Scalable One-Pot Synthesis of Bioactive Furoindole Scaffolds for Pharmaceutical Applications
Scalable One-Pot Synthesis of Bioactive Furoindole Scaffolds for Pharmaceutical Applications
The development of efficient synthetic routes for complex heterocyclic scaffolds remains a cornerstone of modern pharmaceutical process chemistry, particularly when targeting bioactive cores that combine multiple pharmacophores. Patent CN101974004A introduces a robust and economically viable methodology for constructing the furoindole skeleton, a privileged structure found in numerous biologically active compounds ranging from antimicrobial agents to potential antitumor candidates. This technology leverages a tandem multicomponent reaction strategy followed by an intramolecular cyclization, effectively merging the structural benefits of both indole and furan rings into a single, rigid framework. By utilizing readily available starting materials such as o-halobenzaldehydes and specific hydroxy-containing precursors, this process addresses the critical industry need for streamlined manufacturing of high-purity pharmaceutical intermediates. The resulting compounds, characterized by the general structure shown below, exhibit significant potential for drug discovery applications due to their unique electronic properties and conformational stability.
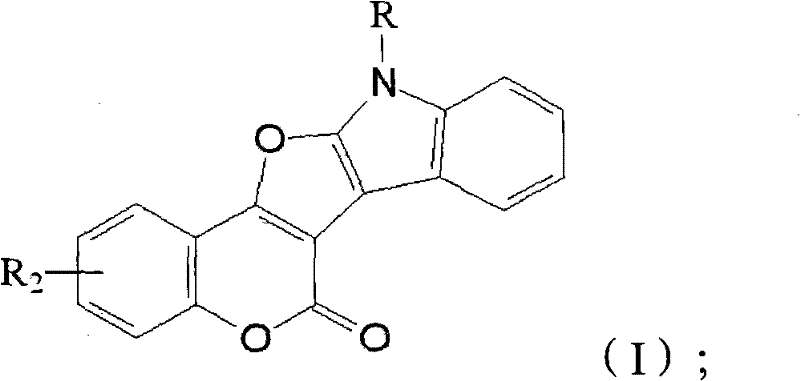
Furthermore, the versatility of this synthetic approach allows for the facile introduction of diverse functional groups at multiple positions on the fused ring system, enabling medicinal chemists to rapidly explore structure-activity relationships without being constrained by cumbersome synthetic limitations. The ability to access these complex architectures through a convergent pathway represents a substantial advancement over linear syntheses that often suffer from low overall yields and excessive waste generation. As the demand for novel heterocyclic building blocks continues to rise in the sectors of oncology and infectious disease research, mastering such efficient coupling technologies becomes essential for maintaining a competitive edge in the global supply chain of fine chemical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes to fused indole-furan systems have historically been plagued by operational complexity and poor atom economy, often requiring harsh reaction conditions that are incompatible with sensitive functional groups. Prior art methodologies frequently rely on multi-step sequences involving the separate preparation of intermediates, each necessitating distinct isolation and purification protocols that dramatically increase production costs and lead times. For instance, earlier approaches described in related literature often utilize strong mineral acids like sulfuric acid or expensive transition metal catalysts that pose significant challenges for waste disposal and environmental compliance on an industrial scale. These conventional processes typically involve the condensation of salicylic acid derivatives followed by separate cyclization steps, creating bottlenecks that hinder the rapid scale-up required for clinical trial material production. Moreover, the reliance on stoichiometric amounts of reagents and the generation of substantial byproduct streams make these legacy methods increasingly unsustainable in the context of modern green chemistry initiatives.
The Novel Approach
In stark contrast, the methodology disclosed in CN101974004A revolutionizes the construction of the furoindole core by employing a telescoped, one-pot procedure that seamlessly integrates bond-forming events without the need for intermediate workup. This innovative strategy initiates with a multicomponent cascade reaction between an o-halobenzaldehyde, a hydroxy-containing compound, and an isonitrile, generating a reactive intermediate in situ that is immediately subjected to cyclization conditions. By eliminating the isolation step, the process not only saves valuable time and solvent but also minimizes material loss associated with physical handling and purification of unstable intermediates. The use of a copper-based catalytic system further distinguishes this approach, offering a cost-effective alternative to precious metal catalysis while maintaining high levels of regioselectivity and yield. This streamlined workflow significantly enhances the feasibility of commercial-scale manufacturing, allowing producers to respond more agilely to market demands for specialized heterocyclic intermediates used in the synthesis of advanced therapeutic agents.
