Scalable Synthesis of 5-Trifluoromethyl-1,2,4-Triazoles for Advanced Pharmaceutical Intermediates
Scalable Synthesis of 5-Trifluoromethyl-1,2,4-Triazoles for Advanced Pharmaceutical Intermediates
The introduction of trifluoromethyl groups into heterocyclic scaffolds is a cornerstone strategy in modern medicinal chemistry, significantly enhancing the electronegativity, metabolic stability, and lipophilicity of drug candidates. Patent CN110467579B discloses a groundbreaking preparation method for 5-trifluoromethyl substituted 1,2,4-triazole compounds, addressing critical bottlenecks in the synthesis of these valuable pharmaceutical intermediates. As a leading entity in fine chemical manufacturing, we recognize that the ability to access these fluorinated motifs efficiently is paramount for developing next-generation therapeutics. The disclosed technology leverages a non-metallic iodine-promoted cyclization strategy that operates under remarkably mild conditions, bypassing the stringent requirements often associated with fluorination chemistry. This innovation not only streamlines the synthetic route but also aligns perfectly with the industry's demand for greener, more cost-effective manufacturing processes.
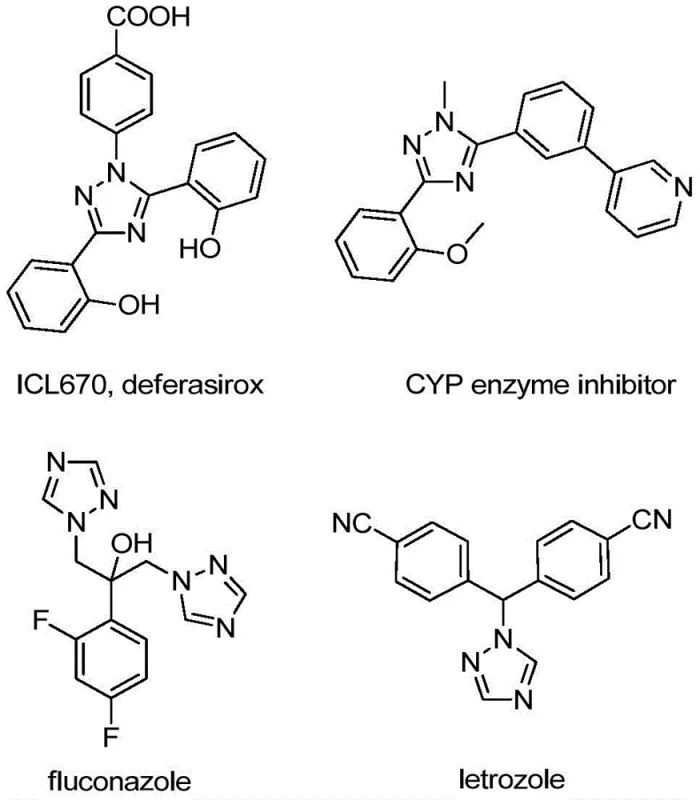
The significance of the 1,2,4-triazole ring cannot be overstated, as it serves as a privileged scaffold in numerous bioactive molecules, ranging from antifungal agents like fluconazole to iron chelators like deferasirox. By integrating a trifluoromethyl group at the 5-position, chemists can fine-tune the pharmacokinetic profiles of these molecules, often resulting in improved oral bioavailability and prolonged half-life in vivo. The method described in CN110467579B provides a robust platform for generating diverse libraries of these compounds, enabling rapid structure-activity relationship (SAR) studies. For R&D teams focused on oncology or infectious diseases, having access to a reliable supply of high-purity 5-trifluoromethyl-1,2,4-triazoles is a strategic advantage that accelerates lead optimization campaigns.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of trifluoromethyl-substituted nitrogen heterocycles has been fraught with challenges that hinder efficient cost reduction in API manufacturing. Traditional approaches often rely on the direct trifluoromethylation of pre-synthesized heterocycles, which necessitates the use of specialized and frequently expensive reagents such as Togni reagents or Umemoto reagents. Alternatively, methods employing trifluorodiazoethane as a synthon are plagued by safety concerns due to the explosive nature of diazo compounds, requiring specialized equipment and rigorous safety protocols that drive up operational costs. Furthermore, many existing protocols depend on transition metal catalysis, which introduces the risk of heavy metal contamination in the final product, necessitating complex and costly purification steps to meet regulatory limits for pharmaceutical ingredients.
