Scalable Electrochemical Synthesis of Beta-Acetaminocarbonyl Compounds for Commercial Pharmaceutical Production
Introduction to Green Electrochemical Manufacturing
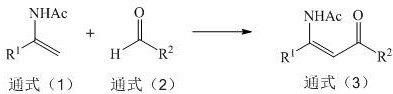
The pharmaceutical and fine chemical industries are currently undergoing a paradigm shift towards sustainable manufacturing technologies, driven by the urgent need to reduce environmental footprints and optimize production costs. A pivotal development in this sector is detailed in Chinese Patent CN113373466A, which discloses a novel electrochemical synthesis method for preparing beta-acetaminocarbonyl compounds. These compounds serve as critical building blocks in the synthesis of chiral beta-amino acids and various drug molecules, traditionally requiring complex and hazardous synthetic routes. The patented technology leverages the power of electrons as clean reagents to facilitate the oxidative cross-coupling of enamides with formates or aldehydes, effectively bypassing the need for stoichiometric chemical oxidants or transition metal catalysts. This breakthrough not only enhances the atom economy of the reaction but also drastically simplifies the downstream processing required to achieve high-purity standards essential for pharmaceutical applications. By utilizing electricity to drive the redox potential, this method offers a robust alternative to conventional thermal chemistry, promising significant improvements in safety and operational efficiency for manufacturers of high-value organic intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of beta-acetaminocarbonyl compounds has relied heavily on transition metal catalysis or the use of hazardous nitrogen-containing reagents, presenting substantial challenges for large-scale production. As illustrated in the comparative reaction schemes, traditional pathways often involve the preparation of unstable hydrazine carboxylates, which require multi-step syntheses starting from toxic hydrazine hydrate and dimethyl carbonate. Furthermore, established methods frequently employ expensive iron-porphyrin complexes or other transition metal catalysts to facilitate C-H bond activation or alkoxycarbonylation, introducing the risk of heavy metal contamination in the final product. The removal of these metal residues necessitates additional purification steps, such as specialized scavenging or recrystallization, which increases both the processing time and the overall cost of goods sold. Additionally, the use of strong chemical oxidants in these conventional routes generates significant amounts of hazardous waste, complicating compliance with increasingly stringent environmental regulations regarding "three wastes" discharge. The complexity of controlling selectivity in these thermal reactions often leads to lower yields and the formation of difficult-to-separate by-products, further eroding the economic viability of the process for commercial scale-up.

The Novel Approach
In stark contrast to these legacy methods, the electrochemical synthesis described in the patent utilizes a direct anodic oxidation strategy that fundamentally simplifies the reaction manifold. By employing a constant current electrolysis setup with inexpensive electrode materials such as platinum sheets or graphite rods, the system generates reactive radical intermediates in situ without the addition of external oxidizing agents. This approach allows for the direct coupling of readily available enamide substrates with simple formates or aldehydes under mild conditions, typically ranging from 25°C to 50°C. The absence of transition metal catalysts means that the reaction mixture is inherently cleaner, reducing the burden on downstream purification and minimizing the risk of metal leaching into the product stream. Moreover, the electrochemical potential can be precisely tuned to favor the desired oxidative cross-coupling pathway, thereby enhancing regioselectivity and suppressing side reactions that commonly plague thermal methods. This technological leap transforms the synthesis from a complex, multi-variable chemical process into a more controllable physical-chemical operation, where the rate of reaction is governed by the applied current rather than the concentration of unstable reagents.
Mechanistic Insights into Electrochemical Oxidative Cross-Coupling
The core mechanism of this transformation relies on the anodic oxidation of the enamide substrate to generate a reactive cation radical species, which subsequently undergoes nucleophilic attack by the formate or aldehyde derivative. In the electrochemical cell, the anode serves as the source of electrons, abstracting an electron from the pi-system of the enamide to initiate the catalytic cycle. This electron transfer process is highly efficient and occurs at the electrode surface, creating a localized high-energy environment that drives the formation of new carbon-carbon bonds without the need for thermal activation energies that might degrade sensitive functional groups. The supporting electrolyte, such as tetrabutylammonium tetrafluoroborate or ammonium perchlorate, plays a crucial role in maintaining conductivity and stabilizing the charged intermediates within the acetonitrile solvent system. Unlike chemical oxidants which are consumed stoichiometrically and produce equivalent amounts of reduced waste, the "electron" acts as a traceless reagent, ensuring that the only by-products are typically hydrogen gas at the cathode or benign protonated species. This mechanistic elegance ensures that the reaction proceeds with high atom economy, as nearly all atoms from the starting materials are incorporated into the final beta-acetaminocarbonyl structure.
