Revolutionizing Cycloheptatriene Derivative Production: A Rapid, Metal-Free Route for High-Value Pharmaceutical Intermediates
Revolutionizing Cycloheptatriene Derivative Production: A Rapid, Metal-Free Route for High-Value Pharmaceutical Intermediates
In the rapidly evolving landscape of pharmaceutical intermediate manufacturing, the efficiency and safety of synthetic routes are paramount determinants of commercial viability. Patent CN109369672B introduces a groundbreaking methodology for the preparation of polysubstituted cycloheptatriene derivatives, a structural motif frequently found in bioactive molecules with potent anti-tumor, antibacterial, and antiviral properties. This innovation represents a significant paradigm shift from traditional transition-metal catalyzed processes to a hypervalent iodine-mediated oxidation strategy. By leveraging the unique reactivity of iodobenzene diacetate (PIDA) in trifluoroethanol (TFE), this protocol achieves rapid cyclization at room temperature within merely one minute. For R&D directors and procurement specialists alike, this development signals a new era of cost-effective and environmentally benign synthesis for complex heterocyclic scaffolds essential in modern drug discovery.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the cycloheptatriene core has relied heavily on transition metal catalysis, often involving rhodium or ruthenium complexes to facilitate carbene generation and subsequent ring expansion reactions. As illustrated in the prior art reaction scheme (Formula I), these conventional pathways typically require the use of hazardous diazo compounds as precursors, which pose significant safety risks regarding explosivity and toxicity during handling and storage. Furthermore, the reliance on noble metal catalysts not only inflates the raw material costs but also introduces stringent purification challenges to meet residual metal specifications required for pharmaceutical applications. The reaction times associated with these metal-catalyzed processes often exceed one hour, limiting throughput and increasing energy consumption, while the harsh conditions necessary for carbene formation can lead to unpredictable side reactions and lower overall selectivity.
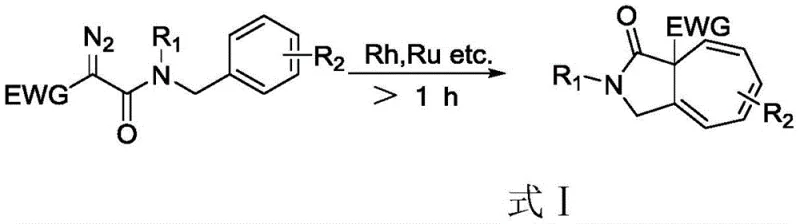
The Novel Approach
In stark contrast, the novel approach detailed in the patent utilizes a metal-free oxidative cyclization strategy driven by hypervalent iodine(III) reagents. The general reaction pathway, depicted in Formula III, demonstrates the transformation of readily available substituted N-alkoxy benzamides bearing alkynyl tethers into complex fused tricyclic systems. This method operates under exceptionally mild conditions, utilizing trifluoroethanol as a solvent at room temperature, thereby eliminating the need for external heating or cooling infrastructure. The reaction kinetics are remarkably fast, reaching completion in approximately one minute, which drastically enhances process throughput. By replacing dangerous diazo substrates with stable N-alkoxy benzamides and substituting expensive noble metals with commodity iodine reagents, this route offers a safer, faster, and more economically attractive alternative for the synthesis of high-value cycloheptatriene derivatives.
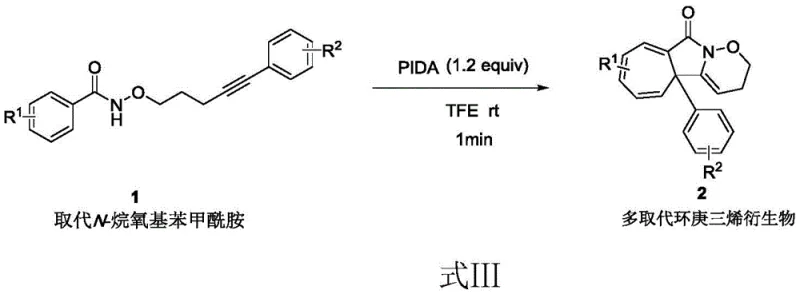
Mechanistic Insights into Hypervalent Iodine Mediated Oxidative Cyclization
The mechanistic underpinning of this transformation involves the activation of the amide nitrogen or the proximal alkyne by the electrophilic iodine(III) species, initiating an intramolecular cascade that constructs the seven-membered ring. Unlike radical-based metal carbene insertions, this ionic or concerted pathway allows for precise control over the regioselectivity of the cyclization. The hypervalent iodine acts as a two-electron oxidant, facilitating the formation of key C-N and C-C bonds simultaneously or sequentially to forge the rigid spiro-fused architecture observed in the products. This mechanism is particularly advantageous for preserving sensitive functional groups on the aromatic rings, such as halogens, methoxy, or cyano groups, which might otherwise be compromised under the harsh thermal or photochemical conditions required for traditional carbene chemistry. The use of TFE as a solvent likely stabilizes cationic intermediates through hydrogen bonding, further accelerating the reaction rate and ensuring high conversion efficiency.
