Advancing Pharmaceutical Intermediate Production via Efficient Nickel-Catalyzed 3-Thioether Isoindolinone Synthesis
Advancing Pharmaceutical Intermediate Production via Efficient Nickel-Catalyzed 3-Thioether Isoindolinone Synthesis
The landscape of pharmaceutical intermediate manufacturing is constantly evolving, driven by the need for more sustainable, cost-effective, and robust synthetic methodologies. A significant breakthrough in this domain is detailed in patent CN110437128B, which introduces a novel synthetic route for 3-thioether-based isoindolinone compounds. These scaffolds are critical building blocks in medicinal chemistry, frequently appearing in bioactive natural products and complex drug molecules. The traditional reliance on expensive noble metal catalysts or harsh acidic and basic conditions has long been a bottleneck for process chemists aiming for scalable production. This new technology leverages earth-abundant Nickel(II) catalysis to facilitate the in situ generation of imine cations, which subsequently undergo nucleophilic addition with thiol compounds. By shifting the paradigm from precious metals to base metals, this method not only addresses economic concerns but also aligns with green chemistry principles by simplifying post-reaction processing and waste management.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of carbon-sulfur bonds in isoindolinone frameworks has relied heavily on sophisticated and costly catalytic systems. Conventional approaches often utilize gold(I), palladium(II), or ruthenium complexes, which, while effective, impose a substantial financial burden on large-scale manufacturing operations. Beyond the direct cost of the catalyst, these methods frequently require stringent reaction conditions, including strong acids or bases, to activate the substrate. Such harsh environments pose significant risks to functional group tolerance, often leading to the degradation of sensitive moieties within complex molecular architectures. Furthermore, reactions conducted under extreme pH conditions necessitate extensive neutralization and washing steps during the workup phase. This not only increases the volume of aqueous waste generated but also complicates the isolation of the pure product, thereby extending production timelines and increasing the overall environmental footprint of the synthesis.
The Novel Approach
In stark contrast to these legacy methods, the technology disclosed in CN110437128B utilizes inexpensive and readily available Nickel(II) salts to drive the transformation efficiently. This approach operates under significantly milder thermal conditions, typically between 80°C and 120°C, eliminating the need for extreme heating or cooling. The core innovation lies in the ability of the Ni(II) species to promote the formation of reactive imine cation intermediates directly from 3-hydroxy-2-benzyl-isoindolin-1-one precursors. These electrophilic intermediates are then trapped by various thiol nucleophiles to forge the desired carbon-sulfur bond with high fidelity. As illustrated in the general reaction scheme below, this one-pot procedure streamlines the synthetic workflow, removing the need for pre-functionalization or protecting group strategies that are common in older routes.
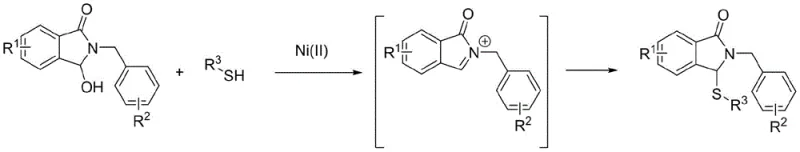
The operational simplicity of this novel approach cannot be overstated. By avoiding the use of corrosive acids or caustic bases, the reaction mixture remains relatively neutral, which drastically simplifies the downstream purification process. The products can often be isolated through standard extraction and silica gel chromatography without the need for complex neutralization protocols. This reduction in unit operations translates directly into lower operational expenditures and a smaller physical footprint for manufacturing facilities. For procurement managers and supply chain directors, this represents a tangible opportunity to reduce the cost of goods sold (COGS) while simultaneously improving the reliability of the supply chain by minimizing the risk of batch failures due to sensitive reaction conditions.
Mechanistic Insights into Ni(II)-Catalyzed Imine Cation Formation
Understanding the mechanistic underpinnings of this transformation is crucial for R&D directors evaluating its feasibility for complex API synthesis. The reaction initiates with the coordination of the Nickel(II) catalyst to the hydroxyl group of the isoindolinone substrate. This coordination activates the C-O bond, facilitating its departure and generating a highly electrophilic N-acyl iminium ion intermediate in situ. This cationic species is resonance-stabilized by the adjacent carbonyl group and the nitrogen atom, yet remains sufficiently reactive to undergo rapid nucleophilic attack. The thiol compound, acting as a soft nucleophile, attacks the electrophilic carbon center of the iminium ion. This step is the rate-determining factor in many similar transformations, but the Lewis acidic nature of the nickel catalyst ensures that the concentration of the reactive iminium species is maintained at an optimal level throughout the reaction course.
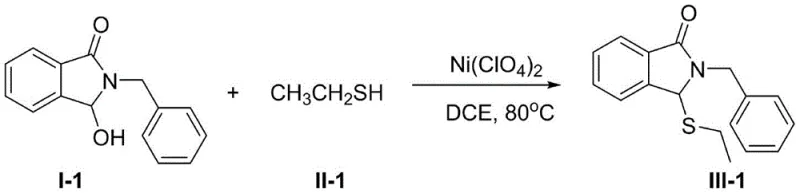
From an impurity control perspective, this mechanism offers distinct advantages over radical-based or oxidative coupling methods. Because the pathway proceeds through a polarized ionic mechanism rather than a radical chain process, the formation of homocoupled disulfide byproducts is minimized. Additionally, the mild conditions prevent the epimerization of chiral centers that might be present on the thiol or the isoindolinone ring, preserving the stereochemical integrity of the molecule. The high yields reported, ranging from 82% to 95% across diverse substrates as seen in specific examples like the synthesis of compound III-1, indicate that side reactions such as polymerization or hydrolysis are effectively suppressed. This high level of chemoselectivity is particularly valuable when synthesizing advanced intermediates where purity specifications are rigorous, ensuring that the final product meets the stringent quality standards required for pharmaceutical applications.
