Scalable A3B-Type Porphyrin Synthesis and Metalloporphyrin Catalysis for Industrial Oxidation
The landscape of fine chemical synthesis is constantly evolving, driven by the need for more efficient, sustainable, and cost-effective pathways to complex molecular architectures. A pivotal development in this domain is documented in patent CN103214492A, which introduces a novel preparation method for A3B-type porphyrins and their corresponding metalloporphyrin derivatives. These compounds are not merely academic curiosities; they represent a class of high-value functional materials with profound implications for biomimetic catalysis and pharmaceutical intermediate production. The core innovation lies in a modified mixed-aldehyde condensation strategy that circumvents the tedious isolation of dipyrromethane intermediates, a bottleneck that has historically plagued the scalable production of asymmetric porphyrins. By optimizing the reaction sequence and solvent systems, this technology offers a robust route to generating low-symmetry porphyrin macrocycles substituted with electron-withdrawing groups like chlorine and nitro functionalities.
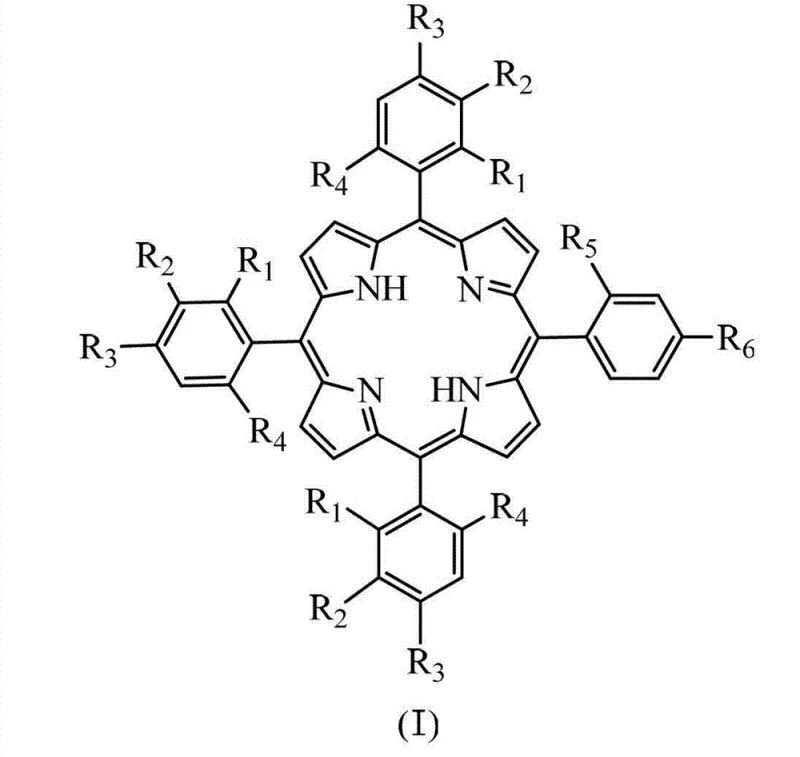
For R&D directors and process chemists, the structural versatility offered by this patent is particularly compelling. The ability to precisely install three identical aryl groups (A-rings) and one distinct aryl group (B-ring) allows for fine-tuning of the electronic properties of the porphyrin core. This is critical for applications in catalytic oxidation, where the redox potential of the central metal ion must be carefully balanced against the stability of the macrocycle. The patent specifically highlights the utility of chloro- and nitro-substituted phenyl rings, which enhance the catalytic activity of the resulting metalloporphyrins by lowering the energy gap between the highest occupied and lowest unoccupied molecular orbitals. This electronic modulation is essential for activating molecular oxygen under mild conditions, a key requirement for green chemistry initiatives in the fine chemical sector.
Understanding the limitations of conventional synthesis methods is crucial for appreciating the technological leap provided by this patent. Historically, the synthesis of A3B-type porphyrins has relied heavily on two primary strategies: the statistical mixed-aldehyde method and the stepwise dipyrromethane (2+2) approach. The mixed-aldehyde method, while operationally simple, suffers from a fundamental lack of selectivity. When two different aldehydes react with pyrrole, they generate a statistical mixture of four different porphyrin species (A4, A3B, A2B2, AB3), necessitating difficult and yield-limiting chromatographic separations to isolate the desired A3B product. This randomness leads to significant waste of expensive starting materials and solvents, creating a substantial burden on both cost and environmental compliance.
