Advanced Photocatalytic Synthesis of Alpha Alpha Difluoro Beta Carbonyl Sulfone Intermediates for Commercial Scale
Advanced Photocatalytic Synthesis of Alpha Alpha Difluoro Beta Carbonyl Sulfone Intermediates for Commercial Scale
The rapid evolution of fluorine chemistry has become a cornerstone in modern medicinal chemistry, driven by the unique ability of fluorine atoms to modulate the metabolic stability and oral bioavailability of bioactive molecules. A pivotal development in this field is detailed in patent CN111269153B, which discloses a groundbreaking synthetic method for α,α-difluoro-β-carbonyl sulfone compounds. These structural motifs are increasingly recognized as valuable pharmacophores, serving as robust surrogates for hydroxyl groups while offering superior resistance to metabolic degradation. The disclosed technology represents a significant paradigm shift from classical stoichiometric approaches to a catalytic, visible-light-driven process. By leveraging the power of photoredox catalysis, this method enables the construction of complex fluorinated scaffolds under exceptionally mild conditions, addressing long-standing challenges in the synthesis of sulfur-containing fluorinated intermediates. For R&D teams and process chemists, this innovation offers a streamlined pathway to access high-value building blocks that were previously difficult to manufacture efficiently.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to this technological breakthrough, the synthesis of α,α-difluoro-β-carbonyl sulfone compounds was fraught with significant operational hurdles and safety concerns. Historical precedents, such as the methodology reported by the Hu group in 2009, relied heavily on the use of methyl benzoate and difluoromethyl sulfone precursors. Critically, these traditional routes necessitated the use of extremely strong bases, specifically lithium hexamethyldisilazide (LHMDS), to drive the deprotonation steps required for carbon-sulfur bond formation. Furthermore, these reactions had to be conducted at cryogenic temperatures, often as low as -98°C, to control reactivity and prevent decomposition. Such苛刻 conditions impose a massive burden on manufacturing infrastructure, requiring specialized low-temperature reactors and extensive energy consumption for cooling. Additionally, the handling of strong bases and the limited functional group tolerance associated with these harsh conditions restricted the scope of accessible derivatives, making the scale-up of these intermediates economically and technically prohibitive for many fine chemical applications.
The Novel Approach
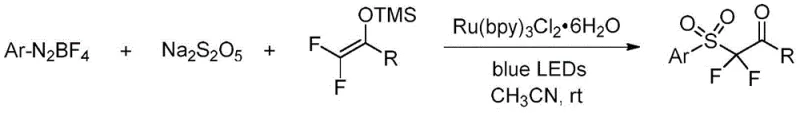
In stark contrast to the legacy methods, the novel photocatalytic strategy outlined in the patent data utilizes a radical-based mechanism that operates seamlessly at room temperature. The core of this innovation lies in the in situ generation of aryl sulfonyl radicals from readily available aryl diazonium salts and sodium metabisulfite (Na2S2O5). Under the irradiation of blue LEDs and in the presence of a ruthenium photosensitizer, these precursors undergo a smooth transformation to generate the reactive radical species without the need for external oxidants or strong bases. This radical intermediate then adds efficiently to 2,2-difluoro enol silyl ethers, followed by a single-electron oxidation step to furnish the target α,α-difluoro-β-carbonyl sulfone products. This approach not only bypasses the need for cryogenic cooling but also utilizes solid sulfur dioxide substitutes, which are safer and easier to handle than gaseous SO2. The result is a robust, scalable, and environmentally friendlier protocol that dramatically lowers the barrier to entry for producing these high-value intermediates.
