Advanced One-Pot Synthesis of (2-Hydroxyphenyl)(2,4,6-trimethoxyphenyl)(phenyl)methane for Industrial Scale-up
Introduction to Next-Generation Triarylmethane Manufacturing
The landscape of fine chemical synthesis is constantly evolving, driven by the urgent need for more sustainable, cost-effective, and operationally simple methodologies. A significant breakthrough in this domain is documented in Chinese Patent CN111484397B, which discloses a highly efficient method for synthesizing (2-hydroxyphenyl)(2,4,6-trimethoxyphenyl)(phenyl)methane. This specific triarylmethane derivative holds immense potential as a critical building block in medicinal chemistry and material science applications. The patented technology represents a paradigm shift from traditional multi-step sequences to a streamlined 'one-pot' reaction strategy. By leveraging the synergistic effects of inexpensive catalysts such as iron(III) chloride or molecular iodine, this process eliminates the need for complex ligand systems and harsh basic conditions. For R&D directors and procurement specialists alike, this innovation signals a pathway toward drastically simplified supply chains and reduced manufacturing overheads. The ability to introduce three distinct aryl groups in a single operational step from commercially abundant starting materials underscores the robustness of this chemical transformation.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of triarylmethane scaffolds has relied heavily on classical Friedel-Crafts alkylation or transition-metal-catalyzed cross-coupling reactions. These conventional pathways are fraught with significant technical and economic bottlenecks that hinder large-scale adoption. Friedel-Crafts methodologies, while conceptually straightforward, often suffer from poor regioselectivity, leading to complex mixtures of isomeric by-products that are difficult and costly to separate. Furthermore, these reactions frequently necessitate the use of stoichiometric amounts of strong Lewis acids or harsh reaction conditions that can degrade sensitive functional groups. Alternatively, modern cross-coupling approaches, though more selective, typically depend on precious metal catalysts like palladium or rhodium, coupled with expensive phosphine ligands. The reliance on such scarce resources not only inflates the raw material costs but also introduces stringent requirements for residual metal removal, a critical quality control hurdle in pharmaceutical intermediate manufacturing. Additionally, many existing routes require pre-functionalized substrates, adding extra synthetic steps and reducing the overall atom economy of the process.
The Novel Approach
In stark contrast to these legacy methods, the technology outlined in CN111484397B offers a revolutionary 'one-pot' solution that directly assembles the target molecule from salicylaldehyde, phenylboronic acid, and 1,3,5-trimethoxybenzene. This approach bypasses the need for pre-activation of substrates or the use of exotic reagents. The reaction proceeds under mild thermal conditions, typically between 40°C and 120°C, and achieves completion within a remarkably short timeframe of 20 to 80 minutes. The absence of base or ligand additives simplifies the reaction matrix, thereby facilitating easier downstream processing. As illustrated in the reaction scheme below, the convergence of these three components is mediated efficiently by either iodine or ferric chloride, demonstrating exceptional chemical selectivity.
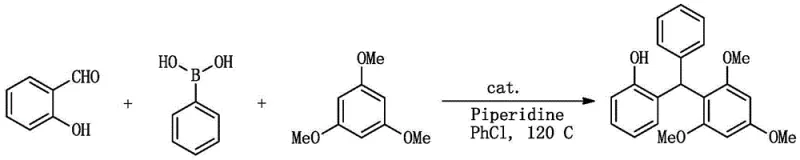
This visual representation highlights the elegance of the transformation, where the aldehyde functionality serves as the electrophilic center, activated by the catalyst to undergo sequential nucleophilic attacks by the electron-rich aromatic rings. The result is a high-yielding process that minimizes waste generation and maximizes throughput, addressing the core pain points of traditional triarylmethane synthesis.
Mechanistic Insights into FeCl3-Catalyzed Cyclization
Understanding the mechanistic underpinnings of this transformation is crucial for optimizing process parameters and ensuring consistent product quality. The reaction likely proceeds through a Lewis acid-mediated activation of the carbonyl group in salicylaldehyde. When iron(III) chloride is employed, it coordinates with the oxygen atom of the aldehyde, increasing the electrophilicity of the carbonyl carbon. This activated species is then susceptible to nucleophilic attack by the highly electron-rich 1,3,5-trimethoxybenzene, which acts as a potent nucleophile due to the resonance donation of its methoxy substituents. Following the initial addition and dehydration steps, the resulting benzylic cation intermediate is further intercepted by the phenyl group derived from phenylboronic acid. While the exact role of the boronic acid in this specific non-basic medium may involve a unique transmetallation or direct electrophilic aromatic substitution pathway facilitated by the iron species, the net outcome is the precise installation of the phenyl ring. The presence of piperidine in the reaction mixture likely serves to moderate the acidity and assist in the final aromatization or proton transfer steps, ensuring the stability of the phenolic hydroxyl group.
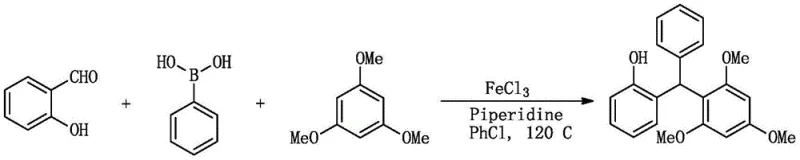
The alternative pathway utilizing molecular iodine suggests a halogen-bonding or electrophilic activation mechanism, where iodine acts as a mild Lewis acid to activate the aldehyde similarly. This dual-catalyst flexibility provides manufacturers with the option to select the most cost-effective or operationally convenient reagent for their specific facility constraints. Crucially, the high chemoselectivity observed ensures that the phenolic hydroxyl group remains intact without requiring protection-deprotection strategies, a feature that significantly reduces the step count and improves the overall yield. Impurity profiles are tightly controlled by the specificity of the catalyst-substrate interaction, minimizing the formation of poly-alkylated side products that commonly plague Friedel-Crafts reactions.