Conversely, the dipyrromethane method offers better control over substitution patterns but introduces severe operational complexities. Traditional 2+2 syntheses require the separate preparation, isolation, and purification of dipyrromethane intermediates before they can be condensed with aldehydes to form the macrocycle. These intermediates are often acid-sensitive and prone to scrambling or decomposition upon standing, requiring stringent handling conditions and immediate use. Furthermore, the multi-step nature of this approach inherently accumulates yield losses at each isolation stage. The patent CN103214492A addresses these pain points by introducing a hybrid approach that retains the simplicity of the mixed-aldehyde method while mitigating its selectivity issues through a controlled addition sequence. By pre-reacting the B-aldehyde with excess pyrrole and then introducing this mixture to the A-aldehyde solution, the method biases the condensation towards the desired A3B architecture without requiring the physical isolation of the dipyrromethane species.
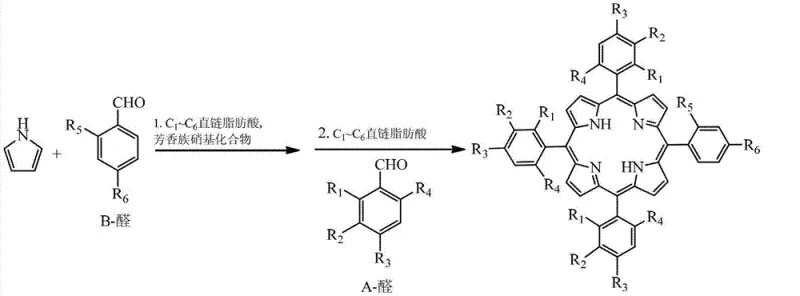
The mechanistic insights into this novel synthetic route reveal a sophisticated manipulation of reaction kinetics and thermodynamics. The process begins with the acid-catalyzed condensation of the B-aldehyde with pyrrole in a solvent system comprising straight-chain fatty acids and aromatic nitro compounds. This initial step generates a reactive pool of oligomers, including dipyrromethanes and tripyrromethanes, in situ. Crucially, instead of quenching and isolating these intermediates, the entire reaction mixture is transferred directly into a refluxing solution containing the A-aldehyde. This "one-pot" transfer minimizes exposure of the acid-labile intermediates to harsh workup conditions, thereby preserving their integrity for the subsequent macrocyclization step. The presence of electron-withdrawing nitro compounds in the solvent matrix may also play a role in stabilizing the cationic intermediates formed during the condensation, although the primary driver is the kinetic control afforded by the sequential addition.
Following the formation of the free-base porphyrin, the patent details a robust metallation protocol to generate the active catalyst species. The insertion of metal ions such as Iron (Fe), Manganese (Mn), Cobalt (Co), Copper (Cu), Zinc (Zn), or Nickel (Ni) is achieved by refluxing the porphyrin ligand with the corresponding metal salt in a mixture of N,N-dimethylformamide (DMF) and glacial acetic acid. For specific high-valent metal centers like Iron and Manganese, the addition of concentrated hydrochloric acid post-reaction ensures the formation of the chloro-metalloporphyrin species, which are often the active forms in oxidation catalysis. The electron-withdrawing nature of the peripheral chloro and nitro substituents significantly influences the coordination chemistry of the central metal. As illustrated in the structural diagrams, these groups pull electron density away from the porphyrin ring, making the central metal more electrophilic and thus more capable of activating oxidants like molecular oxygen or peroxides.
For procurement managers and supply chain heads, the commercial advantages of this synthesis method are rooted in its operational efficiency and resource optimization. By eliminating the isolation of dipyrromethane intermediates, the process drastically reduces the volume of solvents required for extraction, washing, and purification. In large-scale manufacturing, solvent purchase, recovery, and disposal constitute a major portion of operating expenses. Reducing the number of unit operations directly translates to lower energy consumption for heating, cooling, and distillation. Furthermore, the method improves atom economy by utilizing by-products like tripyrromethanes that would otherwise be discarded in traditional dipyrromethane syntheses. This comprehensive utilization of reaction mass means that less raw material is needed to produce the same amount of final product, providing a buffer against volatility in the pricing of specialty aldehydes and pyrrole.
