Advanced Cu-Catalyzed Synthesis of 5,6-Dihydropyrimidinone Derivatives for Commercial Scale-Up
Advanced Cu-Catalyzed Synthesis of 5,6-Dihydropyrimidinone Derivatives for Commercial Scale-Up
The pharmaceutical industry constantly seeks robust synthetic routes for heterocyclic scaffolds that possess significant biological activity. Patent CN103193715A introduces a highly efficient preparation method for 5,6-dihydropyrimidinone derivatives, a core structure found in numerous top-selling drugs such as Valtrex and Viagra. This technology leverages a monovalent copper-catalyzed cyclization between N-acyl substituted propargyl amines and sulfonyl azides. By operating under mild conditions ranging from 30 to 80 degrees Celsius in the presence of 4A molecular sieves, this method overcomes many limitations associated with classical heterocycle synthesis. For R&D directors and procurement specialists, understanding this pathway is critical for developing reliable pharmaceutical intermediate supplier networks that can deliver high-purity compounds with consistent quality.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the dihydropyrimidinone skeleton has relied on classical condensation reactions such as the Biginelli reaction or multi-component couplings involving beta-keto esters and ureas. These traditional pathways often suffer from significant drawbacks, including the requirement for harsh acidic conditions, prolonged reaction times, and difficult purification processes due to the formation of complex byproduct mixtures. Furthermore, solid-phase synthesis methods, while effective for library generation, are often prohibitively expensive and operationally cumbersome for large-scale manufacturing. The reliance on specialized reagents and the generation of substantial chemical waste in these older protocols create bottlenecks in cost reduction in API manufacturing, making them less attractive for modern, green chemistry-focused production facilities.
The Novel Approach
In stark contrast, the methodology disclosed in CN103193715A offers a streamlined, one-pot solution that drastically simplifies the synthetic workflow. By utilizing readily available N-acyl propargyl amines and sulfonyl azides, the process eliminates the need for pre-functionalized complex starting materials. The reaction proceeds smoothly in common organic solvents such as acetonitrile, dichloromethane, or toluene, without the need for extreme temperatures or pressures. This novel approach not only enhances the versatility of the reaction scope, allowing for various substituents on the aromatic rings, but also ensures that the feeding and post-processing steps are exceptionally simple. This operational simplicity translates directly into reduced labor costs and faster turnaround times for pilot plant operations.
Mechanistic Insights into Cu-Catalyzed Cyclization
The core of this innovation lies in the copper(I)-catalyzed interaction between the alkyne moiety of the propargyl amine and the azide group of the sulfonyl azide. This transformation is mechanistically related to the Copper-Catalyzed Azide-Alkyne Cycloaddition (CuAAC), but with a specific intramolecular cyclization trajectory that forms the six-membered dihydropyrimidinone ring. The monovalent copper species, which can be introduced as cuprous iodide, bromide, chloride, or triflate, activates the terminal alkyne, facilitating the nucleophilic attack by the azide. The presence of a base, such as pyridine, triethylamine, or potassium carbonate, is essential to neutralize the acidic byproducts and maintain the catalytic cycle. Crucially, the inclusion of 4A molecular sieves serves as a desiccant to remove trace water, which could otherwise hydrolyze the sensitive sulfonyl azide or deactivate the copper catalyst, thereby ensuring high conversion rates and minimizing impurity profiles.
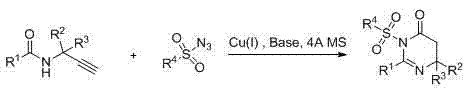
From an impurity control perspective, this mechanism offers distinct advantages over acid-catalyzed condensations. The specificity of the copper catalyst directs the reaction towards the desired heterocyclic product, reducing the formation of polymeric tars or regioisomers often seen in thermal cycloadditions. The mild temperature range of 30 to 80 degrees Celsius further suppresses thermal decomposition of the reactants. For quality control teams, this means the crude reaction mixture is cleaner, requiring less aggressive purification steps. The ability to tune the electronic properties of the substrates—by selecting different R groups on the acyl or sulfonyl moieties—allows for fine-tuning the reaction kinetics, ensuring that even sterically hindered substrates can be converted efficiently without compromising the structural integrity of the final high-purity OLED material or pharmaceutical intermediate.
How to Synthesize 5,6-Dihydropyrimidinone Efficiently
Implementing this synthesis requires strict adherence to the optimized parameters outlined in the patent to ensure reproducibility and safety. The process begins with the preparation of the sulfonyl azide, typically generated in situ or separately from sulfonyl chlorides and sodium azide, followed by the main cyclization step. Operators must maintain an inert atmosphere using nitrogen or argon to prevent oxidation of the copper catalyst. The molar ratios are critical, with a typical stoichiometry of 1:1 for the amine and azide, 0.1 equivalents of copper catalyst, and 1 to 3 equivalents of base. Detailed standardized synthesis steps see the guide below for precise execution protocols that guarantee batch-to-batch consistency.
