Scalable Copper-Catalyzed Synthesis of Coumarin Fluorescent Agents for Optoelectronic Applications
The landscape of fluorescent material manufacturing is undergoing a significant transformation driven by the need for cost-effective and environmentally benign synthetic routes. A pivotal advancement in this domain is detailed in patent CN110343083B, which discloses a novel method for preparing coumarin-based fluorescent agents utilizing copper acetate catalysis. This technology represents a strategic departure from traditional precious metal-catalyzed processes, offering a robust pathway for the synthesis of high-performance optoelectronic materials. By leveraging a base metal catalyst system comprising copper acetate and 1,8-diazabicyclo[5.4.0]undec-7-ene (DBU), the process achieves high chemical selectivity under remarkably mild conditions. For R&D directors and procurement specialists alike, this innovation signals a potential paradigm shift in how complex fluorophores are sourced, promising enhanced supply chain stability and reduced manufacturing overheads without compromising on the photophysical properties essential for next-generation display and sensing applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of sophisticated coumarin derivatives has relied heavily on transition metal catalysis involving palladium or platinum complexes, which present substantial logistical and economic challenges for large-scale operations. These conventional methodologies often necessitate multi-step coupling sequences that suffer from low atom economy and require stringent exclusion of moisture and oxygen, thereby increasing operational complexity and energy consumption. Furthermore, the reliance on precious metals introduces significant cost volatility and creates rigorous downstream purification requirements to meet the stringent heavy metal limits imposed by regulatory bodies in the pharmaceutical and electronic sectors. The accumulation of toxic metal residues not only complicates waste management but also poses risks to the final product's performance stability, particularly in sensitive optoelectronic devices where trace impurities can act as quenching sites. Consequently, the industry has long sought a catalytic system that balances efficiency with economic and environmental sustainability.
The Novel Approach
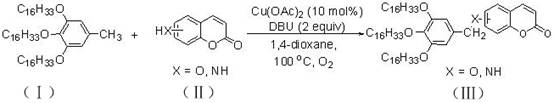
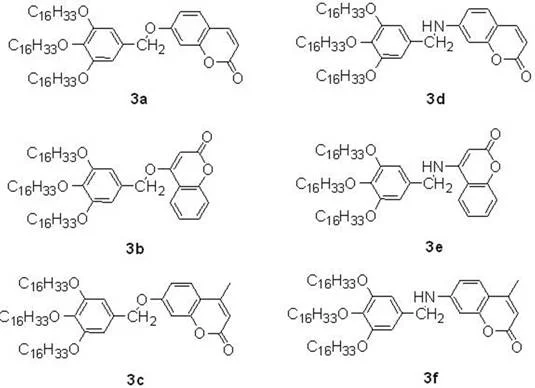
In stark contrast to these legacy methods, the copper acetate-catalyzed protocol described in the patent data offers a streamlined, one-pot solution that directly couples 3,4,5-trihexadecyloxytoluene with various coumarin precursors. This approach utilizes earth-abundant copper species which are not only inexpensive but also exhibit lower toxicity profiles, aligning perfectly with modern green chemistry principles. The reaction proceeds efficiently in 1,4-dioxane at a moderate temperature of 100°C under an ambient air atmosphere, eliminating the need for expensive inert gas shielding or high-pressure reactors. This simplicity translates directly into operational ease, allowing for the direct functionalization of the coumarin core with long-chain alkoxy groups that enhance solubility and processability in organic matrices. The versatility of this system is further evidenced by its tolerance to different substituents on the coumarin ring, enabling the rapid generation of diverse fluorescent libraries.
Mechanistic Insights into Copper Acetate-Catalyzed Oxidative Coupling
The mechanistic underpinning of this transformation likely involves a copper-mediated oxidative C-H functionalization pathway, where the copper(II) species acts as both a Lewis acid activator and a redox mediator. In the presence of DBU, a strong non-nucleophilic base, the benzylic C-H bond of the trihexadecyloxytoluene substrate is activated, facilitating the formation of a reactive organocopper intermediate or a radical species. Molecular oxygen from the air serves as the terminal oxidant, regenerating the active copper catalyst and driving the thermodynamic equilibrium towards the formation of the new carbon-carbon or carbon-heteroatom bond. This aerobic oxidation mechanism is particularly advantageous as it avoids the use of stoichiometric hazardous oxidants, thereby reducing the generation of chemical waste. The coordination of the coumarin substrate to the copper center directs the regioselectivity of the attack, ensuring that the functionalization occurs specifically at the desired position on the heterocyclic ring, which is critical for maintaining the fluorescence quantum yield.
From an impurity control perspective, the use of DBU as a base plays a dual role in suppressing side reactions such as homocoupling or over-oxidation of the sensitive coumarin lactone ring. The specific stoichiometry of 2 equivalents of DBU ensures complete deprotonation of the intermediate species while buffering the reaction medium against acidity that could lead to hydrolysis of the ester moiety. Furthermore, the choice of 1,4-dioxane as a solvent is not arbitrary; its ability to solubilize both the long-chain lipophilic substrates and the polar catalytic species creates a homogeneous reaction environment that maximizes collision frequency and reaction rates. This homogeneity is crucial for preventing the formation of insoluble polymeric byproducts that often plague heterogeneous catalytic systems. The result is a clean reaction profile that yields the target fluorescent agents with minimal structural defects, ensuring consistent batch-to-batch reproducibility which is paramount for commercial applications.