How to Synthesize (2-Hydroxyphenyl)(2,4,6-trimethoxyphenyl)(phenyl)methane Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires strict adherence to the optimized molar ratios and thermal profiles described in the patent. The process is designed to be robust, tolerating slight variations in reagent purity while maintaining high conversion rates. Operators should focus on maintaining the reaction temperature within the specified range of 40°C to 120°C, with 120°C in chlorobenzene being the preferred condition for rapid kinetics. Monitoring the reaction progress via Thin Layer Chromatography (TLC) is essential to determine the precise endpoint, indicated by the disappearance of the starting salicylaldehyde spot. Once the reaction is deemed complete, the workup procedure is straightforward, involving cooling, extraction with ethyl acetate, and standard aqueous washes. For detailed operational protocols and safety guidelines, please refer to the standardized synthesis steps provided below.
- Mix salicylaldehyde, phenylboronic acid, 1,3,5-trimethoxybenzene, piperidine, and catalyst (I2 or FeCl3) in a solvent like chlorobenzene.
- Heat the mixture to 120°C under stirring for 20 to 80 minutes until TLC indicates complete consumption of the aldehyde.
- Cool to room temperature, extract with ethyl acetate, wash with water and brine, dry over sodium sulfate, and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this patented methodology offers profound advantages for procurement managers and supply chain heads tasked with optimizing the sourcing of complex pharmaceutical intermediates. The shift away from precious metal catalysts to earth-abundant iron or iodine represents a fundamental restructuring of the cost base for this molecule. By eliminating the dependency on volatile palladium markets and expensive ligand libraries, manufacturers can achieve substantial cost savings in raw material procurement. Furthermore, the simplification of the synthetic route directly translates to reduced operational expenditures, as fewer unit operations and less specialized equipment are required to manage the reaction and purification stages.
- Cost Reduction in Manufacturing: The economic impact of replacing noble metal catalysts with iron(III) chloride or iodine cannot be overstated. These catalysts are commodity chemicals with stable pricing and widespread availability, removing the supply risk associated with strategic metals. Additionally, the 'one-pot' nature of the reaction eliminates the need for intermediate isolation and purification steps, which are typically the most resource-intensive phases of chemical manufacturing. This consolidation of steps leads to a drastic simplification of the process flow, reducing labor costs, solvent consumption, and energy usage. The high yields reported in the patent examples further enhance the economic viability by maximizing the output per batch, ensuring that the cost per kilogram of the final active intermediate is significantly lower than that produced via conventional multi-step routes.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of universally available starting materials. Salicylaldehyde, phenylboronic acid, and 1,3,5-trimethoxybenzene are bulk chemicals produced by numerous global suppliers, ensuring a diversified and secure supply base. This contrasts sharply with specialized intermediates that may rely on single-source vendors. The robustness of the reaction conditions also means that the process can be easily transferred between different manufacturing sites without significant re-validation efforts. The short reaction time of under 80 minutes allows for high throughput and flexible scheduling, enabling manufacturers to respond rapidly to fluctuations in market demand. This agility is critical for maintaining continuity of supply in the fast-paced pharmaceutical sector.
- Scalability and Environmental Compliance: The environmental footprint of this synthesis is markedly reduced compared to traditional methods. The absence of heavy metal residues simplifies waste treatment protocols and lowers the cost of effluent management. The use of common organic solvents like chlorobenzene or ethanol facilitates solvent recovery and recycling, aligning with green chemistry principles. From a scalability standpoint, the exothermic profile of the reaction is manageable, and the lack of sensitive reagents allows for safe scale-up from gram to ton quantities. The simple post-treatment involving filtration and column chromatography (or potentially crystallization upon further optimization) ensures that the process remains practical even at commercial scales, supporting the production of high-purity intermediates required for regulatory compliance.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a clear understanding of the process capabilities and limitations. Understanding these details is essential for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: What are the primary advantages of this one-pot synthesis over traditional Friedel-Crafts alkylation?
A: Unlike traditional Friedel-Crafts methods which often suffer from poor regioselectivity and require harsh conditions, this novel approach utilizes mild Lewis acid or halogen catalysis to achieve high chemical selectivity in a single step without expensive ligands.
Q: Which catalysts are suitable for this triarylmethane derivative production?
A: The patent specifies two highly effective and economically viable catalyst options: molecular iodine (I2) and iron(III) chloride (FeCl3), both of which are inexpensive and readily available compared to noble metal catalysts.
Q: Is this process scalable for commercial manufacturing of pharmaceutical intermediates?
A: Yes, the process is designed for scalability, featuring short reaction times (20-80 minutes), simple post-treatment involving standard extraction and chromatography, and the use of common industrial solvents like chlorobenzene.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (2-Hydroxyphenyl)(2,4,6-trimethoxyphenyl)(phenyl)methane Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the synthesis method described in CN111484397B for the production of high-value triarylmethane derivatives. As a premier CDMO partner, we possess the technical expertise and infrastructure to translate this innovative laboratory protocol into a robust commercial reality. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with unwavering consistency. We are committed to delivering products that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Whether you require custom synthesis or large-scale toll manufacturing, our facility is designed to handle complex organic transformations with the highest standards of safety and quality.
We invite you to collaborate with us to leverage this cost-effective technology for your next project. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our manufacturing capabilities can drive efficiency and value for your organization. Let us be your trusted partner in navigating the complexities of fine chemical supply chains.
