Advanced Polymeric Chiral Catalysts for High-Purity Optically Active Cyclocarbonates Manufacturing
The chemical industry is constantly seeking more efficient pathways to synthesize chiral building blocks, and patent CN101270112A presents a significant advancement in this domain by introducing a novel polymeric chiral catalyst system. This technology addresses the critical challenge of preparing optically active cyclic carbonates directly from racemic alkylene oxides and carbon dioxide through a catalytic asymmetric cycloaddition reaction. Unlike traditional methods that rely on stoichiometric chiral auxiliaries or expensive enzymatic processes, this invention utilizes a robust two-component catalyst system comprising a polymeric chiral Schiff base-trivalent metal complex and a specific cocatalyst. The strategic design of the catalyst backbone allows for exceptional stability and ease of recovery, positioning this method as a highly viable route for the commercial scale-up of complex pharmaceutical intermediates. By leveraging carbon dioxide as a C1 building block, this process not only achieves high atom economy but also aligns with green chemistry principles, making it an attractive option for manufacturers aiming to reduce their environmental footprint while maintaining rigorous quality standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of optically active cyclic carbonates has been plagued by significant technical and economic hurdles that hinder large-scale adoption. Conventional approaches often rely on small-molecule chiral catalysts, such as Jacobsen-type complexes, which, while effective in laboratory settings, present severe difficulties in industrial processing. A primary limitation is the difficulty in separating the homogeneous catalyst from the product mixture, often necessitating energy-intensive distillation or costly chromatographic purification steps that drastically increase production costs. Furthermore, literature analysis reveals discrepancies in reported performance; for instance, some prior art claims high enantiomeric excess (e.e.) values that are not reproducible under standard conditions, with actual e.e. values dropping significantly as temperature increases from -20°C to 25°C. Additionally, these small-molecule catalysts frequently suffer from deactivation after a single run, rendering them economically unfeasible for continuous manufacturing processes where catalyst turnover number (TON) is a key metric for profitability.
The Novel Approach
The methodology disclosed in the patent overcomes these deficiencies by employing a polymeric architecture for the chiral ligand, fundamentally changing the physical properties of the catalyst without compromising its stereochemical induction capabilities. By incorporating chiral binaphthol or diamine units into a polymer chain, the catalyst becomes heterogeneous in nature, allowing for simple precipitation and filtration to separate it from the reaction mixture. This innovation facilitates the reducing lead time for high-purity cyclic carbonates by eliminating complex downstream processing. The system operates effectively under mild conditions, with carbon dioxide pressures ranging from atmospheric pressure up to 2 MPa and temperatures between 0°C and 60°C. Crucially, the polymeric catalyst demonstrates remarkable reusability, maintaining consistent activity and selectivity over more than ten consecutive cycles, which represents a paradigm shift towards sustainable and cost-effective fine chemical manufacturing.
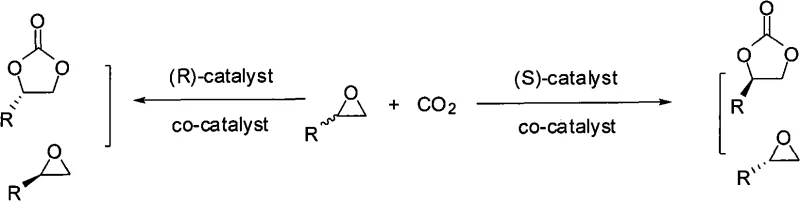
Mechanistic Insights into Polymeric Chiral Schiff Base-Metal Catalysis
The core of this technology lies in the sophisticated design of the polymeric chiral Schiff base-trivalent metal complex, which acts as the primary driver for stereochemical induction. The catalyst features a backbone derived from chiral sources such as (R,R) or (S,S) cyclic diamines or binaphthol derivatives, linked to salicylaldehyde moieties that coordinate with trivalent metal ions like Cobalt (Co), Chromium (Cr), Iron (Fe), or Aluminum (Al). This coordination creates a rigid chiral environment around the metal center, which is essential for distinguishing between the enantiomers of the racemic epoxide substrate during the ring-opening step. The presence of specific counter-ions, such as fluoride (F-), chloride (Cl-), or trifluoroacetate (CF3CO2-), further modulates the Lewis acidity of the metal center, optimizing the activation of the epoxide ring for nucleophilic attack by the carbonate species generated from CO2 and the cocatalyst.