From an impurity control perspective, the electrochemical method offers superior selectivity profiles compared to free-radical initiators used in thermal chemistry. In traditional radical reactions, uncontrolled chain propagation often leads to polymerization of the enamide or over-oxidation of the aldehyde component, resulting in complex impurity profiles that are difficult to characterize. However, in the electrochemical regime, the flux of electrons is strictly regulated by the galvanostat, limiting the concentration of radical intermediates to a steady-state level that favors the cross-coupling event over homocoupling or decomposition. The mild reaction temperatures further suppress thermal degradation pathways, preserving the integrity of the acetamido group and preventing hydrolysis or elimination side reactions. Consequently, the crude reaction mixtures obtained from this process exhibit higher purity, often requiring only standard column chromatography or crystallization to meet the stringent specifications required for pharmaceutical intermediates. This inherent cleanliness of the electrochemical mechanism translates directly into reduced solvent consumption for purification and higher overall process yields, validating its utility for the synthesis of high-value chiral precursors.
How to Synthesize Beta-Acetaminocarbonyl Compounds Efficiently
Implementing this electrochemical protocol requires careful attention to cell configuration and parameter optimization to ensure reproducibility and safety during operation. The patent outlines a straightforward procedure where the enamide and coupling partner are dissolved in a conductive solvent system containing a supporting electrolyte, creating a homogeneous reaction medium suitable for electrolysis. Operators must select appropriate electrode materials, with platinum serving as a robust anode for oxidation and a compatible cathode for reduction, maintaining a fixed inter-electrode distance to minimize cell resistance. The reaction is driven by a constant current power supply, typically set between 10 mA and 15 mA for laboratory-scale batches, ensuring a steady rate of electron transfer that matches the kinetics of the chemical transformation. Detailed standardized operating procedures regarding electrode preparation, solvent drying, and workup protocols are essential for transferring this technology from the bench to pilot plant scales.
- Prepare the electrolytic cell by dissolving supporting electrolyte (NH4ClO4 or Bu4NBF4) in acetonitrile or acetonitrile/water mixture within a three-necked flask.
- Add the enamide substrate (0.3 mmol) and the formate or aldehyde coupling partner (0.9-1.2 mmol) to the solution, then insert platinum or graphite electrodes.
- Apply a constant current of 10-15 mA at 25-50°C for 5-6 hours, followed by aqueous quenching, extraction, and column chromatography purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this electrochemical synthesis route presents a compelling value proposition centered on cost stability and operational simplicity. By eliminating the dependency on volatile and often expensive transition metal catalysts, manufacturers can decouple their production costs from the fluctuating market prices of precious metals like palladium or specialized iron complexes. Furthermore, the removal of hazardous hydrazine derivatives from the bill of materials significantly reduces the regulatory burden associated with the storage, handling, and disposal of toxic substances, thereby lowering insurance and compliance costs. The simplified workflow, which combines reaction and oxidation into a single step, shortens the overall manufacturing cycle time, allowing for faster turnover of inventory and improved responsiveness to market demand fluctuations. This streamlined process also reduces the consumption of auxiliary chemicals and solvents needed for extensive purification, contributing to a leaner and more cost-effective supply chain operation.
