Advanced Cobalt-Catalyzed Synthesis of 3-Thio-1-Glycals for Scalable Pharmaceutical Manufacturing
Introduction to Next-Generation Thiosugar Synthesis
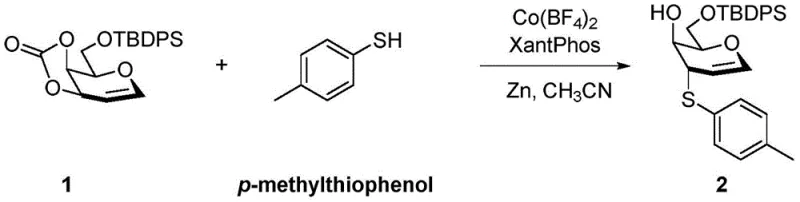
The landscape of carbohydrate chemistry is undergoing a significant transformation driven by the demand for efficient, scalable access to specialized glycosyl donors. Patent CN111995638A introduces a groundbreaking methodology for the synthesis of 3-thio-1-ene sugar compounds, addressing long-standing challenges in regioselectivity and catalyst cost. Unlike traditional approaches that rely on scarce precious metals, this innovation leverages an inexpensive cobalt-based catalytic system to achieve exceptional yields and stereocontrol. For R&D directors and procurement specialists in the pharmaceutical sector, this represents a pivotal shift towards more sustainable and economically viable manufacturing processes for complex carbohydrate intermediates. The technology specifically targets the synthesis of 3-thio-1-glycals, which serve as critical building blocks for bioactive molecules including anti-rheumatic agents and potential anti-tumor compounds.
This novel synthetic route utilizes a synergistic combination of a cobalt salt, a bidentate phosphine ligand, and zinc powder to facilitate the thioglycosylation reaction. The process operates under relatively mild thermal conditions in acetonitrile, demonstrating remarkable tolerance for various thiol and phenol substrates. By replacing expensive noble metals with earth-abundant cobalt, the method not only reduces raw material costs but also simplifies the regulatory burden associated with heavy metal residues in final drug substances. As a reliable pharmaceutical intermediate supplier, understanding these mechanistic advancements is crucial for optimizing supply chains and ensuring the continuous availability of high-purity building blocks for drug discovery programs.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 3-thio-1-enose compounds has been fraught with significant technical and economic hurdles. Conventional methodologies often depend on the use of rare and expensive transition metals such as gold or samarium, which drastically inflate the cost of goods sold (COGS) for large-scale production. Furthermore, these traditional routes frequently suffer from poor regioselectivity, leading to complex mixtures of 1-thio and 3-thio isomers that require tedious and yield-eroding separation processes. The reliance on precious metal catalysts also introduces stringent purification requirements to meet pharmaceutical standards for residual metals, adding multiple processing steps and extending lead times. Additionally, many existing protocols exhibit limited substrate scope, failing to accommodate diverse functional groups without compromising reaction efficiency or stereoselectivity.
The Novel Approach
In stark contrast, the method disclosed in CN111995638A offers a robust solution by employing a cheap cobalt catalyst paired with a specialized ligand system. This approach achieves high regioselectivity and stereoselectivity, effectively suppressing the formation of unwanted isomers and streamlining the purification workflow. The introduction of zinc powder as a promoter further enhances the catalytic turnover, driving reactions to near-completion with impressive isolated yields. This strategy not only mitigates the cost volatility associated with precious metals but also aligns with green chemistry principles by utilizing more abundant materials. The versatility of this system allows for the efficient incorporation of various thiol and phenol derivatives, expanding the chemical space accessible for medicinal chemistry campaigns.

Mechanistic Insights into Cobalt-Catalyzed Thioglycosylation
The core of this technological breakthrough lies in the precise orchestration of the cobalt catalytic cycle. The reaction initiates with the activation of the cobalt precursor, typically Co(BF4)2·6H2O, by the Xantphos ligand to form a highly active catalytic species. This complex facilitates the oxidative addition or coordination with the glycal substrate, creating a reactive intermediate poised for nucleophilic attack. The presence of zinc powder is mechanistically critical; it serves to regenerate the active low-valent cobalt species, ensuring the catalytic cycle proceeds smoothly without deactivation. This redox mediation prevents the accumulation of inactive cobalt species, thereby maintaining high turnover numbers throughout the reaction duration. The specific geometry imposed by the bulky Xantphos ligand also plays a pivotal role in directing the incoming thiol nucleophile to the C3 position, enforcing the observed high regioselectivity.
From an impurity control perspective, this mechanism offers distinct advantages over radical-based or Lewis acid-mediated pathways. The concerted nature of the cobalt-catalyzed process minimizes side reactions such as polymerization or non-selective addition across the double bond. The result is a cleaner reaction profile with fewer byproducts, which translates directly to higher purity in the crude product. This inherent selectivity reduces the load on downstream purification units, such as column chromatography or crystallization, allowing for more efficient resource utilization. For process chemists, understanding this mechanism provides a roadmap for further optimization, such as tuning the electronic properties of the ligand to accommodate even more challenging substrates while maintaining the rigorous quality standards required for GMP manufacturing.