Mechanistic Insights into Copper-Catalyzed Intramolecular Ullmann Coupling
The success of this synthetic transformation hinges on the precise orchestration of a copper-catalyzed intramolecular Ullmann-type coupling, which serves as the key ring-closing step to forge the indole nitrogen-carbon bond. Following the initial formation of the acyclic or semi-cyclic intermediate via the multicomponent reaction, the addition of cuprous halide (preferably CuI) and L-proline facilitates the activation of the aryl halide moiety present in the substrate. The L-proline acts as a crucial bidentate ligand that stabilizes the copper center, preventing aggregation and enhancing the solubility of the catalyst in the organic medium, thereby promoting efficient oxidative addition and reductive elimination cycles. This catalytic cycle enables the nucleophilic attack of the nitrogen atom onto the adjacent aromatic ring, effectively closing the five-membered pyrrole ring of the indole system under relatively mild thermal conditions. The mechanistic pathway ensures high fidelity in bond formation, minimizing the formation of homocoupling byproducts that are often observed in less optimized cross-coupling reactions.
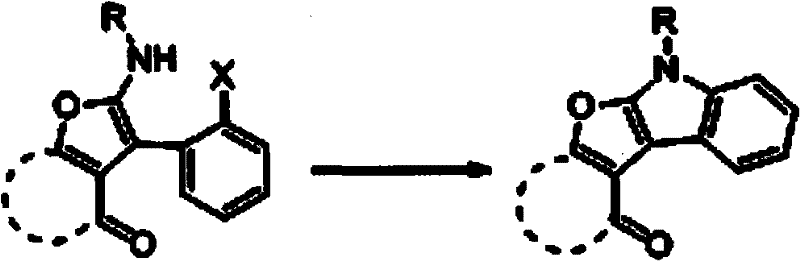
From an impurity control perspective, this mechanism offers distinct advantages by operating under neutral to slightly basic conditions provided by inorganic bases like potassium carbonate or cesium carbonate. The absence of strongly acidic or oxidizing environments preserves sensitive functional groups such as esters or ketones that might otherwise degrade during harsher cyclization protocols. Furthermore, the choice of solvent, typically toluene, provides an optimal boiling point range (90°C to 120°C) that supplies sufficient thermal energy to drive the coupling to completion without inducing thermal decomposition of the product. The rigorous control over reaction parameters, including the molar ratios of catalyst to substrate, ensures that the reaction proceeds with high conversion rates, thereby simplifying downstream purification and ensuring the final API intermediate meets stringent quality specifications required by regulatory bodies.
How to Synthesize Furoindole Heterocycles Efficiently
The practical implementation of this synthesis involves a straightforward two-stage protocol that can be executed in standard glass-lined reactors equipped with reflux condensers and inert gas lines. Operators begin by charging the reactor with the o-halobenzaldehyde, the specific hydroxy-containing precursor (such as 4-hydroxycoumarin), and the chosen isonitrile in a suitable organic solvent like toluene. The mixture is heated to reflux temperatures between 90°C and 120°C for a duration ranging from 2 to 30 hours to ensure complete consumption of the starting materials and formation of the intermediate species. Upon confirmation of the first stage completion, the reaction mixture is cooled slightly before the direct addition of the copper catalyst system, comprising CuI, L-proline, and an inorganic base, without any filtration or extraction steps. The detailed standardized synthesis steps for this process are outlined in the guide below.
- Mix o-halobenzaldehyde, hydroxy-containing compound, and isonitrile in toluene and reflux at 90-120°C for 2-30 hours to generate the intermediate.
- Without isolating the intermediate, add CuI, L-proline, and an inorganic base directly to the reaction mixture.
- Continue refluxing the mixture at 90-120°C for 10-40 hours to effect intramolecular cyclization, then purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented technology translates into tangible strategic benefits that extend far beyond simple laboratory curiosity. The consolidation of multiple synthetic steps into a single vessel operation fundamentally alters the cost structure of manufacturing these complex heterocycles, removing the capital expenditure and operational overhead associated with intermediate isolation equipment and storage. By reducing the total number of unit operations, facilities can achieve higher throughput rates with existing infrastructure, effectively debottlenecking production lines that were previously constrained by lengthy batch cycles. This efficiency gain is critical for maintaining supply continuity in the face of fluctuating raw material availability, as the process relies on commodity chemicals that are widely sourced from the global petrochemical and fine chemical markets.