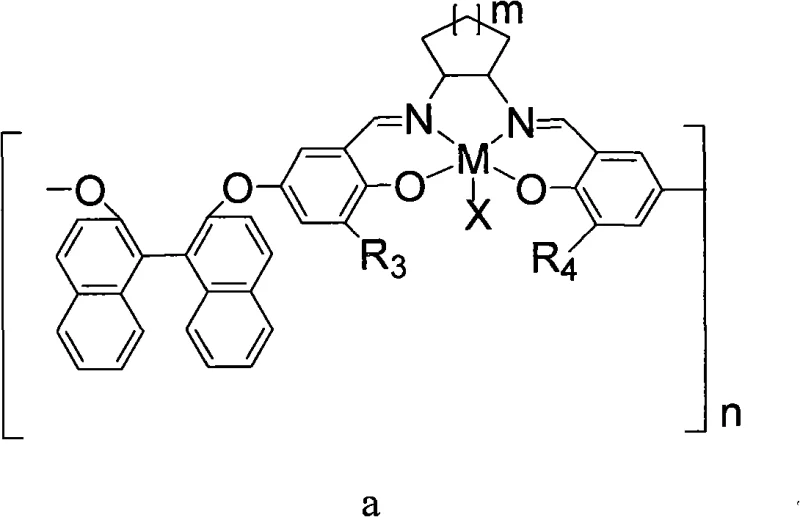
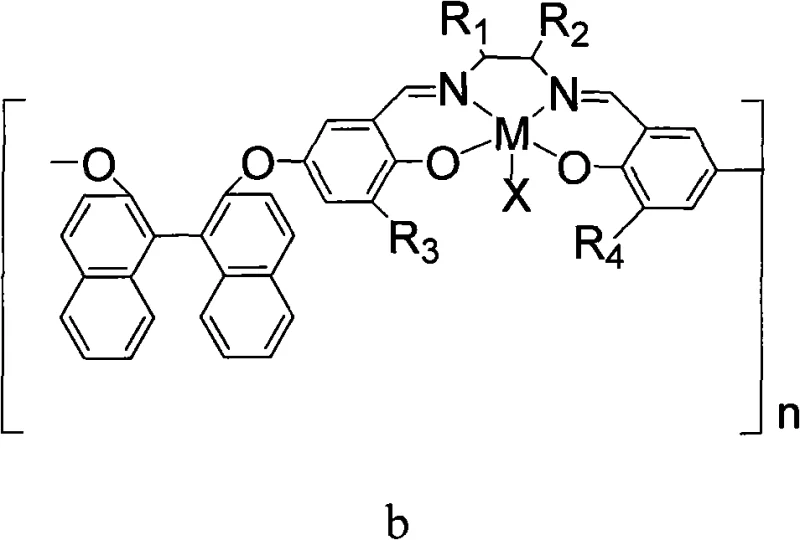
From an impurity control perspective, the polymeric nature of the catalyst offers a distinct advantage by minimizing metal leaching into the final product, a critical parameter for high-purity pharmaceutical intermediates. In traditional homogeneous catalysis, trace metal residues often require additional scavenging steps to meet regulatory limits, adding complexity and cost. However, the insolubility of the polymeric catalyst in the reaction medium ensures that the metal remains bound to the solid support throughout the reaction cycle. Furthermore, the kinetic resolution mechanism inherently enriches the unreacted epoxide enantiomer, providing a dual output of valuable chiral materials from a single racemic starting material. The precise tuning of the cocatalyst, typically a quaternary ammonium salt like tetrabutylammonium fluoride, ensures that the ring-closing step proceeds rapidly without racemization, thereby preserving the optical integrity of the synthesized cyclic carbonate throughout the process.
How to Synthesize Optically Active Cyclocarbonate Efficiently
The synthesis protocol outlined in the patent provides a robust framework for producing these valuable chiral intermediates with high reproducibility. The process begins with the preparation of the chiral polymer ligand through a series of well-defined organic transformations, including Grignard reactions and etherification, followed by complexation with metal salts under inert atmosphere conditions. Once the catalyst is prepared, the cycloaddition reaction is conducted in a pressurized vessel where racemic epoxide and CO2 are introduced in the presence of the cocatalyst. The detailed standardized synthesis steps see the guide below, which outlines the specific molar ratios, temperature profiles, and workup procedures required to achieve optimal yields and enantiomeric excess values.
- Preparation of the chiral polymer ligand via etherification of brominated salicylaldehyde derivatives with chiral binaphthol or diamine backbones.
- Complexation of the polymer ligand with trivalent metal salts (such as Co, Cr, Fe) followed by oxidation to generate the active trivalent metal species.
