Advanced Calcium-Catalyzed Synthesis of Tri-Substituted 1,3-Dienes for Commercial Pharmaceutical Applications
Advanced Calcium-Catalyzed Synthesis of Tri-Substituted 1,3-Dienes for Commercial Pharmaceutical Applications
The landscape of organic synthesis for pharmaceutical intermediates is constantly evolving, driven by the urgent need for greener, more efficient, and cost-effective methodologies. A significant breakthrough in this domain is documented in patent CN109970560B, which discloses a novel preparation method for tri-substituted 1,3-diolefin compounds. These conjugated diene structures are pivotal scaffolds found in numerous natural products and biologically active compounds, such as Palmerolide A and Rapamycin, underscoring their critical importance in modern drug discovery and development. The patented technology introduces a transformative approach by utilizing allyl alcohols directly as raw materials in the presence of a calcium-based catalyst system, effectively bypassing the traditional reliance on hazardous allyl halides. This innovation not only streamlines the synthetic pathway but also aligns perfectly with the principles of green chemistry by minimizing toxic waste generation. For R&D directors and procurement specialists alike, this method represents a substantial leap forward in the reliable supply of high-purity pharmaceutical intermediates.
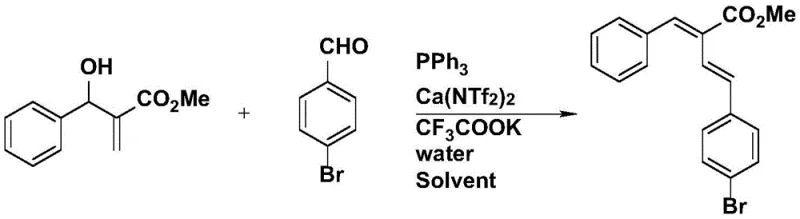
The core of this technological advancement lies in its ability to generate allyl phosphorus ylides in situ from allyl alcohols, which then undergo a Wittig-type olefination with aryl aldehydes. Traditionally, the construction of 1,3-diene frameworks has been fraught with challenges related to reagent stability, toxicity, and purification complexity. By shifting the paradigm to a calcium-catalyzed system operating in benign solvents like isopropanol, the process achieves remarkable atom economy. The reaction conditions are robust, typically proceeding at 100°C under an inert argon atmosphere, ensuring high reproducibility and safety profiles suitable for industrial scale-up. This patent provides a comprehensive solution for the commercial scale-up of complex pharmaceutical intermediates, addressing both the technical hurdles of synthesis and the economic imperatives of modern manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 1,3-diene compounds has relied heavily on two primary strategies: the classical Wittig reaction using pre-formed allyl phosphorus ylides and transition metal-catalyzed coupling reactions. The conventional preparation of allyl phosphorus ylides typically involves the reaction of allyl halides with phosphines, a process that inevitably generates stoichiometric amounts of hydrogen halides as toxic byproducts. These acidic byproducts necessitate the use of additional bases for neutralization, complicating the workup procedure and increasing the burden on waste treatment facilities. Furthermore, transition metal coupling methods, while effective, often suffer from limited substrate scope and the persistent issue of heavy metal contamination. For pharmaceutical applications, removing trace amounts of palladium or nickel to meet stringent regulatory standards requires expensive scavenging resins and extensive purification steps, drastically inflating the cost of goods sold. Additionally, traditional methods often lack the step economy required for rapid iteration in drug discovery, as they may require multi-step sequences to install the necessary functional groups prior to the diene formation.
The Novel Approach
In stark contrast, the methodology outlined in patent CN109970560B offers a streamlined, one-pot solution that directly utilizes allyl alcohols, thereby eliminating the need for pre-functionalized halides. This novel approach leverages a unique catalytic system comprising triphenylphosphine, calcium bis(trifluoromethylsulfonyl)imide, and potassium trifluoroacetate to activate the hydroxyl group and facilitate the formation of the reactive ylide species in situ. The beauty of this system is its simplicity and environmental compatibility; the only byproduct generated during the activation of the alcohol is water, which is inherently non-toxic and easy to remove. This shift from halide-based chemistry to alcohol-based chemistry represents a fundamental improvement in process safety and operational ease. Moreover, the reaction demonstrates exceptional versatility, accommodating a wide range of aryl aldehydes including those with electron-withdrawing or electron-donating substituents, as well as heterocyclic variants. This broad substrate tolerance makes it an invaluable tool for the synthesis of diverse chemical libraries required for high-throughput screening in pharmaceutical research.

