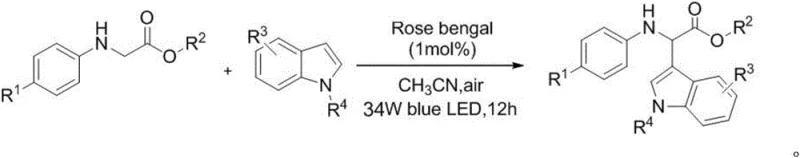
Scalable Photocatalytic Synthesis of Alpha-Indole Glycine Derivatives for Pharma Intermediates
The pharmaceutical industry continuously seeks robust and environmentally benign pathways for constructing complex heterocyclic scaffolds, particularly those serving as critical precursors for non-steroidal anti-inflammatory drugs (NSAIDs). A significant breakthrough in this domain is detailed in patent CN110452151B, which discloses a novel synthetic methodology for alpha-indole glycine derivatives. These compounds are pivotal intermediates in the manufacture of clinically significant agents such as etodolac and pemedolac, which are widely utilized for postoperative pain management and inflammation control. The disclosed technology represents a paradigm shift from traditional transition-metal catalysis to visible-light-driven organophotocatalysis, utilizing the organic dye Rose Bengal. This approach not only addresses the growing regulatory pressure regarding heavy metal impurities in active pharmaceutical ingredients but also offers a streamlined operational profile that is highly attractive for large-scale manufacturing. By leveraging ambient air as the terminal oxidant and operating at room temperature, this process effectively lowers the barrier to entry for producing high-purity pharmaceutical intermediates while minimizing the environmental footprint associated with synthetic chemistry.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of alpha-indole glycine motifs has relied heavily on Mannich-type Friedel-Crafts reactions or cross-dehydrogenative coupling (CDC) strategies that present substantial logistical and safety challenges. Traditional Mannich pathways necessitate the use of imine intermediates, which are notoriously unstable in atmospheric conditions and prone to hydrolysis, thereby complicating storage and handling protocols while introducing variability in reaction outcomes. Furthermore, existing CDC methodologies frequently depend on stoichiometric amounts of toxic transition metals like copper or iron, or precious metal photocatalysts based on ruthenium and iridium complexes. The reliance on these metallic species introduces a critical bottleneck in the supply chain, as removing trace metal residues to meet stringent pharmacopeial standards requires additional, costly purification steps such as scavenging or recrystallization. Additionally, many conventional oxidative coupling protocols demand the use of hazardous organic peroxides or pure oxygen atmospheres, creating significant safety risks related to explosion hazards and requiring specialized pressure-rated equipment that increases capital expenditure for production facilities.
The Novel Approach
In stark contrast to these legacy methods, the innovative protocol described in the patent utilizes a metal-free organic photocatalyst, Rose Bengal, to drive the cross-dehydrogenative coupling between glycine derivatives and substituted indoles. This method operates under exceptionally mild conditions, specifically at room temperature and under ambient air, eliminating the need for energy-intensive heating or cooling cycles and inert gas purging. The use of visible light irradiation, typically from commercially available blue LEDs, provides the necessary energy to excite the catalyst without generating excessive heat or requiring high-pressure setups. This transition to an organocatalytic system fundamentally alters the economic and safety profile of the synthesis by removing the dependency on volatile oxidants and expensive metal salts. Consequently, the process facilitates a cleaner reaction profile with fewer byproducts, directly translating to simplified workup procedures and higher overall throughput for manufacturers aiming to scale up the production of these valuable anti-inflammatory intermediates without compromising on safety or purity standards.
Mechanistic Insights into Rose Bengal-Catalyzed Cross-Dehydrogenative Coupling
The efficacy of this synthetic route is underpinned by a sophisticated single-electron transfer (SET) mechanism initiated by visible light absorption. Upon irradiation with blue light, the Rose Bengal catalyst absorbs photons and transitions from its ground state to an excited singlet state, which rapidly undergoes intersystem crossing to a long-lived triplet excited state. In this highly energetic state, the catalyst acts as a potent oxidant, accepting a single electron from the nitrogen atom of the glycine ester derivative. This electron transfer event generates a glycine-derived radical cation intermediate and a Rose Bengal radical anion. The radical cation subsequently undergoes deprotonation at the alpha-carbon position to form a neutral carbon-centered radical, which is a key reactive species in the bond-forming sequence. Simultaneously, the reduced Rose Bengal radical anion transfers its extra electron to molecular oxygen present in the air, regenerating the ground-state catalyst and producing a superoxide radical anion. This catalytic cycle ensures that the organic dye is continuously recycled, allowing for high turnover numbers with only catalytic loadings, typically around 1 mol%, which is economically advantageous for industrial applications.
The final stage of the mechanism involves the oxidation of the carbon-centered radical to an electrophilic iminium ion species, driven by the superoxide radical or other oxygen-derived reactive species generated in situ. This highly reactive iminium intermediate is then susceptible to nucleophilic attack by the electron-rich C3 position of the indole ring. This carbon-carbon bond formation step constructs the core alpha-indole glycine skeleton with high regioselectivity. The use of ambient air as the terminal oxidant is particularly elegant, as it avoids the accumulation of hazardous waste streams associated with chemical oxidants. Furthermore, the mild nature of this radical pathway ensures compatibility with a wide range of functional groups, including halogens and alkoxy substituents, which might be sensitive to harsher acidic or basic conditions found in traditional Friedel-Crafts chemistry. This mechanistic robustness allows for the synthesis of diverse derivatives without the need for extensive protecting group strategies, thereby shortening the overall synthetic timeline.