The Novel Approach
In stark contrast, the novel approach detailed in the patent utilizes readily available trifluoroethylimidoyl chloride and hydrazones as starting materials, catalyzed by inexpensive elemental iodine. This methodology represents a paradigm shift towards atom economy and operational simplicity. The reaction proceeds smoothly in common organic solvents like 1,2-dichloroethane (DCE) at moderate temperatures around 80°C, eliminating the need for cryogenic conditions or inert atmospheres. By avoiding precious metal catalysts and hazardous diazo species, this route significantly lowers the barrier to entry for producing these complex intermediates. The broad substrate tolerance allows for the incorporation of various functional groups, including halogens, alkyls, and alkoxy groups, providing medicinal chemists with the flexibility to explore diverse chemical spaces without redesigning the entire synthetic pathway.
Mechanistic Insights into Iodine-Promoted Cyclization
The mechanistic pathway of this transformation is a fascinating example of oxidative heterocycle construction. The reaction likely initiates with a base-promoted intermolecular carbon-nitrogen bond formation between the trifluoroethylimidoyl chloride and the hydrazone, yielding a trifluoroacetamidine intermediate. This species subsequently undergoes isomerization, setting the stage for the critical cyclization event. The addition of elemental iodine acts as a mild oxidant, facilitating an oxidative iodination that generates a reactive iodine-containing intermediate. This intermediate then undergoes an intramolecular electrophilic substitution, followed by spontaneous aromatization to furnish the final 5-trifluoromethyl-1,2,4-triazole core. Understanding this mechanism is crucial for process chemists aiming to optimize reaction parameters and minimize impurity formation during scale-up.
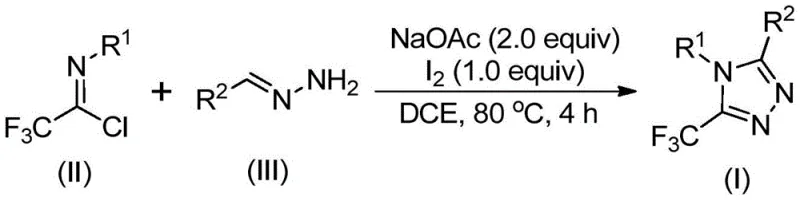
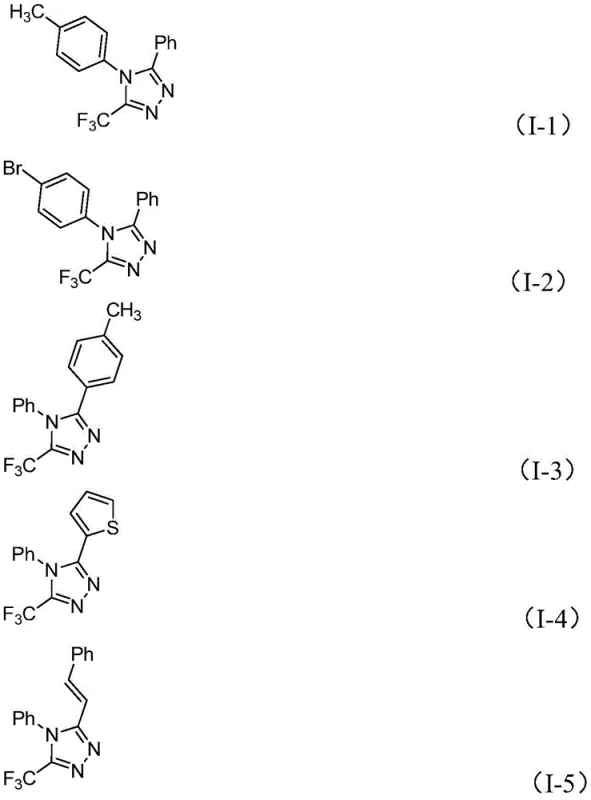
From an impurity control perspective, the mildness of the iodine promotion is a significant advantage. Harsh oxidative conditions often lead to over-oxidation or decomposition of sensitive functional groups on the aromatic rings. However, the controlled release of oxidative potential by molecular iodine in the presence of sodium acetate ensures high chemoselectivity. The patent data indicates excellent yields across a range of substrates, with electron-rich and electron-deficient aryl groups both performing well. For instance, substrates bearing methyl, methoxy, or bromo substituents on the phenyl ring afford the corresponding triazoles in high yields, demonstrating the robustness of the catalytic system. This reliability is essential for maintaining consistent quality in the production of high-purity pharmaceutical intermediates.
