Advanced Copper-Catalyzed Synthesis of 1,2-Bis(arylsulfonyl) Ethylene Derivatives for Commercial Scale
Advanced Copper-Catalyzed Synthesis of 1,2-Bis(arylsulfonyl) Ethylene Derivatives for Commercial Scale
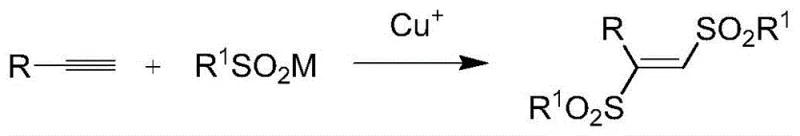
The landscape of organic synthesis for sulfone-containing molecules is undergoing a significant transformation driven by the need for greener, more efficient, and cost-effective manufacturing processes. A pivotal development in this field is documented in patent CN110590621B, which discloses a novel method for synthesizing 1,2-bis(arylsulfonyl) ethylene derivatives via copper-catalyzed terminal alkyne sulfonylation. This technology represents a substantial leap forward from classical approaches, eliminating the reliance on harsh oxidants and difficult-to-source precursors. For R&D directors and procurement managers in the fine chemical and pharmaceutical sectors, this innovation offers a robust pathway to access high-purity intermediates essential for drug discovery and material science applications. The method leverages abundant terminal alkynes and sulfinate compounds, utilizing a monovalent copper catalyst to drive the reaction under remarkably mild conditions, thereby reducing both operational complexity and environmental impact.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 1,2-bis(arylsulfonyl) ethylene derivatives has been plagued by significant synthetic hurdles that impede efficient commercial production. Traditional routes, such as those reported by Ottorio De Lucchi in 1984, often necessitated the reaction of 1,2-dichloroethylene with benzenethiol salts followed by a separate oxidation step, introducing multiple unit operations and potential yield losses. Other methodologies, like the nucleophilic vinyl substitution described by Masahito Ochiai in 1993, relied on specialized and expensive starting materials such as β-((phenylsulfonyl)alkenyl)iodonium tetrafluoroborate, which are not readily available on the bulk chemical market. Furthermore, many prior art processes required the use of strong oxidants to convert sulfide intermediates into the desired sulfones, creating safety hazards and generating toxic waste streams that complicate regulatory compliance. The necessity for inert atmospheres in several of these legacy methods further escalated capital expenditure requirements for reactor systems, making them less attractive for large-scale manufacturing where cost containment is paramount.
The Novel Approach
In stark contrast to these cumbersome legacy techniques, the methodology disclosed in CN110590621B introduces a streamlined, one-pot synthesis that directly couples terminal alkynes with sulfinate compounds. This approach fundamentally simplifies the supply chain by utilizing terminal alkynes, which are commercially ubiquitous and structurally diverse, allowing for the rapid generation of a wide library of derivatives. The reaction proceeds efficiently at temperatures not less than 60°C, typically optimized around 100°C, without the need for an inert atmosphere, significantly lowering the barrier to entry for production facilities. By employing a cuprous salt catalyst in conjunction with specific additives like ethyl difluorobromoacetate, the process achieves high conversion rates while avoiding the formation of toxic by-products associated with oxidative methods. This shift not only enhances the safety profile of the manufacturing process but also drastically reduces the downstream purification burden, resulting in a more economically viable route for producing high-value sulfone intermediates.
Mechanistic Insights into Copper-Catalyzed Terminal Alkyne Sulfonylation
The core of this technological breakthrough lies in the unique catalytic cycle facilitated by monovalent copper species in the presence of fluorinated additives. The reaction initiates with the activation of the terminal alkyne by the cuprous catalyst, forming a reactive copper-acetylide intermediate that is poised for subsequent functionalization. The presence of additives such as ethyl difluorobromoacetate plays a critical role in modulating the electronic environment of the reaction mixture, likely facilitating the generation of sulfonyl radicals from the sodium sulfinate precursor. These radicals then engage in a selective addition across the activated alkyne triple bond, constructing the carbon-sulfur bonds necessary for the bis-sulfonyl architecture. The precise control over regioselectivity and stereoselectivity is achieved through the careful tuning of reaction parameters, ensuring that the thermodynamically stable E-isomer or specific Z-isomer is favored depending on the substrate electronics.
