Advanced Manganese-Catalyzed Synthesis of Polysubstituted Alkenyl Amidines for Commercial Scale-up
Advanced Manganese-Catalyzed Synthesis of Polysubstituted Alkenyl Amidines for Commercial Scale-up
The landscape of organic synthesis is continually evolving towards more sustainable and economically viable pathways, particularly for high-value structural motifs found in pharmaceutical agents. A significant breakthrough in this domain is documented in Chinese Patent CN109796372B, which discloses a novel method for preparing polysubstituted alkenyl amidines. This technology leverages a manganese-catalyzed C-H activation strategy to directly couple amidines with alkynes, bypassing the need for pre-functionalized precursors. For R&D directors and procurement specialists seeking reliable pharmaceutical intermediate suppliers, this patent represents a pivotal shift towards more efficient manufacturing protocols. The ability to construct complex alkenyl amidine scaffolds in a single operational step using earth-abundant manganese catalysts addresses critical pain points regarding cost, step economy, and environmental impact in fine chemical production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of vicinal polysubstituted alkenyl amidines has been fraught with synthetic challenges that hinder large-scale commercialization. Traditional routes typically rely on the preparation of ortho-cyano-substituted 1,2-disubstituted ethylenes or related complex derivatives as starting materials. The preparation of these specific precursors is often不完善 (imperfect) and involves multiple synthetic steps, leading to accumulated yield losses and increased waste generation. Furthermore, the reliance on such specialized starting materials severely limits the diversity of substrates that can be accessed, restricting the chemical space available for medicinal chemistry optimization. The繁琐 (complicated) nature of these legacy processes translates directly into higher manufacturing costs and longer lead times, creating bottlenecks for supply chain heads who require consistent and scalable sources of high-purity intermediates.
The Novel Approach
In stark contrast, the methodology outlined in CN109796372B introduces a streamlined, one-step synthesis that directly utilizes readily available amidines and alkynes. By employing a manganese catalyst in the presence of a Grignard reagent, this process achieves direct C-H activation and subsequent C-C bond formation. This approach eliminates the need for pre-installed leaving groups or complex precursor synthesis, drastically simplifying the overall workflow. The reaction conditions are robust, utilizing common solvents like 1,4-dioxane or tetrahydrofuran, and tolerate a wide range of functional groups. This innovation not only enhances the atom economy of the transformation but also opens up new avenues for structural diversification, allowing for the rapid generation of libraries of polysubstituted alkenyl amidines essential for drug discovery programs.
Mechanistic Insights into Manganese-Catalyzed C-H Activation
The core of this technological advancement lies in the unique reactivity of the manganese catalyst, specifically complexes such as pentacarbonyl manganese bromide (MnBr(CO)5) or decacarbonyl dimanganese (Mn2(CO)10). Unlike traditional noble metal catalysts, manganese offers a distinct electronic environment that facilitates the activation of inert C-H bonds under relatively mild thermal conditions. The mechanism likely involves the coordination of the amidine nitrogen to the manganese center, directing the metal to the ortho-position of the aromatic ring. Subsequent insertion of the alkyne into the Mn-C bond, followed by reductive elimination or protonolysis, yields the desired alkenyl amidine product. The presence of the Grignard reagent acts as a crucial promoter, potentially assisting in the generation of the active catalytic species or facilitating the turnover of the catalytic cycle.
Understanding the impurity profile is critical for R&D teams focused on purity specifications. The high selectivity of this manganese-catalyzed system minimizes the formation of side products often associated with radical pathways or non-selective coupling. The reaction demonstrates excellent functional group tolerance, accommodating electron-donating groups like methyl and methoxy, as well as electron-withdrawing groups such as trifluoromethyl and halogens on the aromatic rings. This tolerance ensures that the final crude product contains fewer difficult-to-remove impurities, simplifying downstream purification processes like column chromatography or crystallization. Consequently, this leads to higher overall recovery rates and ensures that the final high-purity pharmaceutical intermediates meet stringent quality control standards required for clinical applications.
How to Synthesize Polysubstituted Alkenyl Amidine Efficiently
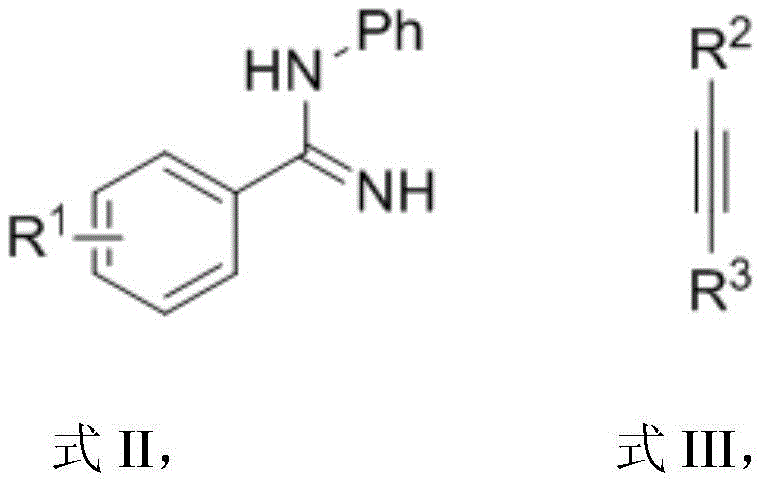
Implementing this synthesis route requires careful attention to reaction parameters to maximize yield and reproducibility. The patent details a standardized protocol where the amidine substrate and alkyne are combined in a specific molar ratio, typically around 1:2.5, to drive the reaction to completion. The choice of solvent and the precise loading of the manganese catalyst are pivotal factors that influence the reaction kinetics. Detailed below is the procedural framework derived from the patent examples, which serves as a foundation for process optimization and scale-up activities.
