Advanced Asymmetric Allylation for High-Purity Rocaglaol Intermediates and Commercial Scale-Up
The pharmaceutical industry is constantly seeking robust methodologies to access complex chiral scaffolds with high fidelity, and patent CN113912577A presents a groundbreaking advancement in this domain. This intellectual property discloses a highly efficient preparation method for chiral allylated benzofuranone compounds, which serve as critical intermediates in the synthesis of Rocaglaol and its biologically active derivatives. The core innovation lies in a sophisticated dual-catalytic system that simultaneously controls two distinct chiral centers, overcoming the longstanding limitations of racemic synthesis. By leveraging a synergistic combination of Lewis acids, chiral nitrogen-oxygen ligands, iridium catalysts, and chiral phosphoramidite ligands, this technology enables the production of intermediates with exceptional diastereoselectivity and enantioselectivity. 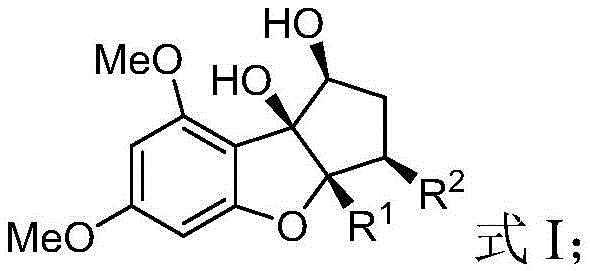 For R&D directors and process chemists, this represents a paradigm shift towards more predictable and scalable asymmetric synthesis, ensuring that the resulting pharmaceutical intermediates meet the stringent purity specifications required for modern drug development pipelines.
For R&D directors and process chemists, this represents a paradigm shift towards more predictable and scalable asymmetric synthesis, ensuring that the resulting pharmaceutical intermediates meet the stringent purity specifications required for modern drug development pipelines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Rocaglaol and related natural products has relied heavily on classical transformations such as Nazarov cyclization, [3+2] cycloaddition, or ethylene oxide ring-opening rearrangements. While these methods established the foundational chemistry for the scaffold, they suffer from significant drawbacks that hinder large-scale commercial viability. Primarily, these conventional routes predominantly yield racemic products, necessitating costly and inefficient resolution steps to isolate the desired enantiomer. Furthermore, the atom utilization rate in these traditional processes is often suboptimal, leading to excessive waste generation and higher environmental burdens. The reaction conditions can also be harsh, limiting the tolerance for diverse functional groups and restricting the scope of substrate substitution. Consequently, achieving high diastereoselectivity and enantioselectivity simultaneously has remained a formidable challenge, often resulting in complex impurity profiles that complicate downstream purification and regulatory approval.
The Novel Approach
In stark contrast, the methodology described in patent CN113912577A introduces a streamlined and highly selective allylation strategy that directly addresses these inefficiencies. The novel approach utilizes readily available benzofuranone compounds and allyl carbonates as starting materials, which are subjected to a cooperative catalytic system. This system orchestrates the formation of two chiral centers in a single operational sequence with remarkable precision. The use of a dual-ligand environment allows for independent modulation of the nucleophilic and electrophilic partners, ensuring that the reaction proceeds with high regioselectivity and stereocontrol. This not only drastically simplifies the synthetic route by reducing the number of steps but also significantly enhances the overall yield and purity of the final intermediate. For procurement managers, this translates to a more reliable supply chain for high-purity pharmaceutical intermediates, as the process minimizes the risk of batch-to-batch variability associated with difficult separations.
Mechanistic Insights into Dual-Catalytic Asymmetric Allylation
The mechanistic elegance of this synthesis lies in the precise orchestration of two distinct catalytic cycles that operate in concert. The first cycle involves the activation of the benzofuranone substrate (Compound A) by a Lewis acid, such as nickel triflate or cobalt tetrafluoroborate hexahydrate, in the presence of a chiral nitrogen-oxygen ligand. This coordination reduces the electron cloud density of the oxygen atom, thereby enhancing the nucleophilicity of the carbon at the reaction site while imposing a specific spatial configuration. Simultaneously, the second cycle activates the allyl carbonate (Compound B) through oxidative addition with an iridium catalyst and a chiral phosphoramidite ligand.  This generates a pi-allyl-iridium electrophile with defined chirality. When these two activated species meet in the presence of a base like potassium carbonate, a highly stereoselective nucleophilic substitution occurs. The synergy between the Lewis acid/chiral N-O ligand pair and the Ir/phosphoramidite pair ensures that both chiral centers are established with high fidelity, effectively suppressing the formation of unwanted diastereomers and enantiomers.
This generates a pi-allyl-iridium electrophile with defined chirality. When these two activated species meet in the presence of a base like potassium carbonate, a highly stereoselective nucleophilic substitution occurs. The synergy between the Lewis acid/chiral N-O ligand pair and the Ir/phosphoramidite pair ensures that both chiral centers are established with high fidelity, effectively suppressing the formation of unwanted diastereomers and enantiomers.