Furthermore, the precursor allyl alcohols used in this process can themselves be synthesized efficiently via green methods, such as the Morita-Baylis-Hillman (MBH) reaction shown in the figure above, which utilizes DABCO as a catalyst. This creates a fully integrated, green synthetic route from simple starting materials like benzaldehyde and methyl acrylate all the way to the complex diene target. The cumulative effect of these improvements is a drastic reduction in the environmental footprint of the synthesis, coupled with a significant enhancement in overall yield and purity. For supply chain managers, this translates to a more reliable and sustainable source of critical intermediates, less susceptible to regulatory crackdowns on hazardous waste disposal.
Mechanistic Insights into Calcium-Catalyzed Olefination
The mechanistic underpinning of this reaction is a fascinating interplay between Lewis acid activation and phosphine nucleophilicity. The calcium catalyst, specifically calcium bis(trifluoromethylsulfonyl)imide (Ca(NTf2)2), acts as a potent Lewis acid that coordinates with the hydroxyl group of the allyl alcohol. This coordination significantly enhances the leaving group ability of the hydroxyl moiety, facilitating its displacement by triphenylphosphine to form the key allyl phosphonium salt intermediate. Unlike traditional methods that require harsh conditions or strong bases to generate the ylide, the presence of potassium trifluoroacetate and water in this system creates a buffered environment that promotes the deprotonation of the phosphonium salt to generate the reactive phosphorus ylide under mild thermal conditions. The generated ylide then undergoes a stereoselective [2+2] cycloaddition with the aryl aldehyde, followed by a retro-[2+2] fragmentation to yield the final 1,3-diene product with high E-selectivity. This mechanism ensures that the reaction proceeds with minimal side reactions, such as polymerization or isomerization, which are common pitfalls in diene synthesis.
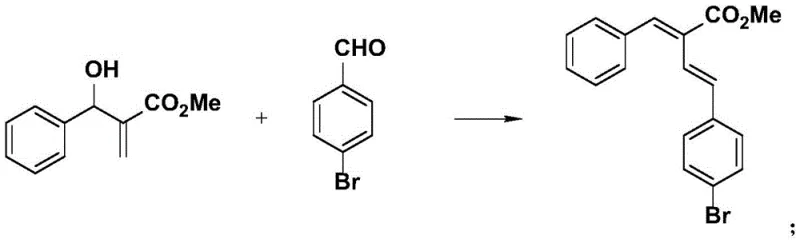
Impurity control is another critical aspect where this mechanism excels. The use of a calcium catalyst, which is an earth-abundant metal, avoids the introduction of toxic heavy metals that are difficult to purge from the final API. The reaction conditions are sufficiently mild to prevent the decomposition of sensitive functional groups often present in advanced intermediates, such as esters, nitriles, and halides. As demonstrated in the specific example above, the reaction tolerates the presence of a bromine atom on the aromatic ring, which is a valuable handle for further downstream functionalization via cross-coupling reactions. The high atom economy of the process means that fewer atoms end up as waste, simplifying the downstream purification process. Typically, the crude reaction mixture can be purified using standard silica gel chromatography with common solvent systems like petroleum ether and ethyl acetate, yielding products of high purity suitable for immediate use in subsequent synthetic steps without the need for recrystallization or specialized scavenging treatments.
How to Synthesize Tri-Substituted 1,3-Diolefin Compounds Efficiently
The practical implementation of this synthesis is designed for ease of operation, making it accessible for both laboratory-scale optimization and pilot-plant production. The protocol involves a straightforward mixing of reagents followed by a controlled heating phase, requiring no specialized equipment beyond standard glassware and heating mantles. The detailed standardized synthesis steps, including precise molar ratios and workup procedures, are provided in the guide below to ensure reproducibility and safety for technical teams adopting this methodology.
- Mix allyl alcohol, triphenylphosphine, calcium bisimide, aryl aldehyde, potassium trifluoroacetate, and water in a reaction solvent like isopropanol.
- Conduct the catalytic reaction under an argon atmosphere at 100°C with stirring for approximately 24 hours.