- Prepare reactants: Mix N-acyl substituted propargyl amine and sulfonyl azide in a 1:1 molar ratio under inert gas.
- Add catalyst system: Introduce monovalent Cu catalyst (0.1 eq), base (1-3 eq), and 4A molecular sieves to the organic solvent.
- Reaction and Purification: Stir at 30-80°C for 1-18 hours, then purify via acid wash, extraction, and column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this technology represents a strategic opportunity to optimize the sourcing of critical heterocyclic building blocks. The primary advantage lies in the accessibility of the raw materials; N-acyl propargyl amines and sulfonyl chlorides are commodity chemicals available from multiple global vendors, reducing the risk of single-source dependency. This abundance ensures enhanced supply chain reliability, as disruptions in the availability of one precursor can be easily mitigated by switching suppliers without reformulating the entire process. Furthermore, the mild reaction conditions imply lower energy consumption compared to high-temperature reflux methods, contributing to substantial cost savings in utility expenditures over the lifecycle of the product.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts beyond simple copper salts, combined with the avoidance of specialized solid-phase resins, leads to a significantly reduced bill of materials. The simplified workup procedure, which involves standard aqueous washing and extraction rather than complex distillation or preparative HPLC, lowers the operational expenditure per kilogram. By removing the need for harsh reagents and extreme conditions, the facility maintenance costs are also minimized, as there is less corrosion and wear on the reactor vessels, resulting in long-term financial efficiency for the manufacturing partner.
- Enhanced Supply Chain Reliability: The robustness of this chemical process allows for flexible production scheduling. Since the reaction tolerates a variety of solvents including dichloromethane, toluene, and acetonitrile, production can continue even if there are temporary shortages of a specific solvent grade. The short reaction times, ranging from 1 to 18 hours depending on the substrate, enable higher throughput in existing reactor farms. This agility ensures reducing lead time for high-purity pharmaceutical intermediates, allowing downstream drug manufacturers to accelerate their own clinical trial timelines and market entry strategies without being bottlenecked by intermediate supply delays.
- Scalability and Environmental Compliance: Scaling this reaction from gram to ton scale is straightforward due to the homogeneous nature of the catalytic system and the absence of exothermic runaway risks associated with strong acid catalysis. The use of 4A molecular sieves, which can be filtered and potentially regenerated, aligns with green chemistry principles by reducing liquid waste volume. The process generates minimal hazardous byproducts, simplifying the wastewater treatment requirements and ensuring compliance with increasingly stringent environmental regulations. This environmental compatibility makes it an ideal candidate for commercial scale-up of complex polymer additives or agrochemical intermediates in regions with strict emission controls.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this synthetic route. These insights are derived directly from the experimental data and background analysis provided in the patent documentation, aiming to clarify the practical aspects of adopting this technology for industrial applications. Understanding these nuances is vital for process chemists who are evaluating the feasibility of integrating this method into their existing production pipelines.
Q: What are the advantages of this Cu-catalyzed method over traditional Biginelli reactions?
A: Unlike traditional methods that often require harsh acidic conditions or multi-step solid-phase synthesis, this patent describes a one-pot reaction under mild temperatures (30-80°C) with readily available starting materials, significantly simplifying the operational complexity.
Q: Why are 4A molecular sieves critical in this synthesis?
A: The 4A molecular sieves act as an efficient water scavenger. Since sulfonyl azides can be sensitive to moisture and water can inhibit the copper catalytic cycle or lead to side reactions, maintaining anhydrous conditions is crucial for maximizing yield and purity.
Q: Is this process suitable for large-scale pharmaceutical manufacturing?
A: Yes, the process utilizes common organic solvents like acetonitrile or dichloromethane and operates at moderate temperatures, making it highly scalable. The simple workup involving standard extraction and chromatography facilitates commercial scale-up of complex pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5,6-Dihydropyrimidinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the copper-catalyzed synthesis described in CN103193715A for the next generation of therapeutic agents. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions seamlessly from the laboratory bench to full-scale manufacturing. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 5,6-dihydropyrimidinone derivative meets the exacting standards required for clinical and commercial use.
We invite you to collaborate with our technical team to explore how this efficient synthetic route can be tailored to your specific molecule requirements. By engaging with us, you gain access to a Customized Cost-Saving Analysis that evaluates the economic benefits of switching to this copper-catalyzed protocol for your specific supply chain. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us demonstrate how we can become your trusted partner in delivering high-quality chemical solutions.