From an impurity control perspective, this mechanism offers substantial advantages over traditional radical or non-catalytic pathways. The mild reaction conditions, typically conducted at temperatures between 0°C and 60°C, prevent thermal degradation and side reactions that often plague high-energy processes. The specific choice of ligands, such as the optimized L2 chiral nitrogen-oxygen ligand and the (S,S,S)-L1 phosphoramidite ligand, creates a rigid chiral pocket that sterically hinders the approach of reactants in unfavorable orientations. This steric guidance is crucial for achieving the reported diastereomeric ratios (d.r.) of up to >19:1 and enantiomeric excess (ee) values exceeding 99%. For quality assurance teams, this inherent selectivity means that the crude product profile is much cleaner, reducing the load on chromatographic purification systems and ensuring that the final API intermediate consistently meets rigorous pharmacopeial standards without extensive reprocessing.
How to Synthesize Chiral Allylated Benzofuranone Efficiently
The practical implementation of this synthesis involves a carefully sequenced addition of reagents to maintain the integrity of the catalytic species. The process begins with the preparation of two separate solutions: one containing the benzofuranone, Lewis acid, and chiral N-O ligand, and the other containing the allyl carbonate, iridium catalyst, and phosphoramidite ligand. These are typically stirred in a mixture of dichloromethane and dichloroethane to ensure optimal solubility and reaction kinetics. This modular approach allows for precise control over the concentration and activation state of each component before they are combined, which is critical for maximizing the synergistic effects of the dual catalytic system.
- Prepare a first mixed solution by reacting benzofuranone compound A with a Lewis acid and chiral nitrogen-oxygen ligand in an organic solvent.
- Prepare a second mixed solution by reacting allyl carbonate compound B with an Iridium catalyst and chiral phosphoramidite ligand.
- Combine both solutions with an alkaline reagent to perform nucleophilic substitution, yielding the chiral allylated benzofuranone with high stereoselectivity.
Commercial Advantages for Procurement and Supply Chain Teams
For stakeholders focused on the bottom line and supply continuity, the adoption of this patented technology offers compelling strategic benefits. The shift from multi-step, low-yield racemic syntheses to a direct, high-selectivity allylation process fundamentally alters the cost structure of producing Rocaglaol intermediates. By eliminating the need for resolution steps and reducing the number of unit operations, manufacturers can achieve significant cost reduction in pharmaceutical intermediates manufacturing. The use of commercially available starting materials further de-risks the supply chain, as there is no dependency on exotic or custom-synthesized precursors that might face availability bottlenecks. Additionally, the high atom utilization rate implies less waste disposal cost and a smaller environmental footprint, aligning with increasingly strict global sustainability mandates.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal removal steps and the reduction in solvent usage due to fewer reaction stages contribute to a leaner production model. The high yields reported in the patent examples suggest that less raw material is required per kilogram of finished product, directly lowering the cost of goods sold (COGS). Furthermore, the ability to run the reaction at near-room temperature reduces energy consumption associated with heating or cryogenic cooling, providing additional operational savings.
- Enhanced Supply Chain Reliability: The robustness of the catalytic system against variations in substrate substitution means that a wider range of derivatives can be produced using the same core infrastructure. This flexibility allows suppliers to respond quickly to changing market demands for specific Rocaglaol analogs without retooling entire production lines. The use of stable ligands and common solvents like DCM and DCE ensures that the process is not vulnerable to supply disruptions of specialized reagents, thereby securing the continuity of supply for downstream drug manufacturers.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing standard organic synthesis techniques that translate well from laboratory to pilot and commercial scales. The high selectivity minimizes the generation of hazardous byproducts, simplifying wastewater treatment and废气 handling. This environmental compatibility facilitates easier regulatory approval for new manufacturing sites and reduces the long-term liability associated with chemical waste management, making it an attractive option for sustainable chemical production.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this asymmetric allylation technology. These insights are derived directly from the experimental data and beneficial effects outlined in the patent documentation, providing clarity on the process capabilities and limitations. Understanding these nuances is essential for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: What are the advantages of this dual-catalytic method over traditional Nazarov cyclization?
A: Unlike traditional methods that often yield racemic mixtures with low atom utilization, this dual-catalytic approach achieves high diastereoselectivity and enantioselectivity (>99% ee) under mild conditions, significantly simplifying downstream purification.
Q: Which catalysts are critical for the stereoselectivity in this synthesis?
A: The process relies on a synergistic system involving a Lewis acid (e.g., Ni(OTf)2 or Co(BF4)2) paired with a chiral nitrogen-oxygen ligand, and an Iridium catalyst paired with a chiral phosphoramidite ligand to control the two chiral centers independently.
Q: What are the primary therapeutic applications of the resulting Rocaglaol derivatives?
A: The synthesized intermediates are pivotal for producing Rocaglaol derivatives, which exhibit potent biological activities against hepatitis C, leukemia, various cancers, and coronaviruses, making them valuable assets in antiviral and oncology drug development.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Benzofuranone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the dual-catalytic asymmetric allylation technology described in CN113912577A for the production of high-value antiviral and anticancer intermediates. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from benchtop discovery to full-scale manufacturing. Our facilities are equipped with stringent purity specifications and rigorous QC labs capable of verifying the high enantiomeric excess and diastereoselectivity required for these complex chiral building blocks. We are committed to delivering consistent quality that supports your clinical and commercial timelines.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis route can optimize your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain a detailed understanding of the economic benefits specific to your volume requirements. We encourage potential partners to contact us for specific COA data and route feasibility assessments, allowing us to demonstrate our capability to deliver reliable pharmaceutical intermediates that meet the highest industry standards.