From an impurity control perspective, the mildness of the reaction conditions plays a critical role in minimizing the formation of polymeric byproducts or decomposition species often associated with reactive carbene intermediates. The short reaction window of one minute limits the exposure of the product to the oxidizing environment, preventing over-oxidation or degradation of the newly formed cycloheptatriene system. This inherent selectivity translates to a cleaner crude reaction profile, simplifying downstream purification processes. For quality control teams, this means a more consistent impurity profile and reduced burden on analytical resources to identify and quantify trace metal contaminants, as the process is entirely free of heavy metal catalysts. The robustness of this mechanism across various electronic environments on the benzene rings (electron-donating methyl/methoxy vs. electron-withdrawing chloro/cyano) underscores its versatility for generating diverse libraries of analogs for SAR studies.
How to Synthesize Polysubstituted Cycloheptatriene Derivatives Efficiently
The operational simplicity of this synthesis makes it highly accessible for both laboratory scale-up and industrial production. The procedure involves a straightforward mixing of reagents followed by a rapid workup, requiring minimal specialized equipment. The following section outlines the standardized protocol derived from the patent examples, ensuring reproducibility and high yield. Detailed step-by-step instructions for the synthesis are provided below to guide process chemists in implementing this technology.
- Charge a reactor with substituted N-alkoxy benzamide and iodobenzene diacetate (PIDA) in a molar ratio of 1: 1.2.
- Add trifluoroethanol (TFE) as the solvent and stir the mixture at room temperature for approximately 1 minute.
- Concentrate the filtrate using a rotary evaporator and purify the crude product via silica gel column chromatography to obtain the target derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this hypervalent iodine-mediated route offers transformative benefits that extend far beyond simple yield improvements. The shift away from noble metal catalysts fundamentally alters the cost structure of the manufacturing process, removing a major variable expense and reducing dependency on volatile precious metal markets. Additionally, the dramatic reduction in reaction time from hours to minutes significantly increases reactor turnover rates, allowing existing infrastructure to produce substantially more material without capital investment in new equipment. The use of stable, non-hazardous starting materials also lowers insurance and compliance costs associated with handling explosive diazo compounds, contributing to a safer and more sustainable operation.
- Cost Reduction in Manufacturing: The elimination of rhodium and ruthenium catalysts results in substantial direct cost savings, as these metals are among the most expensive reagents in organic synthesis. Furthermore, the removal of heavy metal scavenging steps from the downstream processing workflow reduces the consumption of auxiliary materials and simplifies waste treatment protocols. The high atom economy and excellent yields (ranging from 80% to 96% across various substrates) ensure that raw material utilization is maximized, minimizing waste generation and lowering the cost per kilogram of the final active pharmaceutical ingredient (API) intermediate.
- Enhanced Supply Chain Reliability: The reagents required for this process, specifically iodobenzene diacetate (PIDA) and substituted N-alkoxy benzamides, are commercially available commodity chemicals with robust global supply chains. Unlike specialized ligands or sensitive organometallic complexes that may have long lead times or single-source suppliers, these materials can be sourced from multiple vendors, mitigating the risk of supply disruptions. The stability of the starting materials allows for long-term storage without significant degradation, enabling manufacturers to maintain strategic inventory buffers and respond quickly to fluctuating market demands.
- Scalability and Environmental Compliance: Operating at room temperature eliminates the energy costs associated with heating or cryogenic cooling, making the process inherently more energy-efficient and easier to scale. The absence of toxic heavy metals simplifies environmental compliance and waste disposal, aligning with increasingly stringent global regulations on pharmaceutical manufacturing emissions. The rapid reaction kinetics allow for continuous flow processing opportunities, which can further enhance safety and scalability by minimizing the hold-up volume of reactive intermediates, thus facilitating the commercial scale-up of complex pharmaceutical intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis route. These answers are derived directly from the experimental data and technical specifications outlined in the patent documentation, providing clarity for stakeholders evaluating this technology for integration into their production pipelines.
Q: What are the primary advantages of this hypervalent iodine method over traditional transition metal catalysis?
A: The primary advantages include the elimination of expensive and toxic noble metal catalysts (such as Rhodium or Ruthenium), a drastic reduction in reaction time from over 1 hour to just 1 minute, and the ability to operate safely at room temperature without hazardous diazo compounds.
Q: What is the typical yield range for these cycloheptatriene derivatives?
A: According to the patent data, the method consistently provides high yields ranging from 80% to 96% across various substituted substrates, demonstrating excellent functional group tolerance.
Q: Is this process suitable for large-scale commercial manufacturing?
A: Yes, the process is highly scalable due to its mild conditions (room temperature), short reaction time, and the use of commercially available reagents like PIDA, which simplifies supply chain logistics and reduces production costs significantly.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Polysubstituted Cycloheptatriene Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting innovative synthetic methodologies to maintain a competitive edge in the global pharmaceutical market. Our team of expert process chemists has thoroughly analyzed the potential of this hypervalent iodine-mediated cyclization and is fully prepared to translate this laboratory-scale breakthrough into robust commercial manufacturing processes. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from pilot plant to full-scale manufacturing is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of cycloheptatriene derivative we deliver meets the highest standards of quality and consistency required by top-tier pharmaceutical companies.
We invite you to collaborate with us to leverage this cutting-edge technology for your next drug development project. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific production needs, highlighting exactly how this metal-free route can optimize your budget. Please contact our technical procurement team today to request specific COA data for our reference standards and comprehensive route feasibility assessments. Let us help you accelerate your timeline to market with a reliable, cost-effective, and scalable supply of high-purity cycloheptatriene intermediates.