- Cost Reduction in Manufacturing: The economic impact of this process is driven primarily by the elimination of intermediate purification stages, which traditionally account for a significant portion of manufacturing costs due to solvent usage, chromatography media, and labor. By utilizing a base-metal copper catalyst instead of expensive palladium or rhodium complexes, the direct material cost of the catalytic system is drastically lowered, contributing to a more favorable margin profile for the final product. Additionally, the simplified workup procedure reduces the volume of hazardous waste generated, leading to substantial savings in waste disposal fees and environmental compliance costs. These cumulative efficiencies allow suppliers to offer competitive pricing models for high-value pharmaceutical intermediates without compromising on quality or purity standards.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route enhances supply chain resilience by minimizing the risk of batch failures associated with complex multi-step sequences. Since the starting materials—o-halobenzaldehydes, hydroxy-coumarins, and isonitriles—are commercially available from multiple vendors, the risk of single-source dependency is mitigated, ensuring a steady flow of inputs for continuous production. The tolerance of the reaction to various substituents also means that a single production line can be easily adapted to manufacture a library of analogues, providing flexibility to pivot quickly between different product SKUs based on customer demand. This adaptability is crucial for contract manufacturing organizations that need to manage diverse portfolios of active pharmaceutical ingredients and intermediates efficiently.
- Scalability and Environmental Compliance: Scaling this process from gram-scale laboratory experiments to multi-ton commercial production is facilitated by the use of standard reflux conditions and common organic solvents that do not require specialized high-pressure or cryogenic equipment. The "one-pot" nature of the reaction inherently reduces the solvent footprint per kilogram of product, aligning with corporate sustainability goals and increasingly strict environmental regulations regarding volatile organic compound emissions. The use of L-proline, a naturally occurring amino acid, as a ligand further underscores the green chemistry credentials of the process, appealing to partners who prioritize environmentally responsible sourcing. Consequently, this technology supports the long-term viability of manufacturing operations by future-proofing them against tightening regulatory frameworks while maximizing output capacity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this furoindole synthesis technology, derived directly from the patent specifications and process data. Understanding these details helps stakeholders evaluate the feasibility of integrating this route into their existing manufacturing pipelines or R&D programs. The answers reflect the specific advantages of the copper-catalyzed one-pot method over traditional alternatives, focusing on operational simplicity and product quality.
Q: What are the key advantages of this one-pot synthesis method?
A: The primary advantage is the elimination of intermediate isolation and purification steps, which drastically simplifies the workflow and reduces solvent consumption and waste generation compared to traditional multi-step protocols.
Q: Why is the CuI/L-proline catalyst system preferred over noble metals?
A: Copper iodide paired with L-proline offers a significantly more cost-effective alternative to palladium or other noble metal catalysts, while maintaining high efficiency for the intramolecular Ullmann coupling required to close the indole ring.
Q: Can this method be adapted for different functional groups?
A: Yes, the protocol is highly versatile; by varying the hydroxy-containing starting materials (such as 4-hydroxycoumarin or 2-hydroxy-1,4-naphthoquinone) and the isonitrile component, a diverse library of substituted furoindole derivatives can be accessed.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Furoindole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that efficient synthetic methodologies play in accelerating the development of next-generation therapeutics. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial manufacturing is seamless and reliable. We are committed to delivering high-purity furoindole intermediates that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. By leveraging advanced catalytic technologies like the one described in CN101974004A, we can provide cost-effective solutions that help our partners reduce their overall cost of goods sold while maintaining the highest standards of quality and safety.
We invite you to contact our technical procurement team to discuss how we can support your specific project requirements with tailored manufacturing strategies. Whether you need a Customized Cost-Saving Analysis for your current supply chain or require specific COA data and route feasibility assessments for new targets, our experts are ready to collaborate with you. Let us help you optimize your sourcing strategy and secure a reliable supply of high-quality heterocyclic building blocks for your pharmaceutical applications.