- Cost Reduction in Manufacturing: The streamlined workflow eliminates multiple isolation and purification steps associated with intermediate dipyrromethanes. This reduction in unit operations leads to substantial savings in labor, solvent consumption, and energy usage. By avoiding the need for extensive chromatographic separation of statistical mixtures typical in older mixed-aldehyde methods, the process enhances overall throughput and reduces the cost of goods sold for high-purity A3B porphyrins.
- Enhanced Supply Chain Reliability: The starting materials required for this synthesis, such as various chlorobenzaldehydes, nitrobenzaldehydes, and pyrrole, are commodity chemicals with stable global supply chains. The robustness of the reaction conditions, which tolerate a range of temperatures and solvent ratios, ensures consistent production quality even with minor variations in raw material grades. This reliability minimizes the risk of production delays and ensures a steady supply of critical catalytic intermediates for downstream pharmaceutical and agrochemical applications.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing standard reflux and stirring equipment common in fine chemical plants. The significant reduction in solvent waste and the avoidance of hazardous intermediate handling align with modern environmental, health, and safety (EHS) standards. This compliance facilitates easier regulatory approval for new manufacturing sites and reduces the long-term liability associated with hazardous waste disposal, making it a sustainable choice for long-term production partnerships.
The technical depth of this patent extends beyond mere synthesis; it provides a framework for understanding how molecular design influences catalytic performance. The FAQ section below addresses common queries regarding the stability, purity, and application scope of these materials, drawing directly from the experimental data and claims presented in the documentation. For organizations looking to integrate biomimetic oxidation catalysts into their processes, understanding these nuances is vital for successful technology transfer and process validation.
- Pre-mix B-aldehyde and pyrrole in a solvent system containing fatty acids and aromatic nitro compounds at 0-50°C for 5-60 minutes.
- Add the reaction mixture directly to a refluxing solution containing A-aldehyde and continue refluxing for 0.5-2 hours to form the porphyrin macrocycle.
- For metalloporphyrins, react the purified porphyrin ligand with metal salts (Fe, Mn, Co, Zn, etc.) in DMF/acetic acid under reflux for 1-5 hours.
Frequently Asked Questions (FAQ)
Q: What is the primary advantage of this A3B porphyrin synthesis method?
A: The method eliminates the need to isolate and purify unstable dipyrromethane intermediates, significantly simplifying the workflow and reducing solvent consumption compared to traditional 2+2 synthesis routes.
Q: What applications do these chloro-nitro substituted metalloporphyrins serve?
A: They function as highly effective biomimetic catalysts for the selective oxidation of methyl arenes, such as converting o-cresol to salicylic acid using molecular oxygen.
Q: Is this process suitable for large-scale manufacturing?
A: Yes, the process utilizes standard reflux conditions and commercially available aldehydes, making it adaptable for commercial scale-up of complex fine chemicals without requiring specialized high-pressure equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable A3B-Type Porphyrin Supplier
As the demand for specialized catalysts and high-performance intermediates grows, partnering with an experienced CDMO becomes a strategic imperative. NINGBO INNO PHARMCHEM stands at the forefront of fine chemical manufacturing, possessing extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our technical team is well-versed in the intricacies of porphyrin chemistry, ensuring that the transition from laboratory bench to pilot plant and finally to full commercial scale is seamless. We maintain stringent purity specifications and operate rigorous QC labs equipped with advanced analytical instrumentation to verify the structural integrity and metal content of every batch, guaranteeing that our clients receive materials that meet the highest industry standards.
We invite you to collaborate with us to leverage this innovative synthesis technology for your specific applications. Whether you require custom metalloporphyrins for oxidation catalysis or specialized porphyrin ligands for material science, our team is ready to provide a Customized Cost-Saving Analysis tailored to your project needs. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, ensuring that your supply chain is built on a foundation of scientific excellence and operational reliability.