How to Synthesize 5-Trifluoromethyl-1,2,4-Triazoles Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires careful attention to stoichiometry and addition sequences to maximize yield and purity. The protocol involves a two-stage heating process where the initial condensation is followed by the oxidative cyclization. Detailed standard operating procedures regarding exact molar ratios, solvent volumes, and workup techniques are critical for reproducibility. For a comprehensive guide on executing this reaction with optimal results, please refer to the standardized synthesis steps outlined below.
- Mix sodium acetate, trifluoroethylimidoyl chloride, and hydrazone in an organic solvent such as dichloroethane (DCE).
- Heat the reaction mixture to 80°C and stir for 2 to 4 hours to allow initial cyclization.
- Add elemental iodine to the system and continue heating for an additional 1 to 2 hours to promote oxidative aromatization.
- Filter the mixture, mix with silica gel, and purify via column chromatography to isolate the final triazole product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers tangible benefits that extend beyond mere chemical elegance. The elimination of exotic reagents and precious metal catalysts translates directly into a more resilient and cost-efficient supply chain. By relying on commodity chemicals like hydrazones and imidoyl chlorides, manufacturers can mitigate the risks associated with sourcing specialized reagents that may have long lead times or volatile pricing. Furthermore, the simplified workup procedure reduces the consumption of silica gel and solvents during purification, contributing to substantial cost savings in waste management and raw material usage.
- Cost Reduction in Manufacturing: The absence of heavy metal catalysts such as palladium or copper removes the necessity for expensive scavenging resins and rigorous metal testing, which are significant cost drivers in API production. Additionally, the use of elemental iodine, a low-cost promoter, compared to sophisticated organometallic complexes, drastically lowers the catalyst cost per kilogram of product. The mild reaction conditions also imply lower energy consumption for heating and cooling, further enhancing the overall economic viability of the process for large-scale commercial scale-up of complex pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: The starting materials, specifically the hydrazones and trifluoroethylimidoyl chlorides, are derived from widely available aldehydes and amines, ensuring a stable and continuous supply stream. This reduces the dependency on single-source suppliers for niche reagents, thereby minimizing the risk of production delays due to raw material shortages. The robustness of the reaction against moisture and oxygen means that storage and handling requirements are less stringent, simplifying logistics and warehousing operations for the manufacturing facility.
- Scalability and Environmental Compliance: The process is inherently scalable, as demonstrated by its successful expansion to gram levels in the patent examples, with clear potential for multi-kilogram production. The avoidance of toxic heavy metals aligns with increasingly strict environmental regulations regarding heavy metal discharge and residual limits in pharmaceuticals. This green chemistry profile facilitates easier regulatory approval and reduces the environmental footprint of the manufacturing site, supporting corporate sustainability goals while ensuring reducing lead time for high-purity intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding this synthesis method, derived from the specific advantages and operational details found in the patent literature. These insights are intended to clarify the feasibility and benefits of adopting this technology for your specific project needs.
Q: What are the advantages of this iodine-promoted method over traditional trifluoromethylation?
A: This method avoids the use of expensive and potentially hazardous trifluoromethylation reagents or unstable trifluorodiazoethane. It utilizes cheap, commercially available hydrazones and trifluoroethylimidoyl chloride under mild conditions without requiring strict anhydrous or anaerobic environments.
Q: Does this synthesis require heavy metal catalysts?
A: No, the process is promoted by non-metallic elemental iodine. This eliminates the need for costly transition metal catalysts like palladium or copper, simplifying downstream purification and reducing heavy metal residue risks in the final API.
Q: Is this process suitable for large-scale manufacturing?
A: Yes, the patent explicitly states the method can be easily expanded to the gram level and beyond. The use of common solvents like DCE and the tolerance for ambient conditions make it highly amenable to commercial scale-up for industrial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Trifluoromethyl-1,2,4-Triazole Supplier
At NINGBO INNO PHARMCHEM, we understand that the transition from bench-scale discovery to commercial production requires a partner with deep technical expertise and robust infrastructure. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can move seamlessly from clinical trials to market launch. We maintain stringent purity specifications and operate rigorous QC labs equipped with advanced analytical instrumentation to guarantee that every batch of 5-trifluoromethyl-1,2,4-triazole meets the highest industry standards for potency and impurity profiles.
We invite you to collaborate with us to leverage this innovative synthetic technology for your drug development programs. Our technical sales team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you optimize your supply chain for success.