How to Synthesize Alpha-Indole Glycine Derivatives Efficiently
The practical implementation of this photocatalytic method is straightforward and designed for ease of adoption in both laboratory and pilot plant settings. The procedure typically involves dissolving the glycine ester substrate and the substituted indole in acetonitrile, a polar aprotic solvent that effectively solubilizes both organic components and supports the photoredox cycle. A catalytic amount of Rose Bengal is added to the mixture, which is then stirred at room temperature while being exposed to blue LED light sources. The reaction progress is monitored until completion, usually within 12 hours, after which standard extraction and chromatographic techniques are employed to isolate the pure product. This operational simplicity removes the need for specialized high-pressure reactors or glovebox environments, making it accessible for contract development and manufacturing organizations looking to optimize their intermediate production lines. For detailed standardized synthesis steps and specific molar ratios, please refer to the guide below.
- Combine glycine derivative and substituted indole in acetonitrile solvent with 1 mol% Rose Bengal catalyst.
- Stir the mixture at room temperature under air atmosphere while irradiating with 34W blue LED light for 12 hours.
- Purify the crude reaction mixture via column chromatography using petroleum ether and ethyl acetate to isolate the target derivative.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic procurement and supply chain perspective, the adoption of this Rose Bengal-mediated photocatalytic process offers compelling advantages that directly impact the bottom line and operational resilience. The elimination of precious metal catalysts such as ruthenium or iridium removes a significant cost driver and mitigates supply chain volatility associated with the mining and refining of rare earth elements. Furthermore, the ability to run reactions under ambient air conditions rather than requiring pure oxygen or inert nitrogen blankets simplifies facility requirements and reduces utility costs related to gas consumption and monitoring. The mild reaction conditions also imply lower energy consumption for heating or cooling, contributing to a more sustainable manufacturing profile that aligns with modern corporate sustainability goals. These factors collectively enhance the reliability of supply by reducing the complexity of the manufacturing process and minimizing the risk of batch failures due to equipment malfunction or reagent instability.
- Cost Reduction in Manufacturing: The substitution of expensive transition metal catalysts with commodity-grade organic dyes like Rose Bengal results in substantial raw material cost savings. Since the catalyst loading is low and the dye is inexpensive, the overall cost of goods sold (COGS) for the intermediate is significantly reduced. Additionally, the avoidance of heavy metals eliminates the need for costly metal scavenging resins and extensive analytical testing for residual metals, further streamlining the purification budget. The use of simple acetonitrile solvent and ambient conditions also reduces energy overheads, making the process economically superior to thermal or high-pressure alternatives.
- Enhanced Supply Chain Reliability: By relying on stable, commercially available starting materials and avoiding moisture-sensitive imines or hazardous peroxides, the supply chain becomes more robust against disruptions. The reagents required, such as substituted indoles and glycine esters, are widely sourced from multiple global suppliers, reducing single-source dependency risks. The operational simplicity means that production can be easily transferred between different manufacturing sites without the need for highly specialized infrastructure, ensuring continuity of supply even during regional logistical challenges or facility maintenance periods.
- Scalability and Environmental Compliance: The photochemical nature of the reaction is inherently scalable using modern flow chemistry or large-surface-area batch reactors, allowing for seamless transition from gram-scale development to multi-ton commercial production. The green chemistry credentials of the process, characterized by the use of air as an oxidant and the generation of minimal toxic waste, facilitate easier regulatory approval and environmental permitting. This compliance advantage accelerates time-to-market for new drug formulations relying on these intermediates and reduces the long-term liability associated with hazardous waste disposal and environmental remediation.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this photocatalytic technology. These answers are derived directly from the experimental data and beneficial effects reported in the patent literature, providing a clear understanding of the process capabilities and limitations. Understanding these details is crucial for R&D teams evaluating the feasibility of integrating this route into their existing pipelines and for procurement officers assessing the long-term viability of the supply source.
Q: What are the advantages of using Rose Bengal over traditional metal catalysts?
A: Rose Bengal is an inexpensive organic dye that eliminates the risk of heavy metal residues common with copper or ruthenium catalysts, significantly simplifying downstream purification and regulatory compliance for pharmaceutical applications.
Q: Does this photocatalytic method require specialized equipment?
A: No, the process operates at room temperature under ambient air conditions using standard blue LED light sources, removing the need for expensive inert gas lines, high-pressure reactors, or cryogenic cooling systems.
Q: What is the substrate scope for this synthesis method?
A: The method demonstrates broad compatibility with various substituents on both the glycine ester and the indole ring, including electron-donating methoxy groups and electron-withdrawing halogens, maintaining high yields across diverse structures.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alpha-Indole Glycine Derivatives Supplier
As the global demand for high-quality anti-inflammatory intermediates continues to rise, partnering with a technically proficient manufacturer is essential for securing a competitive edge. NINGBO INNO PHARMCHEM stands at the forefront of this sector, leveraging advanced photocatalytic technologies to deliver superior alpha-indole glycine derivatives. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the volumetric requirements of major pharmaceutical clients without compromising on quality. We maintain stringent purity specifications through our rigorous QC labs, employing state-of-the-art analytical instrumentation to verify that every batch meets the exacting standards required for API synthesis, thereby safeguarding your downstream production processes.
We invite you to collaborate with us to optimize your supply chain for these critical pharmaceutical building blocks. Our technical sales team is prepared to provide a Customized Cost-Saving Analysis tailored to your specific volume needs, demonstrating how our efficient synthetic routes can reduce your overall procurement costs. We encourage you to contact our technical procurement team today to request specific COA data for our current inventory and to discuss route feasibility assessments for your custom projects, ensuring a seamless integration of our high-purity intermediates into your manufacturing workflow.