Mechanistic Insights into Visible-Light Photoredox Catalysis
The mechanistic elegance of this transformation relies on the precise interplay between the photosensitizer and the radical precursors. Upon absorption of blue light photons, the ruthenium catalyst, specifically Ru(bpy)3Cl2·6H2O, transitions to an excited state capable of engaging in single-electron transfer (SET) processes. This excited species facilitates the reduction of the aryl diazonium salt or the activation of the sulfur source, leading to the homolytic cleavage that generates the crucial aryl sulfonyl radical. This radical species is highly electrophilic and adds rapidly to the electron-rich double bond of the 2,2-difluoro enol silyl ether. The resulting carbon-centered radical intermediate is then oxidized by the oxidized form of the photocatalyst, regenerating the ground-state catalyst and completing the catalytic cycle. This redox-neutral or oxidative quenching cycle ensures that the reaction proceeds with high atom economy and minimal waste generation. Understanding this mechanism is vital for process optimization, as it highlights the importance of maintaining an oxygen-free environment to prevent radical quenching by atmospheric oxygen, which could otherwise lead to side products and reduced yields.
From an impurity control perspective, the mildness of the reaction conditions plays a pivotal role in ensuring product purity. Traditional strong-base mediated reactions often suffer from competing elimination reactions or nucleophilic attacks on sensitive functional groups, leading to complex impurity profiles that are difficult to separate. In this photocatalytic system, the neutrality of the reaction medium preserves sensitive functionalities such as esters, halides, and heterocycles, which are common in pharmaceutical intermediates. The use of sodium metabisulfite as a solid SO2 surrogate further minimizes the risk of over-sulfonylation or the formation of sulfonic acid byproducts that can occur with gaseous SO2. Consequently, the crude reaction mixtures obtained from this process are typically cleaner, reducing the burden on downstream purification steps like column chromatography or crystallization. This inherent selectivity is a key advantage for manufacturing high-purity materials required for clinical trials and eventual commercial drug production.
How to Synthesize Alpha,Alpha-Difluoro-Beta-Carbonyl Sulfone Efficiently
The practical implementation of this synthesis is designed for ease of operation, making it highly attractive for both laboratory discovery and pilot-scale production. The procedure involves a straightforward one-pot setup where the aryl diazonium salt, sodium metabisulfite, and the fluorinated enol silyl ether are combined in acetonitrile. The addition of a catalytic amount of the ruthenium complex initiates the process upon exposure to standard blue LED arrays. The reaction progress can be conveniently monitored via thin-layer chromatography (TLC), and upon completion, the workup involves simple concentration and purification. This simplicity stands in contrast to multi-step sequences often required for similar transformations. For detailed operational parameters, stoichiometry, and specific purification protocols, please refer to the standardized synthesis guide below which encapsulates the critical process parameters derived from the patent examples.
- Charge a reaction vessel with aryl diazonium salt, sodium metabisulfite, 2,2-difluoro enol silyl ether, and Ru(bpy)3Cl2·6H2O catalyst under inert atmosphere.
- Add acetonitrile solvent and irradiate the mixture with 15W blue LEDs at room temperature while stirring until reaction completion.
- Concentrate the reaction mixture under reduced pressure and purify the crude product via column chromatography using petroleum ether and ethyl acetate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this photocatalytic technology translates into tangible strategic benefits that extend beyond mere chemical novelty. The shift from cryogenic, base-mediated chemistry to ambient temperature photocatalysis fundamentally alters the cost structure of manufacturing these specialized intermediates. By eliminating the requirement for ultra-low temperature reactors and the associated energy costs for cooling, the overall utility consumption is drastically reduced. Furthermore, the replacement of hazardous strong bases with benign inorganic salts like sodium metabisulfite simplifies safety compliance and waste disposal protocols. These factors collectively contribute to a more sustainable and cost-effective manufacturing process, allowing for competitive pricing in the supply of these critical building blocks to the pharmaceutical industry.
