Scalable Synthesis of Novel Thiouracil-Triazolopyrimidine Antibacterial Intermediates
Scalable Synthesis of Novel Thiouracil-Triazolopyrimidine Antibacterial Intermediates
In the face of escalating global bacterial resistance, the pharmaceutical industry urgently requires novel scaffolds that operate through unique mechanisms of action. Patent CN111518104B introduces a groundbreaking class of thiouracil-containing 1,2,4-triazolo[1,5-a]pyrimidine compounds, specifically engineered to target the bacterial SecA protein. This innovation represents a significant leap forward in antibacterial drug discovery, offering a robust solution for developing next-generation therapeutics. The structural novelty of these compounds lies in the strategic splicing of two potent pharmacophores: the thiouracil moiety and the fused triazolopyrimidine heterocycle. ![General chemical structure of thiouracil-containing 1,2,4-triazolo[1,5-a]pyrimidine compounds showing variable substituents R1 and R2](/insights/img/thiouracil-triazolopyrimidine-synthesis-pharma-supplier-20260309045236-01.png) As a reliable pharmaceutical intermediate supplier, we recognize that the true value of such a patent lies not just in the molecule itself, but in the feasibility of its manufacture. The disclosed synthesis pathway offers a streamlined, cost-effective route that bypasses the complexities often associated with fused heterocyclic systems, ensuring a stable supply chain for R&D teams aiming to combat superbugs.
As a reliable pharmaceutical intermediate supplier, we recognize that the true value of such a patent lies not just in the molecule itself, but in the feasibility of its manufacture. The disclosed synthesis pathway offers a streamlined, cost-effective route that bypasses the complexities often associated with fused heterocyclic systems, ensuring a stable supply chain for R&D teams aiming to combat superbugs.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional approaches to synthesizing complex nitrogen-rich heterocycles often suffer from multi-step inefficiencies, harsh reaction conditions, and the reliance on scarce or toxic reagents. Conventional methods for constructing triazolopyrimidine cores frequently involve prolonged heating, difficult purification processes due to similar byproduct profiles, and the use of heavy metal catalysts that necessitate rigorous and expensive removal steps to meet regulatory standards for API manufacturing. Furthermore, linking distinct pharmacophores like thiouracil to these cores typically requires protecting group strategies that add unnecessary steps, reduce overall yield, and increase waste generation. These bottlenecks significantly inflate the cost of goods sold (COGS) and extend lead times, creating substantial friction for procurement managers seeking cost reduction in API manufacturing. The inability to easily scale these traditional routes often results in supply discontinuity, posing a risk to long-term drug development projects.
The Novel Approach
The methodology outlined in CN111518104B revolutionizes this landscape through a convergent synthesis strategy that prioritizes operational simplicity and atom economy. By employing an "active substructure splicing" concept, the inventors have devised a route where the thiouracil and triazolopyrimidine fragments are constructed independently and then coupled efficiently via a thioether linkage. 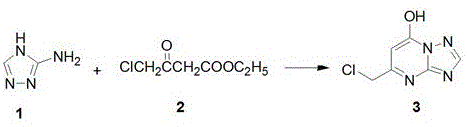 This approach eliminates the need for complex protecting groups and allows for the use of commodity chemicals such as substituted anilines and benzaldehydes as starting materials. The reaction conditions are remarkably mild, utilizing standard solvents like glacial acetic acid and acetonitrile, and common bases like potassium carbonate. This simplicity translates directly into enhanced process robustness, making the commercial scale-up of complex pharmaceutical intermediates far more achievable. For supply chain heads, this means a process that is not only easier to validate but also inherently safer and more environmentally compliant, reducing the burden of waste treatment and regulatory oversight.
This approach eliminates the need for complex protecting groups and allows for the use of commodity chemicals such as substituted anilines and benzaldehydes as starting materials. The reaction conditions are remarkably mild, utilizing standard solvents like glacial acetic acid and acetonitrile, and common bases like potassium carbonate. This simplicity translates directly into enhanced process robustness, making the commercial scale-up of complex pharmaceutical intermediates far more achievable. For supply chain heads, this means a process that is not only easier to validate but also inherently safer and more environmentally compliant, reducing the burden of waste treatment and regulatory oversight.
Mechanistic Insights into Convergent Heterocyclic Assembly
The synthetic elegance of this patent lies in its stepwise construction of the two key intermediates before the final coupling. The formation of the triazolopyrimidine core begins with the condensation of 3-amino-1,2,4-triazole and ethyl 4-chloroacetoacetate in glacial acetic acid. This cyclization is driven by the nucleophilic attack of the amino group on the ketone carbonyl, followed by intramolecular ring closure to form the pyrimidine ring fused to the triazole. Subsequent chlorination with phosphorus oxychloride (POCl3) activates the 7-position of the heterocycle, converting the hydroxyl group into a good leaving group (chloride), which is crucial for the subsequent nucleophilic aromatic substitution. This activation step is critical for ensuring high reactivity in the following amination step, where various substituted anilines are introduced to diversify the R1 position, allowing for extensive SAR (Structure-Activity Relationship) exploration without altering the core synthetic logic.
Parallel to this, the thiouracil fragment (Intermediate 6) is generated via a classic Knoevenagel-type condensation involving substituted benzaldehyde, ethyl cyanoacetate, and thiourea, catalyzed by piperidine. This one-pot reaction efficiently constructs the pyrimidine-2-thione ring with high regioselectivity. The final coupling step exploits the nucleophilicity of the sulfur atom in the thiouracil intermediate. Under basic conditions provided by potassium carbonate in acetonitrile, the thiolate anion attacks the chloromethyl group on the triazolopyrimidine intermediate (Intermediate 5). This SN2-type substitution forms the stable thioether bridge that links the two pharmacophores. The use of mild base and polar aprotic solvent ensures that side reactions are minimized, leading to a cleaner crude product profile. This mechanistic clarity allows process chemists to precisely control impurity levels, ensuring that the final high-purity pharmaceutical intermediates meet the stringent specifications required for preclinical and clinical evaluation.
