven by the drastic reduction in reagent consumption and waste treatment costs. Since the basic ionic liquid can be reused multiple times without significant loss of catalytic activity, the effective cost per kilogram of catalyst is negligible compared to single-use bases. Furthermore, the option to run the reaction under solvent-free conditions eliminates the purchase, storage, and recovery of large volumes of organic solvents, leading to significant reductions in utility costs for distillation and solvent management. This lean manufacturing approach ensures that the final product remains cost-competitive even when scaling to multi-ton quantities.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions contributes to a more resilient supply chain. The tolerance for various substituents means that the same process platform can be adapted for a wide range of derivatives without extensive re-validation, reducing the lead time for new product introductions. Moreover, the mild temperature range of 20~100℃ reduces the risk of thermal runaway incidents, ensuring safer operations and minimizing the likelihood of production stoppages due to safety concerns. This reliability is essential for maintaining continuous supply to downstream customers who depend on just-in-time delivery models for their own production schedules.
- Scalability and Environmental Compliance: Scaling this process from gram to ton scale is facilitated by the simplicity of the workup procedure, which avoids complex filtration of inorganic salts often encountered with traditional bases. The reduction in hazardous waste generation aligns with increasingly stringent global environmental regulations, mitigating the risk of compliance penalties. The ability to operate under solvent-free conditions further enhances the environmental profile, making the process attractive for companies aiming to reduce their carbon footprint. This alignment with green chemistry principles not only future-proofs the manufacturing process but also appeals to end-users who prioritize sustainable sourcing in their vendor selection criteria.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this ionic liquid catalyzed synthesis. These insights are derived directly from the experimental data and claims within the patent documentation, providing a factual basis for evaluating the technology's fit within your existing manufacturing portfolio. Understanding these nuances is critical for technical teams assessing feasibility and procurement teams negotiating supply agreements.
Q: What are the advantages of using basic ionic liquids over traditional bases like sodium ethoxide?
A: Basic ionic liquids serve as both catalyst and solvent, allowing for milder reaction conditions (20-100°C vs harsh reflux), significantly shorter reaction times (1-10h vs 12-80h), and crucially, the ability to recycle the catalyst multiple times without loss of activity, reducing waste and cost.
Q: Can this synthesis method be performed without organic solvents?
A: Yes, the patent explicitly describes solvent-free conditions where the ionic liquid acts as the reaction medium. This eliminates the need for volatile organic compounds (VOCs), simplifying downstream processing and enhancing environmental compliance.
Q: What is the substrate scope for this aromatization reaction?
A: The method demonstrates broad compatibility with various substituents including alkyl, cycloalkyl, phenyl, halogenated phenyl, and heteroaryl groups on both the malononitrile and nitroethylene starting materials, yielding diverse polysubstituted benzene scaffolds.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Polysubstituted Benzene Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of advanced catalytic technologies like the one described in CN101445470B for producing high-value chemical intermediates. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless. We are committed to delivering products with stringent purity specifications, supported by our rigorous QC labs that employ state-of-the-art analytical instrumentation to verify identity and assay. Whether you require custom synthesis of complex aromatic scaffolds or reliable supply of established intermediates, our infrastructure is designed to meet the demanding requirements of the global pharmaceutical and agrochemical industries.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis route can be tailored to your specific project needs. By leveraging our expertise in process optimization, we can provide a Customized Cost-Saving Analysis that quantifies the potential efficiencies for your specific target molecule. Please contact us to request specific COA data for related compounds or to initiate a dialogue regarding route feasibility assessments. Let us partner with you to accelerate your development timelines and secure a competitive advantage in the marketplace through superior chemical manufacturing solutions.
