Advanced C-H Activation Strategy for Scalable 3-Acylated Indole Production
Advanced C-H Activation Strategy for Scalable 3-Acylated Indole Production
The pharmaceutical industry constantly seeks more efficient pathways to construct complex heterocyclic scaffolds, and the recent technological breakthrough detailed in patent CN109265386B represents a significant leap forward in indole chemistry. This novel methodology introduces a robust, one-step synthesis of 3-acylated indole derivatives utilizing aromatic amidines and α-carbonyl sulfur ylides as key building blocks. By leveraging transition metal-catalyzed C-H activation, this process bypasses the limitations of classical multi-step routes, offering a streamlined approach that is particularly valuable for the rapid generation of diverse chemical libraries. For R&D teams focused on drug discovery, the ability to access these privileged structures with high regioselectivity and under mild conditions opens new avenues for exploring biological activity without the burden of complex synthetic planning.
From a commercial manufacturing perspective, the implications of this patent are profound, addressing critical pain points related to cost, scalability, and environmental impact. The elimination of pre-functionalization steps and the use of readily available starting materials position this technology as a highly attractive option for reliable pharmaceutical intermediate suppliers aiming to optimize their production portfolios. As we delve deeper into the technical specifics, it becomes clear that this C-H activation strategy not only enhances synthetic efficiency but also aligns perfectly with modern green chemistry principles, making it an ideal candidate for cost reduction in API manufacturing where margin compression is a constant challenge.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the indole core has relied heavily on classical methodologies such as the Fischer indole synthesis, Nenitzescu synthesis, or various aniline derivative cyclizations. While these methods established the foundation of indole chemistry, they are increasingly viewed as suboptimal for modern high-throughput and large-scale applications. The Fischer synthesis, for instance, often struggles with poor regioselectivity when unsymmetrical ketones are employed, leading to difficult-to-separate isomeric mixtures that compromise overall yield and purity. Furthermore, many traditional routes necessitate harsh reaction conditions, including the use of strong acids, high temperatures, or stoichiometric amounts of toxic oxidants like copper or silver salts, which generate substantial hazardous waste.
Another significant drawback of conventional nitro-derivative synthesis methods is their limited scope regarding intermolecular reactions; they are frequently restricted to intramolecular cyclizations of acetylene bonds, severely limiting the structural diversity accessible to chemists. The requirement for protecting groups in many of these older pathways adds unnecessary synthetic steps, increasing both the material cost and the time-to-market for new drug candidates. Additionally, the sensitivity of some traditional methods to moisture and oxygen requires specialized equipment and rigorous exclusion protocols, further complicating the scale-up process and increasing operational expenditures for manufacturing facilities.
The Novel Approach
In stark contrast to these legacy methods, the novel approach disclosed in the patent utilizes a direct C-H activation strategy that fundamentally simplifies the synthetic landscape. By employing aromatic amidines as intrinsic directing groups, the reaction achieves precise regiocontrol, specifically targeting the 3-position of the emerging indole ring without the need for pre-installed halogen handles or other activating groups. This atom-economical transformation couples the amidine directly with α-carbonyl sulfur ylides, effectively constructing the entire indole skeleton and installing the acyl functionality in a single operational step. The reaction proceeds under remarkably mild conditions, typically at 80°C in common organic solvents, which drastically reduces energy consumption compared to high-temperature alternatives.
The versatility of this new method is evidenced by its exceptional functional group tolerance, accommodating a wide range of substituents including halogens, nitro groups, esters, and ethers without degradation or side reactions. This robustness eliminates the need for complex protection-deprotection sequences, thereby shortening the synthetic route and improving the overall mass balance of the process. For process chemists, this means a significant reduction in unit operations and solvent usage, directly translating to a more sustainable and economically viable manufacturing process. The ability to synthesize 3-acylated indoles efficiently makes this technology a powerful tool for accessing bioactive molecules found in natural products and pharmaceutical agents.
Mechanistic Insights into Rh/Ir-Catalyzed C-H Activation Cyclization
The core of this transformative synthesis lies in the sophisticated mechanism of transition metal-catalyzed C-H bond activation, specifically utilizing Rhodium (Rh) or Iridium (Ir) complexes. The reaction initiates with the coordination of the amidine nitrogen to the metal center, which acts as a directing group to facilitate the selective cleavage of the proximal C-H bond on the aromatic ring. This metallacycle formation is the critical determinant of regioselectivity, ensuring that the subsequent insertion of the sulfur ylide occurs exclusively at the desired position to form the indole core. The use of pentamethylcyclopentadienyl (Cp*) ligands on the metal center provides the necessary steric and electronic environment to stabilize the active catalytic species throughout the cycle.
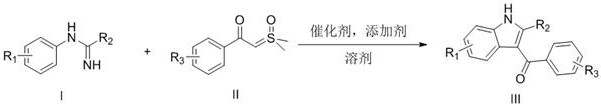
Following the C-H activation step, the α-carbonyl sulfur ylide undergoes insertion into the metal-carbon bond, followed by a series of rearrangements and eliminations that ultimately release the sulfur byproduct and close the pyrrole ring. The presence of additives such as silver hexafluoroantimonate or acetates plays a crucial role in generating the cationic active catalyst species and facilitating the departure of leaving groups. This mechanistic pathway is highly efficient, as demonstrated by the reported yields ranging from 72% to 86% across various substrates in the patent examples. The mild thermal requirements (80°C) suggest a low activation energy barrier for the rate-determining steps, which is advantageous for maintaining the integrity of sensitive functional groups during the transformation.