- Remove the reaction solvent using a vacuum rotary evaporator and purify the residue via column chromatography to obtain the target diene.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this calcium-catalyzed technology offers compelling economic and logistical benefits that extend far beyond the laboratory bench. The elimination of expensive transition metal catalysts and the associated purification infrastructure results in a leaner, more cost-efficient manufacturing process. By removing the dependency on precious metals like palladium, companies can insulate their supply chains from the volatility of commodity metal prices and the geopolitical risks associated with their sourcing. Furthermore, the simplified workup procedure, which avoids complex aqueous extractions and heavy metal scavenging, significantly reduces the consumption of solvents and consumables, leading to lower operational expenditures. The robustness of the reaction conditions also implies a higher success rate in production batches, minimizing the risk of costly batch failures and ensuring a consistent supply of high-quality intermediates to meet production schedules.
- Cost Reduction in Manufacturing: The primary driver for cost reduction in this process is the substitution of hazardous allyl halides with inexpensive and readily available allyl alcohols. This change eliminates the need for stoichiometric bases and the subsequent neutralization steps, thereby reducing the volume of chemical waste generated. Additionally, the use of calcium, an abundant and low-cost metal, replaces expensive noble metal catalysts, directly lowering the raw material cost per kilogram of product. The simplified purification process further contributes to cost savings by reducing the time and resources spent on chromatography and quality control testing for metal residues. Overall, the process delivers a substantial reduction in the cost of goods sold, enhancing the profit margins for downstream pharmaceutical products.
- Enhanced Supply Chain Reliability: The reliance on stable, non-hazardous starting materials significantly improves supply chain resilience. Allyl alcohols and aryl aldehydes are commodity chemicals with established global supply networks, reducing the risk of shortages compared to specialized organometallic reagents. The operational simplicity of the reaction, which does not require rigorous exclusion of moisture beyond a standard inert atmosphere, allows for greater flexibility in manufacturing locations and reduces the need for highly specialized containment facilities. This accessibility ensures that production can be scaled up rapidly to meet surging demand without the long lead times typically associated with setting up complex transition metal catalysis lines. Consequently, partners can rely on a more agile and responsive supply chain capable of adapting to market dynamics.
- Scalability and Environmental Compliance: From an environmental compliance perspective, this technology is a game-changer. The generation of water as the sole byproduct aligns perfectly with increasingly stringent environmental regulations regarding volatile organic compounds (VOCs) and hazardous waste disposal. The absence of toxic halogenated byproducts simplifies the permitting process for new manufacturing facilities and reduces the liability associated with waste management. The process has already been demonstrated to be scalable to gram levels with high efficiency, indicating a clear path toward multi-kilogram and ton-scale production. This scalability, combined with its green profile, makes it an ideal candidate for sustainable manufacturing initiatives, helping companies meet their corporate social responsibility goals while maintaining operational excellence.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this calcium-catalyzed synthesis. These answers are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a reliable basis for decision-making.
Q: What are the advantages of using calcium catalysts over transition metals for diene synthesis?
A: Calcium catalysts eliminate the need for expensive and toxic transition metals like palladium or nickel, significantly reducing heavy metal residue concerns in pharmaceutical intermediates and lowering purification costs.
Q: Can this method accommodate diverse substrate scopes for drug discovery?
A: Yes, the protocol demonstrates excellent tolerance for various substituted phenyl groups, heterocycles, and alkyl chains on the allyl alcohol, making it highly versatile for synthesizing complex bioactive molecules.
Q: How does this process improve environmental sustainability compared to traditional Wittig reactions?
A: By using allyl alcohols directly instead of allyl halides, the reaction produces water as the only byproduct, avoiding the generation of toxic hydrogen halides and improving overall atom economy.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tri-Substituted 1,3-Diene Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the technologies described in patent CN109970560B for the next generation of pharmaceutical intermediates. As a leading CDMO partner, we possess the technical expertise and infrastructure to translate these innovative laboratory protocols into robust, commercial-scale manufacturing processes. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with unwavering consistency. We operate state-of-the-art rigorous QC labs that enforce stringent purity specifications, guaranteeing that every batch of tri-substituted 1,3-diene delivered meets the highest industry standards for impurity profiles and residual solvent content.
We invite you to collaborate with us to leverage this green chemistry advantage for your upcoming projects. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific molecule, demonstrating exactly how this calcium-catalyzed route can optimize your budget. Please contact us today to request specific COA data for our reference standards and to discuss route feasibility assessments for your target compounds. Let us be your partner in driving efficiency and sustainability in your chemical supply chain.