- Cost Reduction in Manufacturing: The economic impact of this new method is profound, primarily driven by the simplification of the reaction infrastructure. Traditional methods requiring -98°C conditions demand specialized equipment and high energy inputs for refrigeration, which significantly inflate the cost of goods sold. By operating at room temperature, this process removes those capital and operational expenditures entirely. Additionally, the use of commercially available and inexpensive starting materials, such as aryl diazonium salts and sodium metabisulfite, ensures that raw material costs remain stable and predictable. The high efficiency of the photocatalyst, used in minute quantities, further minimizes reagent costs, enabling substantial cost savings in fine chemical manufacturing without compromising on yield or quality.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of robust and widely available reagents. Sodium metabisulfite is a commodity chemical with a stable global supply, unlike some specialized organometallic reagents that may face sourcing bottlenecks. The tolerance of the reaction to various functional groups means that a single platform technology can be applied to synthesize a wide array of derivatives, reducing the need for multiple distinct synthetic routes. This versatility allows suppliers to respond more agilely to changing demands from R&D departments. Moreover, the mild conditions reduce the risk of batch failures due to temperature excursions or moisture sensitivity, leading to more consistent delivery schedules and higher reliability for downstream customers relying on these intermediates for their own synthesis campaigns.
- Scalability and Environmental Compliance: Scaling photochemical reactions has historically been a challenge, but advancements in LED technology and flow chemistry have made this increasingly viable. The absence of hazardous gases like sulfur dioxide and the use of solid substitutes align perfectly with modern green chemistry principles and stringent environmental regulations. This facilitates easier permitting and faster scale-up from kilogram to tonne scales. The simplified workup procedure, which avoids complex aqueous quenches of strong bases, reduces the volume of wastewater generated. Consequently, this method supports the commercial scale-up of complex sulfone derivatives while maintaining a low environmental footprint, a critical factor for companies aiming to meet sustainability goals and regulatory standards in the production of pharmaceutical intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this photocatalytic synthesis. These insights are derived directly from the experimental data and scope defined in the patent literature, providing clarity on the practical aspects of adopting this technology. Understanding these nuances helps stakeholders make informed decisions about integrating this method into their existing supply chains and R&D pipelines. The answers reflect the balance between scientific rigor and practical manufacturability that defines this innovative approach.
Q: What are the key advantages of this photocatalytic method over traditional cryogenic synthesis?
A: Unlike traditional methods requiring strong bases like LHMDS and ultra-low temperatures of -98°C, this novel protocol operates at room temperature using visible light. This eliminates the need for expensive cryogenic infrastructure and hazardous strong bases, significantly simplifying the operational workflow and enhancing safety profiles for industrial manufacturing.
Q: What is the substrate scope for the aryl diazonium salts in this reaction?
A: The method demonstrates excellent functional group tolerance, accommodating both electron-withdrawing groups such as halogens and trifluoromethyl groups, as well as electron-donating alkyl or alkoxy substituents. This broad compatibility allows for the efficient synthesis of a diverse library of fluorine-containing sulfone derivatives essential for medicinal chemistry optimization.
Q: Is the ruthenium catalyst essential, or can it be substituted?
A: While Ru(bpy)3Cl2·6H2O is the preferred photosensitizer yielding optimal results, the patent indicates that alternatives like Ir(ppy)3 or Eosin-Y can be used. However, substituting the catalyst may lead to a reduction in reaction yield, so the ruthenium complex is recommended for maximizing efficiency in commercial production settings.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alpha,Alpha-Difluoro-Beta-Carbonyl Sulfone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this visible-light mediated synthesis in accelerating the development of next-generation therapeutics. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop discovery to full-scale manufacturing is seamless. Our facilities are equipped with state-of-the-art photochemical reactors and rigorous QC labs capable of meeting stringent purity specifications required for GMP environments. We understand that the introduction of fluorine and sulfur motifs requires precise control, and our technical team is adept at optimizing these photocatalytic parameters to maximize yield and minimize impurities, delivering high-purity fluorine-containing intermediates that meet the exacting standards of the global pharmaceutical industry.
We invite you to collaborate with us to leverage this advanced technology for your specific project needs. Whether you require custom synthesis of novel analogs or reliable supply of established intermediates, our team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. By partnering with us, you gain access to a supply chain that is not only cost-effective and reliable but also at the forefront of sustainable chemical innovation, ensuring your projects remain competitive and compliant in a rapidly evolving market landscape.