How to Synthesize 3-Thioether Isoindolinone Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires careful attention to reagent stoichiometry and solvent choice to maximize efficiency. The patent outlines a robust protocol where the 3-hydroxy precursor and the thiol are dissolved in a polar aprotic or chlorinated solvent, followed by the addition of the nickel catalyst. The reaction is then heated to the specified temperature range until conversion is complete. While the general principles are straightforward, optimizing parameters such as catalyst loading and reaction time is essential for different substrate combinations. For a comprehensive understanding of the specific operational parameters, including exact molar ratios and workup procedures, please refer to the detailed standardized synthesis steps provided in the guide below.
- Dissolve 3-hydroxy-2-benzyl-isoindolin-1-one and the chosen thiol compound in a suitable solvent such as 1,2-dichloroethane or DMF.
- Add a Ni(II) catalyst, such as Ni(ClO4)2 or NiCl2, to the reaction mixture and stir at temperatures between 80°C and 120°C for 5 to 10 hours.
- Upon completion, cool the mixture, extract with dichloromethane, wash with brine, dry over anhydrous Na2SO4, and purify the residue via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For stakeholders focused on the bottom line and supply continuity, the shift to this nickel-catalyzed methodology offers compelling strategic advantages. The primary driver of value is the substitution of expensive noble metals with commodity-grade nickel salts. This change alone results in a drastic reduction in raw material costs, which is amplified when considering the scale of industrial production. Furthermore, the simplified workup procedure eliminates the need for specialized waste treatment associated with heavy metal removal or neutralization of strong acids. This reduction in processing complexity leads to shorter cycle times and higher throughput, allowing manufacturers to respond more agilely to market demand fluctuations without compromising on quality or safety standards.
- Cost Reduction in Manufacturing: The economic impact of replacing gold or palladium catalysts with nickel derivatives is profound, as nickel is orders of magnitude cheaper and more abundant globally. This substitution removes a significant variable cost from the manufacturing equation, providing a buffer against volatile precious metal markets. Additionally, the elimination of harsh reagents reduces the consumption of auxiliary chemicals used for quenching and neutralization, further lowering the overall cost per kilogram of the produced intermediate. The high atom economy and yield of the reaction ensure that raw material utilization is maximized, minimizing waste disposal costs and enhancing the overall profitability of the production line.
- Enhanced Supply Chain Reliability: Relying on earth-abundant metals like nickel mitigates the supply chain risks associated with geopolitically constrained precious metals. Nickel salts are widely available from multiple global suppliers, ensuring a stable and continuous supply of critical catalysts. The robustness of the reaction conditions means that the process is less susceptible to minor variations in temperature or reagent quality, leading to more consistent batch-to-batch performance. This reliability is crucial for maintaining uninterrupted production schedules and meeting the just-in-time delivery requirements of downstream pharmaceutical clients who depend on a steady flow of high-quality intermediates.
- Scalability and Environmental Compliance: The mild reaction conditions and simple aqueous workup make this process inherently scalable from gram to multi-ton quantities without significant engineering hurdles. The absence of corrosive acids or bases reduces the corrosion load on reactor vessels and piping, extending equipment lifespan and reducing maintenance downtime. From an environmental standpoint, the reduced generation of saline wastewater and heavy metal waste simplifies compliance with increasingly stringent environmental regulations. This eco-friendly profile not only lowers regulatory risk but also enhances the corporate sustainability credentials of the manufacturing organization, which is becoming a key differentiator in the global chemical market.
Frequently Asked Questions (FAQ)
To assist technical teams in evaluating this technology for their specific applications, we have compiled answers to common inquiries regarding the scope and limitations of this nickel-catalyzed method. These insights are derived directly from the experimental data and technical specifications outlined in the patent documentation. Understanding these nuances is vital for assessing the fit of this methodology within existing synthetic routes and for planning future process development campaigns effectively.
Q: What are the primary advantages of using Ni(II) catalysts over noble metals for this synthesis?
A: The use of Ni(II) catalysts significantly reduces raw material costs compared to traditional gold or palladium systems. Furthermore, nickel catalysts operate under milder conditions, reducing energy consumption and simplifying the removal of metal residues from the final pharmaceutical intermediate.
Q: Does this method tolerate acid- or base-sensitive functional groups?
A: Yes, unlike traditional methods requiring harsh acidic or basic conditions which often necessitate neutralization steps, this Ni(II)-catalyzed protocol proceeds under neutral to mild conditions. This makes it highly suitable for substrates containing sensitive functional groups that would otherwise degrade.
Q: What is the typical yield range for this nickel-catalyzed transformation?
A: According to the experimental data in patent CN110437128B, the reaction consistently achieves high yields ranging from 82% to 95% across various thiol substrates, demonstrating robust efficiency for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Thioether Isoindolinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this nickel-catalyzed synthesis for the production of high-value pharmaceutical intermediates. Our team of expert process chemists has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, which employ state-of-the-art analytical techniques to verify identity and assay. By leveraging this efficient synthetic route, we can offer our partners a reliable source of 3-thioether isoindolinone derivatives that balance cost-efficiency with uncompromising quality.
We invite you to engage with our technical procurement team to discuss how this innovative methodology can be integrated into your supply chain. Whether you require a Customized Cost-Saving Analysis for your current projects or need to review specific COA data and route feasibility assessments, we are ready to provide the support you need. Partnering with us means gaining access to a vertically integrated manufacturing capability that prioritizes innovation, sustainability, and reliability, positioning your organization for success in a competitive global marketplace.