From an impurity control perspective, this mechanism offers distinct advantages over radical-based or oxidative cyclizations. Because the reaction proceeds through a well-defined organometallic cycle rather than uncontrolled radical propagation, the formation of polymeric byproducts or random coupling impurities is minimized. The specificity of the amidine directing group ensures that side reactions at other positions on the aromatic ring are suppressed, leading to a cleaner crude reaction profile. This high level of selectivity simplifies downstream purification, often allowing for straightforward silica gel chromatography to achieve analytical purity, which is a critical factor for high-purity pharmaceutical intermediates intended for clinical use.
How to Synthesize 3-Acylated Indole Derivatives Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires careful attention to the stoichiometry and reaction environment to maximize the benefits of the catalytic system. The standard protocol involves charging a clean reactor with the aromatic amidine substrate and the α-carbonyl sulfur ylide in a molar ratio of approximately 1:2, ensuring an excess of the ylide to drive the reaction to completion. A transition metal catalyst, such as dichloro(pentamethylcyclopentadienyl)rhodium(III) dimer, is added at a loading of roughly 5 mol%, along with a silver salt additive like silver hexafluoroantimonate to activate the catalyst. The reaction mixture is then dissolved in a suitable solvent such as 1,2-dichloroethane or toluene and heated to 80°C under an inert argon atmosphere for 24 hours.
- Charge a reactor with aromatic amidine, sulfur ylide, Rh/Ir catalyst, silver/additive salts, and solvent (e.g., DCE) under argon protection.
- Heat the reaction mixture to 80°C in an oil bath and stir continuously for 24 hours to ensure complete conversion.
- Remove solvent under reduced pressure and purify the crude residue via silica gel column chromatography to isolate the target indole derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this C-H activation technology offers tangible strategic benefits that extend beyond simple chemical elegance. The most immediate impact is seen in the simplification of the supply chain; by reducing a multi-step synthesis to a single convergent step, the number of raw materials required is significantly decreased. This consolidation reduces the complexity of vendor management and minimizes the risk of supply disruptions associated with sourcing multiple specialized precursors. Furthermore, the starting materials—aromatic amidines and sulfur ylides—are generally commercially available or easily synthesized from commodity chemicals, ensuring a stable and reliable supply base for long-term production campaigns.
- Cost Reduction in Manufacturing: The economic argument for this technology is compelling, primarily driven by the drastic reduction in processing time and material consumption. By eliminating the need for protecting groups and avoiding stoichiometric oxidants, the process generates significantly less chemical waste, which lowers disposal costs and reduces the environmental footprint of the manufacturing site. The high atom economy of the C-H activation step means that a larger proportion of the input mass ends up in the final product, improving the overall material efficiency. Additionally, the mild reaction conditions (80°C) reduce energy consumption compared to high-temperature processes, contributing to lower utility costs per kilogram of product produced.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route translates directly into improved supply chain reliability and shorter lead times. Because the reaction tolerates a wide variety of functional groups without requiring specific protection strategies, the same core process can be applied to a broad range of analogues with minimal re-optimization. This flexibility allows manufacturers to respond quickly to changing demand for different indole derivatives without the need for extensive process development cycles. The use of standard solvents and common catalysts further ensures that the process is not dependent on exotic or hard-to-source reagents, mitigating the risk of raw material shortages.
- Scalability and Environmental Compliance: Scaling this process from gram to tonnage quantities is facilitated by the homogeneous nature of the catalytic system and the absence of hazardous high-pressure or high-temperature requirements. The reaction can be safely conducted in standard glass-lined or stainless steel reactors equipped with basic heating and stirring capabilities. From an environmental compliance standpoint, the reduction in waste generation and the avoidance of heavy metal oxidants align with increasingly stringent global regulations on pharmaceutical manufacturing. This makes the technology future-proof against tightening environmental standards, ensuring continuous operation without the need for costly retrofits or waste treatment upgrades.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel indole synthesis method. These insights are derived directly from the experimental data and technical disclosures within the patent documentation, providing a clear understanding of the process capabilities and limitations. Understanding these details is essential for technical teams evaluating the feasibility of integrating this route into their existing manufacturing workflows.
Q: What are the primary advantages of this C-H activation method over traditional Fischer synthesis?
A: Unlike Fischer synthesis which often suffers from poor regioselectivity, this method utilizes amidine as a directing group to achieve precise 3-position acylation. Furthermore, it operates under significantly milder conditions (80°C) without requiring stoichiometric oxidants or harsh bases, resulting in higher product purity and yield.
Q: Which catalysts are compatible with this synthetic route?
A: The process demonstrates excellent compatibility with pentamethylcyclopentadienyl rhodium and iridium complexes. Specifically, dimers such as [Cp*RhCl2]2 and [Cp*IrCl2]2, as well as cationic species like [Cp*Rh(MeCN)3](SbF6)2, have been proven effective in driving the cyclization efficiently.
Q: Does this method require protecting groups for sensitive functional groups?
A: No, one of the distinct commercial advantages of this technology is its exceptional functional group tolerance. The mild reaction environment allows substrates containing halogens, nitro groups, and esters to react directly without the need for tedious protection and deprotection steps, thereby shortening the overall production timeline.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Acylated Indole Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced C-H activation technologies in accelerating drug development and optimizing production costs. As a leading CDMO partner, we possess the technical expertise and infrastructure to translate complex laboratory methodologies like the one described in CN109265386B into robust, commercial-scale processes. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from discovery to market is seamless. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee the quality of every batch of high-purity pharmaceutical intermediates we deliver.
We invite you to collaborate with us to leverage this efficient synthetic route for your next project. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. Please contact our technical procurement team today to request specific COA data for our existing indole inventory or to discuss route feasibility assessments for your custom synthesis needs. Let us help you achieve your supply chain goals with precision, reliability, and cost-effectiveness.