How to Synthesize Thiouracil-Triazolopyrimidine Efficiently
The patented procedure provides a detailed, reproducible protocol for generating these high-value antibacterial agents. The process is divided into five distinct operational stages, each optimized for yield and purity. The initial steps focus on building the heterocyclic cores, while the final stages involve the critical coupling and purification. The use of standard laboratory equipment such as round-bottom flasks, reflux condensers, and rotary evaporators indicates that this chemistry is not restricted to specialized facilities, enhancing its accessibility for contract research organizations and manufacturing partners. For a comprehensive breakdown of the specific molar ratios, temperature profiles, and workup procedures, please refer to the standardized guide below.
- Condense 3-amino-1,2,4-triazole with ethyl 4-chloroacetoacetate in glacial acetic acid under reflux to form Intermediate 3.
- React Intermediate 3 with phosphorus oxychloride (POCl3) under reflux to chlorinate the hydroxyl group, yielding Intermediate 4.
- Perform nucleophilic substitution on Intermediate 4 with substituted anilines in isopropylamine to generate Intermediate 5.
- Synthesize Intermediate 6 via piperidine-catalyzed condensation of substituted benzaldehyde, ethyl cyanoacetate, and thiourea.
- Couple Intermediate 5 and Intermediate 6 using potassium carbonate in acetonitrile under reflux to obtain the final target compound.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this synthetic route offers profound advantages for stakeholders focused on cost efficiency and supply security. The elimination of transition metal catalysts is a primary driver for cost reduction, as it removes the need for expensive scavengers and extensive analytical testing for residual metals, which are major cost centers in modern API production. Furthermore, the reliance on bulk commodity chemicals like anilines and benzaldehydes ensures that raw material costs remain stable and predictable, shielding the project from the volatility often seen with specialty reagents. The simplicity of the workup procedures, which primarily involve filtration and recrystallization rather than complex chromatography, significantly reduces solvent consumption and processing time. This operational efficiency translates into substantial cost savings in manufacturing, allowing for more competitive pricing of the final active pharmaceutical ingredient.
Supply chain reliability is further bolstered by the robustness of the reaction conditions. The use of reflux in common solvents like acetic acid and acetonitrile implies that the process can be easily transferred from laboratory glassware to industrial stainless steel reactors without significant re-engineering. This scalability reduces the lead time for high-purity pharmaceutical intermediates, enabling faster progression from gram-scale screening to kilogram-scale pilot runs. Additionally, the modular nature of the synthesis, where different R1 and R2 groups can be introduced late in the sequence, allows for flexible production scheduling. Manufacturers can produce large batches of the common intermediates and then customize the final step based on specific customer demands, optimizing inventory management and reducing the risk of obsolescence. This flexibility is crucial for maintaining supply continuity in a dynamic market environment.
Environmental compliance and safety are also significantly enhanced by this green chemistry approach. The avoidance of hazardous reagents and the minimization of waste streams align with increasingly strict global environmental regulations. The process generates fewer byproducts, simplifying waste treatment and reducing the environmental footprint of the manufacturing facility. This not only mitigates regulatory risk but also enhances the corporate social responsibility profile of the supply chain. For procurement managers, partnering with a supplier who utilizes such efficient and compliant processes ensures a sustainable source of materials that will not be disrupted by environmental enforcement actions. The overall simplicity and safety of the protocol make it an ideal candidate for continuous manufacturing technologies, which could further drive down costs and improve quality consistency in the future.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these novel compounds. The answers are derived directly from the technical specifications and beneficial effects described in the patent documentation, providing clarity on the feasibility and potential of this technology. Understanding these details is essential for making informed decisions about integrating this scaffold into your drug discovery pipeline.
Q: What is the primary biological target of these thiouracil-triazolopyrimidine compounds?
A: These compounds are designed to inhibit the SecA protein in bacteria, a novel target for overcoming antibiotic resistance, particularly against strains like Staphylococcus aureus and Bacillus subtilis.
Q: Does this synthesis method require expensive transition metal catalysts?
A: No, the patented process utilizes common organic bases like piperidine and potassium carbonate, avoiding costly transition metals and simplifying downstream purification and heavy metal removal.
Q: Is this synthetic route suitable for large-scale industrial production?
A: Yes, the patent explicitly states the method involves simple operations, readily available raw materials, and standard reflux conditions, making it highly amenable to commercial scale-up from kilograms to tons.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Thiouracil-Triazolopyrimidine Supplier
The technological potential of the thiouracil-containing 1,2,4-triazolo[1,5-a]pyrimidine scaffold is immense, offering a viable path toward overcoming multidrug-resistant bacterial infections. At NINGBO INNO PHARMCHEM, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop discovery to market-ready therapy is seamless. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of intermediate delivered meets the highest international standards. We understand the critical nature of antibacterial development and are committed to providing a supply chain that is both resilient and responsive to your evolving needs.
We invite you to collaborate with us to leverage this innovative synthetic route for your next-generation antibiotic programs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how our optimized processes can enhance your project's economic viability. Please contact our technical procurement team today to request specific COA data for our available analogues and to discuss route feasibility assessments for your custom targets. Let us be your partner in turning this promising scientific breakthrough into a life-saving reality.