- Prepare the reaction system under inert atmosphere using a manganese catalyst such as MnBr(CO)5 and a Grignard reagent.
- Combine the amidine substrate (Formula II) and alkyne (Formula III) in a solvent like 1,4-dioxane at a molar ratio of approximately 1: 2.5.
- Heat the mixture to 120°C for 12 hours to facilitate C-H activation and C-C bond formation, followed by standard workup and purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the transition to this manganese-catalyzed process offers tangible strategic benefits beyond mere technical novelty. The primary advantage stems from the substitution of expensive precious metal catalysts with abundant manganese salts. This shift fundamentally alters the cost structure of the manufacturing process, removing the volatility associated with rhodium, palladium, or ruthenium pricing. Additionally, the simplification of the synthetic route from multi-step to one-step significantly reduces labor costs, energy consumption, and solvent usage. These factors collectively contribute to substantial cost savings in pharmaceutical intermediate manufacturing, allowing for more competitive pricing models without compromising on quality or supply reliability.
- Cost Reduction in Manufacturing: The elimination of costly precious metal catalysts and the reduction in synthetic steps directly lower the Bill of Materials (BOM). By avoiding the synthesis of complex ortho-cyano precursors, manufacturers save on reagents and processing time. The use of commercially available alkynes and amidines further stabilizes the supply chain against raw material fluctuations. This economic efficiency allows for the production of high-purity intermediates at a fraction of the cost of traditional methods, providing a significant margin advantage for downstream API production.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions, operating effectively at temperatures around 120°C in common solvents, ensures high process reliability. The broad substrate scope means that supply chains are not dependent on niche, hard-to-source precursors. Instead, the process utilizes commodity chemicals that are widely available from multiple global vendors. This diversification of raw material sources mitigates the risk of supply disruptions, ensuring consistent delivery schedules for critical pharmaceutical building blocks and reducing lead time for high-purity intermediates.
- Scalability and Environmental Compliance: The use of manganese, an environmentally benign metal, aligns with increasing regulatory pressures for green chemistry. The simplified workflow generates less chemical waste compared to multi-step alternatives, easing the burden on waste treatment facilities. Furthermore, the reaction's tolerance to various functional groups suggests that it can be scaled from laboratory benchtop (100 kgs) to full commercial production (100 MT) with minimal re-optimization. This scalability ensures that the technology can meet growing market demands for complex organic molecules while maintaining strict environmental compliance standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this manganese-catalyzed synthesis. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity for stakeholders evaluating this technology for their supply chains.
Q: What are the advantages of using manganese catalysts over precious metals for amidine synthesis?
A: Manganese catalysts, such as MnBr(CO)5, offer a cost-effective and earth-abundant alternative to expensive precious metals like palladium or ruthenium, significantly reducing raw material costs while maintaining high catalytic activity for C-H activation.
Q: What is the substrate scope for this alkenyl amidine synthesis method?
A: The method demonstrates broad substrate tolerance, accommodating various substituents on the benzene ring including alkyl, alkoxy, halogen, and trifluoromethyl groups, as well as heteroaryl alkynes, making it versatile for diverse pharmaceutical intermediate production.
Q: How does this method improve upon traditional synthesis routes for alkenyl amidines?
A: Unlike conventional methods that require complex multi-step preparations of ortho-cyano-1,2-disubstituted ethylene precursors, this direct C-H activation approach simplifies the process to a one-step coupling, enhancing efficiency and reducing waste.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Polysubstituted Alkenyl Amidine Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the manganese-catalyzed C-H activation technology described in CN109796372B. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped to handle air-sensitive reactions and utilize stringent purity specifications to ensure that every batch of polysubstituted alkenyl amidine meets the rigorous demands of the global pharmaceutical industry. Our rigorous QC labs employ advanced analytical techniques to verify structural integrity and impurity profiles, guaranteeing the quality necessary for your drug development pipelines.
We invite you to collaborate with us to leverage this cost-effective and efficient synthesis route for your specific projects. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your target molecule. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring that your supply chain is optimized for both performance and profitability in the competitive landscape of fine chemical manufacturing.