How to Synthesize 3-Thio-1-Glycals Efficiently
The practical implementation of this synthesis involves a straightforward protocol that balances reaction efficiency with operational simplicity. The process begins by charging a reaction vessel with the cobalt catalyst, the Xantphos ligand, and activated zinc powder under an inert atmosphere to prevent oxidation. The glycal substrate and the chosen thiol or phenol are then introduced in acetonitrile, and the mixture is heated to temperatures between 100°C and 120°C. Reaction progress is monitored via TLC until the starting material is fully consumed, typically resulting in high conversion rates. The detailed standardized synthesis steps, including specific molar ratios and workup procedures, are outlined in the guide below to ensure reproducibility and safety in your laboratory operations.
- Combine cobalt tetrafluoroborate hydrate catalyst, Xantphos ligand, and zinc powder with 3,4-O-carbonate galactalose in acetonitrile solvent.
- Add the specific thiol or phenol reactant (e.g., p-toluenethiophenol) to the reaction mixture under inert atmosphere.
- Heat the mixture to 100-120°C with stirring until TLC indicates complete consumption of the starting material, then purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this cobalt-catalyzed methodology presents a compelling value proposition centered on cost stability and operational resilience. By shifting away from volatile precious metal markets, manufacturers can secure a more predictable cost structure for their key intermediates. The elimination of expensive metal scavengers and the reduction in purification complexity directly contribute to substantial cost savings in pharmaceutical intermediate manufacturing. Furthermore, the robustness of the reaction conditions ensures consistent batch-to-batch quality, minimizing the risk of production delays caused by failed runs or out-of-specification results. This reliability is essential for maintaining uninterrupted supply lines to downstream API producers.
- Cost Reduction in Manufacturing: The substitution of gold or palladium catalysts with inexpensive cobalt salts fundamentally alters the economic model of thiosugar production. This change eliminates the need for costly metal recovery processes and reduces the capital expenditure associated with specialized equipment for handling toxic heavy metals. Additionally, the high yields achieved reduce the amount of raw material required per unit of product, further driving down the variable costs. The simplified workup procedure also lowers labor and solvent consumption, contributing to a leaner and more cost-effective manufacturing footprint.
- Enhanced Supply Chain Reliability: The reagents utilized in this process, including cobalt salts and common phosphine ligands, are commercially available from multiple global suppliers, reducing the risk of single-source dependency. The robustness of the reaction against minor variations in conditions ensures that production schedules can be met consistently, even in large-scale campaigns. This stability allows supply chain planners to optimize inventory levels and reduce safety stock requirements, freeing up working capital. Moreover, the compatibility with standard solvents like acetonitrile simplifies logistics and waste management protocols.
- Scalability and Environmental Compliance: The protocol is inherently designed for scalability, utilizing standard heating and stirring equipment found in most pilot and production plants. The high selectivity of the reaction minimizes the generation of hazardous waste streams, facilitating easier compliance with increasingly stringent environmental regulations. The absence of persistent heavy metal contaminants in the final product simplifies the regulatory filing process for new drug applications. This environmental advantage not only reduces disposal costs but also enhances the corporate sustainability profile of the manufacturing organization.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and optimization studies presented in the patent literature. They are intended to provide clarity on the practical aspects of adopting this method for your specific production needs. Understanding these details will help in making informed decisions about process integration and resource allocation.
Q: What are the advantages of using cobalt catalysts over precious metals for thiosugar synthesis?
A: Cobalt catalysts are significantly more abundant and cost-effective than traditional gold or palladium systems. Furthermore, the specific cobalt/Xantphos system described eliminates the need for expensive metal scavengers, simplifying downstream purification and reducing overall production costs.
Q: How does the addition of zinc powder impact the reaction efficiency?
A: Zinc powder acts as a crucial additive that promotes the catalytic cycle of the cobalt species. Experimental data indicates that its inclusion boosts isolated yields substantially, transforming moderate conversions into high-efficiency transformations suitable for industrial application.
Q: Is this method scalable for commercial production of glycosyl donors?
A: Yes, the protocol utilizes robust conditions (100-120°C) and common solvents like acetonitrile. The high regioselectivity (>20:1) minimizes byproduct formation, making the purification process straightforward and highly amenable to scaling from laboratory to multi-kilogram production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Thio-1-Glycals Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the success of modern drug development pipelines. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements with precision and speed. We are committed to delivering products that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Our expertise in carbohydrate chemistry allows us to navigate the complexities of glycosyl donor synthesis, providing you with a dependable source for these essential building blocks.
We invite you to collaborate with us to leverage this advanced cobalt-catalyzed technology for your projects. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume and purity needs. Please contact us to request specific COA data and route feasibility assessments for your target molecules. By partnering with us, you gain access to a supply chain that prioritizes efficiency, quality, and long-term strategic value.