How to Synthesize Coumarin Fluorescent Agents Efficiently
The practical implementation of this synthesis route is designed for scalability and ease of execution, making it highly attractive for process chemists aiming to transition from laboratory discovery to pilot plant production. The standardized protocol involves a straightforward charging sequence followed by a thermal incubation period that requires minimal intervention, allowing technical teams to focus on optimization rather than troubleshooting complex equipment setups. Detailed below is the generalized workflow derived from the patent examples, which serves as a foundational guide for establishing robust manufacturing parameters. Adhering to these specific molar ratios and thermal conditions is essential to replicate the high yields and purity levels reported in the intellectual property documentation.
- Charge a reaction vessel with 3,4,5-trihexadecyloxytoluene, coumarin substrate, copper acetate (10 mol%), and DBU (2 equiv) in 1,4-dioxane solvent.
- Heat the reaction mixture to 100°C and stir for 20 hours under an air atmosphere to facilitate oxidative coupling.
- Cool to room temperature, quench with saturated NaCl, extract with ethyl acetate, dry over Na2SO4, and purify via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this copper-catalyzed methodology presents a compelling value proposition centered around cost rationalization and risk mitigation. The substitution of expensive palladium catalysts with commodity-grade copper acetate drastically reduces the raw material cost baseline, insulating the production budget from the volatile fluctuations typical of the precious metals market. Moreover, the elimination of specialized ligands and the use of air as a reagent simplify the bill of materials, reducing the number of SKUs that need to be managed and qualified. This simplification extends to the infrastructure requirements, as the mild reaction conditions allow for the utilization of standard glass-lined or stainless steel reactors without the need for exotic alloys resistant to harsh halogenated environments. Consequently, the barrier to entry for manufacturing these high-value intermediates is significantly lowered, fostering a more competitive and resilient supply base.
- Cost Reduction in Manufacturing: The economic benefits of this process are multifaceted, stemming primarily from the drastic reduction in catalyst costs and the simplification of the purification train. By avoiding precious metals, manufacturers save substantially on both the initial purchase price and the costly recovery or disposal processes associated with heavy metal waste streams. The high conversion rates and selectivity minimize the loss of valuable starting materials, improving the overall mass balance and reducing the cost per kilogram of the final active ingredient. Additionally, the energy demand is optimized by operating at 100°C, which is achievable with standard steam heating systems rather than requiring high-energy electrical heating or cryogenic cooling for exothermic control. These cumulative efficiencies translate into a leaner cost structure that enhances margin potential in a competitive global market.
- Enhanced Supply Chain Reliability: Supply chain continuity is bolstered by the widespread availability of the key reagents, specifically copper acetate and DBU, which are produced at massive scales for various industrial applications. Unlike specialized phosphine ligands or organometallic complexes that may have single-source suppliers and long lead times, these commoditized chemicals can be sourced from multiple vendors globally, mitigating the risk of supply disruptions. The robustness of the reaction to air and moisture further reduces the logistical burden, as reagents do not require stringent anhydrous packaging or nitrogen-blanketed transport. This resilience ensures that production schedules can be maintained even during periods of logistical strain, providing a reliable flow of materials to downstream formulation partners who depend on just-in-time delivery models.
- Scalability and Environmental Compliance: Scaling this process from gram to tonnage quantities is facilitated by the inherent safety and simplicity of the reaction conditions, which do not involve pyrophoric reagents or high-pressure hydrogenation steps. The use of 1,4-dioxane, while requiring careful handling, allows for efficient solvent recovery and recycling due to its favorable boiling point and phase separation characteristics with water. From an environmental compliance standpoint, the lower toxicity of copper compared to other transition metals simplifies the permitting process for new manufacturing lines and reduces the liability associated with effluent discharge. The process aligns well with increasingly strict global regulations regarding heavy metal residues in electronic and consumer products, future-proofing the supply chain against tightening legislative frameworks and enhancing the brand reputation of the end-product manufacturers.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this copper-catalyzed synthesis technology. These insights are derived directly from the experimental data and beneficial effects outlined in the patent documentation, providing clarity on the operational feasibility and strategic advantages of adopting this route. Understanding these nuances is critical for stakeholders evaluating the integration of this chemistry into their existing production portfolios or R&D pipelines.
Q: Why is copper acetate preferred over palladium catalysts for this synthesis?
A: Copper acetate is significantly more cost-effective and less toxic than precious metal catalysts like palladium, reducing both raw material costs and heavy metal removal burdens in downstream processing.
Q: What are the purification advantages of using 1,4-dioxane as the solvent?
A: 1,4-Dioxane is miscible with water, which simplifies the aqueous workup process and allows for efficient separation of organic products from inorganic salts and catalyst residues.
Q: Is this synthetic route suitable for large-scale industrial production?
A: Yes, the reaction operates at atmospheric pressure and moderate temperatures (100°C) using air as the oxidant, eliminating the need for specialized high-pressure equipment or inert gas protection.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Coumarin Fluorescent Agent Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this copper-catalyzed technology in advancing the field of optoelectronic materials and specialty chemicals. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of coumarin fluorescent agent delivers the consistent photophysical performance required for high-end applications. We are committed to leveraging our technical expertise to optimize this specific copper acetate route, tailoring the process parameters to maximize yield and minimize environmental impact for our global clientele.
We invite forward-thinking organizations to collaborate with us to unlock the full commercial potential of these advanced fluorescent materials. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis that quantifies the economic benefits of switching to this catalytic system for your specific volume requirements. We encourage you to reach out today to request specific COA data and route feasibility assessments, allowing us to demonstrate how our manufacturing capabilities can support your supply chain goals and drive innovation in your product development cycles.