- Execution of the cycloaddition reaction between racemic epoxide and CO2 under mild pressure (0.1-2 MPa) using a quaternary ammonium salt cocatalyst.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this polymeric catalyst technology translates into tangible operational efficiencies and risk mitigation strategies. The ability to recover and reuse the catalyst multiple times without significant degradation in performance directly impacts the cost of goods sold (COGS) by amortizing the initial catalyst investment over a much larger production volume. This contrasts sharply with single-use homogeneous catalysts where the cost of the chiral ligand and metal salt is incurred for every batch produced. Moreover, the simplified workup procedure, which relies on filtration rather than chromatography, reduces the consumption of solvents and silica gel, leading to substantial waste reduction and lower disposal costs. These factors collectively contribute to a more resilient supply chain capable of meeting fluctuating market demands without compromising on margin.
- Cost Reduction in Manufacturing: The implementation of this polymeric catalyst system drives cost optimization primarily through the elimination of expensive downstream purification processes. Since the catalyst can be separated via simple precipitation and filtration, the need for resource-intensive chromatographic columns or complex distillation setups is removed. Additionally, the high turnover frequency and reusability of the catalyst mean that the effective catalyst loading per kilogram of product decreases significantly over time. This structural advantage allows manufacturers to achieve competitive pricing structures for optically active cyclic carbonates, making them more accessible for downstream applications in polymer and pharmaceutical synthesis without sacrificing quality.
- Enhanced Supply Chain Reliability: Supply continuity is bolstered by the robustness of the catalyst synthesis and the stability of the reaction conditions. The raw materials required for the catalyst, such as substituted salicylaldehydes and chiral diamines, are commercially available and do not rely on exotic or geopolitically sensitive resources. Furthermore, the reaction tolerates a broad range of operating pressures and temperatures, providing flexibility in plant operations and reducing the risk of batch failures due to minor process deviations. This reliability ensures that a reliable pharmaceutical intermediates supplier can maintain consistent delivery schedules, crucial for clients managing just-in-time inventory systems for critical drug substances.
- Scalability and Environmental Compliance: Scaling this process from laboratory to commercial production is facilitated by the heterogeneous nature of the catalyst, which behaves predictably in larger reactors. The use of carbon dioxide as a feedstock not only utilizes a greenhouse gas but also avoids the generation of stoichiometric byproducts associated with phosgene-based carbonate synthesis routes. The reduced solvent usage and the absence of heavy metal contamination in the product stream simplify wastewater treatment and ensure compliance with stringent environmental regulations. This alignment with green chemistry principles enhances the corporate sustainability profile of manufacturers adopting this technology.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and performance of this catalytic system. These insights are derived directly from the experimental data and embodiments provided in the patent documentation, ensuring that the information reflects the actual capabilities and limitations of the technology. Understanding these nuances is essential for R&D teams evaluating the feasibility of integrating this process into their existing manufacturing workflows.
Q: What are the advantages of polymeric chiral catalysts over small molecule counterparts?
A: Polymeric catalysts offer superior recoverability and reusability. Unlike small molecule catalysts which often require complex chromatographic separation, polymeric variants can be precipitated and filtered, allowing for reuse over 10 cycles without significant loss in enantioselectivity or activity.
Q: How does temperature affect the enantiomeric excess (e.e.) in this process?
A: Temperature plays a critical role in kinetic resolution. Lower temperatures generally favor higher optical purity. For instance, reaction data indicates that while yields may decrease at -20°C compared to 25°C, the e.e. value can significantly increase, reaching levels above 85% under optimized cold conditions.
Q: Which metal centers are most effective for this cycloaddition reaction?
A: The patent discloses a wide range of trivalent metals including Cobalt (Co), Chromium (Cr), Iron (Fe), and Aluminum (Al). Cobalt complexes, particularly those with specific counter-ions like trifluoroacetate or fluoride, have demonstrated robust activity and selectivity profiles suitable for industrial application.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Optically Active Cyclocarbonate Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic technologies like the polymeric chiral system described in CN101270112A for the production of high-value chiral intermediates. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from bench-scale discovery to full-scale manufacturing is seamless and efficient. Our facilities are equipped with state-of-the-art high-pressure reactors and rigorous QC labs capable of handling sensitive chiral chemistries, guaranteeing that every batch meets stringent purity specifications required by global regulatory bodies. We are committed to delivering consistent quality and supply security for your critical projects.
We invite you to collaborate with our technical team to explore how this innovative catalyst technology can be tailored to your specific synthetic needs. By engaging with us, you gain access to a Customized Cost-Saving Analysis that evaluates the economic benefits of switching to this reusable catalyst system for your specific product portfolio. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise can accelerate your path to market while optimizing your production costs.