- Cost Reduction in Manufacturing: The most significant economic benefit arises from the complete elimination of stoichiometric oxidants and expensive catalyst systems, which traditionally account for a substantial portion of raw material costs in fine chemical synthesis. Without the need to purchase and subsequently remove heavy metal catalysts, the downstream processing becomes markedly cheaper, as expensive scavenging resins or multiple recrystallization steps are no longer required to meet residual metal limits. Additionally, the use of electricity as the primary driving force is generally more energy-efficient and cost-predictable than heating large reactors to high temperatures for extended periods, leading to lower utility expenses per kilogram of product. The high atom efficiency of the electrochemical coupling ensures that raw material utilization is maximized, minimizing the waste of valuable enamide substrates and further driving down the variable cost of production.
- Enhanced Supply Chain Reliability: Sourcing complex catalysts and hazardous reagents like hydrazine hydrate often involves long lead times and strict regulatory controls that can disrupt production schedules. By shifting to a methodology that relies on commodity chemicals such as acetonitrile, simple salts, and basic aldehydes, the supply chain becomes far more resilient to external shocks and vendor shortages. The robustness of the electrochemical equipment, which consists of standard power supplies and inert electrodes, reduces the risk of unplanned downtime caused by catalyst deactivation or reactor fouling common in heterogeneous catalysis. This reliability ensures a consistent flow of high-purity intermediates to downstream customers, strengthening the manufacturer's reputation as a dependable partner in the pharmaceutical value chain.
- Scalability and Environmental Compliance: The electrochemical process is inherently scalable through the use of filter-press cells or continuous flow reactors, allowing for seamless expansion from gram-scale R&D to tonne-scale commercial production without re-optimizing the chemistry. From an environmental standpoint, the drastic reduction in chemical waste generation aligns perfectly with global sustainability goals and corporate ESG mandates, facilitating easier permitting and community acceptance of manufacturing facilities. The absence of toxic heavy metals in the effluent simplifies wastewater treatment protocols, reducing the capital expenditure required for environmental protection infrastructure. This green chemistry profile not only mitigates regulatory risk but also enhances the marketability of the final product to environmentally conscious pharmaceutical clients seeking sustainable sourcing options.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this electrochemical technology, derived directly from the patent specifications and comparative data. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this route into existing manufacturing portfolios. The answers provided reflect the specific advantages of the electron-mediated mechanism over traditional thermal and catalytic methods, highlighting the practical benefits for industrial application.
Q: What are the primary advantages of this electrochemical method over traditional iron-catalyzed routes?
A: The electrochemical method eliminates the need for expensive transition metal catalysts like Iron-Porphyrin complexes and toxic hydrazine derivatives, significantly simplifying the purification process and reducing heavy metal contamination risks in the final API intermediate.
Q: Is this process scalable for industrial production of pharmaceutical intermediates?
A: Yes, the process operates under mild conditions (25-50°C, ambient pressure) using simple undivided cells, making it highly amenable to continuous flow electrochemistry and large-scale commercial manufacturing without complex high-pressure equipment.
Q: What represents the main cost saving driver in this synthetic route?
A: The primary cost drivers are removed by avoiding the multi-step synthesis of hazardous hydrazine precursors and eliminating the procurement and subsequent removal of precious or transition metal catalysts, leading to substantial raw material and waste treatment savings.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Beta-Acetaminocarbonyl Compounds Supplier
As the global demand for chiral pharmaceutical intermediates continues to rise, the ability to deliver high-quality building blocks through sustainable and efficient processes is more critical than ever. NINGBO INNO PHARMCHEM stands at the forefront of this innovation, leveraging advanced electrochemical technologies to manufacture beta-acetaminocarbonyl compounds with exceptional purity and consistency. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the rigorous volume requirements of multinational pharmaceutical companies. We maintain stringent purity specifications through our rigorous QC labs, utilizing state-of-the-art analytical instrumentation to verify that every batch meets the exacting standards required for API synthesis, thereby guaranteeing the safety and efficacy of the final drug products.
We invite potential partners to engage with our technical procurement team to discuss how this novel electrochemical route can be tailored to your specific project needs. By collaborating with us, you can access a Customized Cost-Saving Analysis that quantifies the economic benefits of switching from traditional catalytic methods to our green electrochemical platform. We encourage you to contact us today to request specific COA data and route feasibility assessments, allowing you to make informed decisions that optimize your supply chain and accelerate your drug development timelines.
