Advanced Copper-Catalyzed Synthesis of Benzofuropyridine Intermediates for Commercial Pharmaceutical Manufacturing
Introduction to Next-Generation Heterocyclic Synthesis
The pharmaceutical industry continuously seeks robust and scalable methodologies for constructing complex heterocyclic scaffolds that serve as the backbone for novel therapeutic agents. A recent breakthrough detailed in patent CN112574223B introduces a highly efficient copper-catalyzed strategy for synthesizing 1,2-dihydrobenzofuropyridine compounds, which are pivotal precursors to bioactive molecules with Topoisomerase inhibitory activity. This innovation addresses critical bottlenecks in traditional synthetic routes by replacing expensive noble metal catalysts with an earth-abundant copper system, thereby aligning with modern green chemistry principles while maintaining exceptional reaction efficiency. The disclosed method leverages a tandem addition/cyclization/isomerization sequence that transforms aurone-derived azadienes and terminal alkynes into valuable polycyclic structures with yields reaching up to 99 percent. For R&D directors and procurement specialists, this represents a paradigm shift towards more sustainable and cost-effective manufacturing of high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of dihydropyridine and benzofuropyridine cores has relied heavily on strategies involving condensation reactions of amines with carbonyl compounds or partial reductions of pyridine derivatives. These conventional pathways often suffer from significant drawbacks, including the requirement for harsh reaction conditions that can compromise sensitive functional groups present in complex drug candidates. Furthermore, many established protocols depend on noble metal catalysts which not only inflate raw material costs but also introduce stringent regulatory hurdles regarding residual metal limits in final active pharmaceutical ingredients. The limited substrate scope of older methods frequently necessitates extensive protecting group manipulations, leading to longer synthetic sequences, lower overall atom economy, and increased waste generation. Such inefficiencies create substantial barriers for supply chain heads aiming to secure reliable sources of key intermediates at a competitive price point.
The Novel Approach
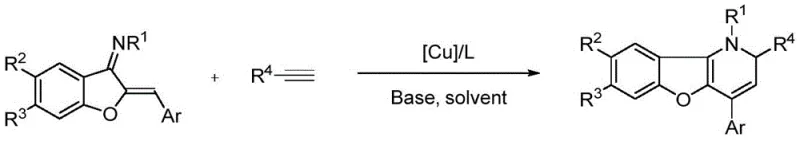
In stark contrast, the novel copper-catalyzed methodology described in the patent data offers a streamlined and versatile alternative that overcomes these historical limitations. By utilizing a catalytic system composed of a copper precursor and a bidentate phosphine ligand, specifically XantPhos, the reaction proceeds smoothly at a mild temperature of 60 degrees Celsius in 1,4-dioxane. This approach enables a direct tandem transformation where the azadiene and alkyne undergo addition, cyclization, and isomerization in a single operational step, drastically reducing processing time and solvent usage. The versatility of this system is demonstrated by its broad tolerance for various substituents on both the azadiene and alkyne components, allowing for the rapid generation of diverse chemical libraries. As illustrated in the reaction scheme below, this method provides direct access to the 1,2-dihydrobenzofuropyridine core, which can be subsequently aromatized to yield the fully conjugated benzofuropyridine system.
Mechanistic Insights into Copper-Catalyzed Tandem Cyclization
The success of this synthetic route hinges on the precise interplay between the copper center and the specialized ligand environment, which facilitates the activation of the terminal alkyne and the subsequent nucleophilic attack on the azadiene. Mechanistic studies suggest that the copper species coordinates with the alkyne to increase its electrophilicity, promoting a regioselective addition to the electron-deficient azadiene double bond. The choice of ligand is paramount; screening data indicates that bulky, bite-angle ligands like XantPhos significantly outperform monodentate phosphines or nitrogen-based ligands in terms of yield and reaction rate. This ligand effect stabilizes the active catalytic species and prevents the formation of inactive copper aggregates, ensuring consistent performance across different substrate combinations. The mild basic conditions provided by triethylamine further assist in the proton transfer steps required for the isomerization phase, driving the equilibrium towards the desired dihydro-product without degrading sensitive moieties.
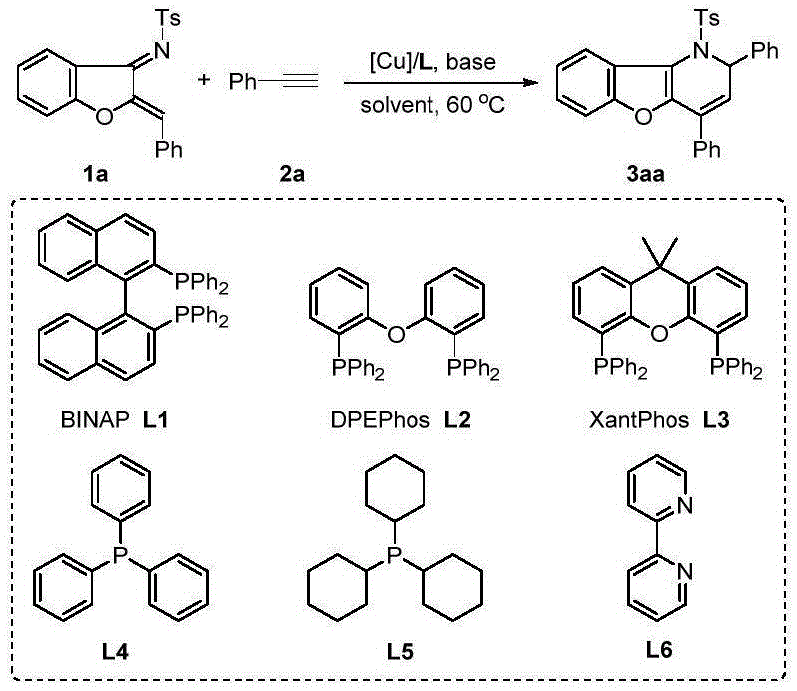
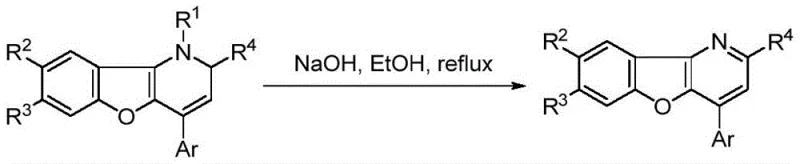
From an impurity control perspective, the high selectivity of this copper-catalyzed system minimizes the formation of side products such as homocoupling dimers or polymerization byproducts that often plague alkyne chemistry. The reaction's reliance on the strong driving force of aromaticity recovery during the cyclization step ensures high conversion rates, often exceeding 95 percent for a wide range of substrates including those with electron-withdrawing or electron-donating groups. This robustness is crucial for maintaining a clean impurity profile, which simplifies downstream purification and reduces the burden on quality control laboratories. Furthermore, the ability to perform a subsequent aromatization step under simple alkaline reflux conditions allows manufacturers to access the fully aromatic benzofuropyridine scaffold, a structure known for its stability and biological relevance, directly from the initial tandem reaction product.
How to Synthesize 1,2-Dihydrobenzofuropyridine Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires careful attention to the stoichiometry and order of addition to maximize the efficiency of the catalytic cycle. The standard protocol involves pre-mixing the copper precursor, typically Cu(CH3CN)4BF4, with the XantPhos ligand in the solvent to generate the active catalyst in situ before introducing the substrates. Maintaining an inert atmosphere is recommended to prevent oxidation of the copper(I) species, although the system demonstrates reasonable stability under the described conditions. Following the tandem reaction, the crude 1,2-dihydrobenzofuropyridine can be isolated via standard column chromatography or crystallization techniques depending on the scale. For applications requiring the aromatic variant, a simple treatment with sodium hydroxide in ethanol under reflux completes the transformation, offering a flexible two-step-one-pot potential for process optimization.
- Prepare the catalytic system by mixing a copper precursor such as Cu(CH3CN)4BF4 with a diphosphine ligand like XantPhos in an organic solvent.
- Add the aurone-derived azadiene substrate, terminal alkyne, and a base such as triethylamine to the reaction mixture.
- Heat the mixture to 60°C in 1,4-dioxane to facilitate the addition/cyclization/isomerization tandem reaction, followed by optional aromatization under alkaline conditions.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this copper-catalyzed technology presents a compelling value proposition centered on cost reduction and supply security. The substitution of scarce and volatile noble metals with abundant copper salts fundamentally alters the cost structure of the synthesis, eliminating the need for expensive catalyst recovery systems and reducing the risk of supply disruptions associated with precious metal markets. Moreover, the use of commodity chemicals such as 1,4-dioxane and triethylamine ensures that raw material sourcing remains straightforward and resilient against geopolitical fluctuations. The high yields reported across a broad substrate scope mean that less starting material is wasted, directly improving the mass balance and reducing the environmental footprint of the manufacturing process, which is increasingly a criterion for vendor selection in the pharmaceutical sector.
- Cost Reduction in Manufacturing: The elimination of precious metal catalysts removes a significant line item from the bill of materials, while the mild reaction conditions reduce energy consumption associated with heating and cooling cycles. Simplified workup procedures due to high selectivity further lower labor and solvent costs, contributing to a more economical overall process that enhances margin potential for high-volume production.
- Enhanced Supply Chain Reliability: By relying on widely available copper salts and commercially accessible ligands, manufacturers can diversify their supplier base and avoid single-source dependencies. The robustness of the reaction against variations in substrate electronics ensures consistent output quality even when sourcing raw materials from different vendors, thereby stabilizing lead times and inventory planning.
- Scalability and Environmental Compliance: The operation at atmospheric pressure and moderate temperatures simplifies reactor design requirements, allowing for easier scale-up from kilogram to tonnage quantities without extensive re-engineering. Additionally, the reduced toxicity profile compared to heavy metal alternatives facilitates waste disposal and helps facilities meet increasingly stringent environmental regulations regarding heavy metal discharge.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and scope defined in the patent literature, providing a factual basis for evaluating the feasibility of this route for your specific project needs. Understanding these details is essential for making informed decisions about process adoption and vendor qualification.
Q: What are the advantages of using copper catalysis over noble metals for benzofuropyridine synthesis?
A: Copper catalysis offers significant cost advantages due to the abundance of copper compared to precious metals like palladium or rhodium. Additionally, the copper/XantPhos system operates under milder conditions (60°C) with excellent functional group tolerance, reducing energy consumption and simplifying purification processes.
Q: Can this synthesis method be scaled for industrial production?
A: Yes, the method utilizes readily available starting materials and standard solvents like 1,4-dioxane. The mild reaction temperature and high yields (up to 99%) indicate strong potential for commercial scale-up without the safety hazards associated with high-pressure or high-temperature noble metal catalysis.
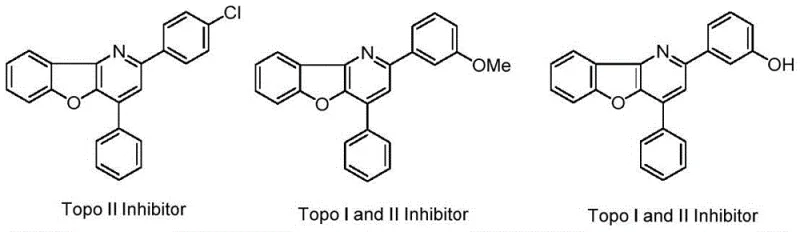
Q: What is the biological significance of the synthesized benzofuropyridine compounds?
A: The resulting benzofuropyridine scaffolds are valuable intermediates for developing Topoisomerase I and II inhibitors. These compounds exhibit potential cytotoxicity against human cancer cells, making them critical building blocks for oncology drug discovery pipelines.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Benzofuropyridine Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of efficient heterocyclic synthesis in accelerating drug discovery timelines. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from bench-scale optimization to industrial manufacturing is seamless and compliant. We maintain stringent purity specifications and operate rigorous QC labs equipped to handle complex impurity profiling, guaranteeing that every batch of benzofuropyridine intermediate meets the exacting standards required for clinical and commercial applications. Our commitment to quality and reliability makes us an ideal partner for organizations seeking to secure their supply chain for oncology research materials.
We invite you to engage with our technical procurement team to discuss how this advanced copper-catalyzed route can be tailored to your specific molecular targets. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits of switching to this methodology. We encourage you to contact us today to obtain specific COA data and route feasibility assessments that will empower your R&D and supply chain teams to make data-driven decisions for your upcoming projects.