Furthermore, the mechanism inherently suppresses the formation of common impurities such as over-oxidized sulfones or polymerized alkyne by-products, which are frequent challenges in radical sulfonylation chemistries. The copper catalyst acts not only as an initiator but also as a regulator, ensuring that the radical flux remains controlled throughout the reaction duration. This controlled radical pathway allows for the tolerance of various functional groups on both the alkyne and the sulfinate components, including electron-donating and electron-withdrawing substituents on aryl rings. The ability to accommodate heteroaryl and alkyl groups without significant degradation of yield underscores the robustness of the catalytic system. For process chemists, understanding this mechanistic nuance is vital for troubleshooting and optimizing the reaction for specific target molecules, ensuring that the final product meets the stringent purity specifications required for pharmaceutical applications.
How to Synthesize 1,2-Bis(arylsulfonyl) Ethylene Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires adherence to specific protocol parameters to maximize yield and reproducibility. The general procedure involves dissolving the terminal alkyne and sodium sulfinate compound in a polar aprotic solvent, with acetonitrile demonstrating superior performance compared to alternatives like DMF or dichloroethane. The addition of the cuprous iodide catalyst and the difluoromethylene additive must be timed correctly to ensure homogeneous mixing before the application of heat. As demonstrated in the patent examples, maintaining the reaction temperature at approximately 100°C for a duration of one hour is sufficient to drive the conversion to completion, as monitored by TLC. The workup procedure is straightforward, involving aqueous quenching, organic extraction, and standard silica gel chromatography, which facilitates the isolation of the pure product without complex crystallization steps.
- Combine terminal alkyne, sodium sulfinate compound, and a difluoromethylene additive (e.g., ethyl difluorobromoacetate) in a suitable solvent like acetonitrile.
- Add a cuprous salt catalyst, such as cuprous iodide (CuI), to the reaction mixture at a concentration of approximately 20% relative to raw materials.
- Heat the reaction mixture to a temperature between 60°C and 120°C, preferably around 100°C, and stir for approximately 1 hour until completion.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing and supply chain perspective, the adoption of this copper-catalyzed methodology offers profound advantages that translate directly into bottom-line improvements and operational resilience. The elimination of strong oxidants and the avoidance of inert atmosphere requirements significantly reduce the complexity of the manufacturing infrastructure, allowing for production in standard glass-lined or stainless steel reactors without specialized gas handling systems. This simplification leads to a substantial reduction in capital expenditure and operational overhead, making the process highly attractive for contract manufacturing organizations aiming to optimize their asset utilization. Moreover, the reliance on commodity chemicals like terminal alkynes and sodium sulfinates ensures a stable and diversified supply base, mitigating the risks associated with single-source dependencies on exotic reagents.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the simplification of the synthetic route and the use of inexpensive, widely available raw materials. By condensing the synthesis into a single step that avoids costly oxidation reagents and multi-step sequences, the overall cost of goods sold is significantly lowered. The high atom economy of the reaction minimizes waste generation, which in turn reduces the costs associated with waste disposal and environmental compliance. Additionally, the mild reaction conditions decrease energy consumption compared to high-temperature or cryogenic processes, contributing to further operational savings. The ability to achieve high yields with minimal purification steps means that solvent usage and processing time are also reduced, compounding the financial advantages for large-scale production runs.
- Enhanced Supply Chain Reliability: The use of terminal alkynes and sodium sulfinates as starting materials greatly enhances supply chain security, as these compounds are produced by numerous global suppliers and are not subject to the same geopolitical or logistical bottlenecks as specialized organometallic reagents. This abundance ensures that production schedules can be maintained consistently without the risk of delays caused by raw material shortages. The robustness of the reaction conditions also means that the process is less sensitive to minor variations in reagent quality or environmental factors, leading to more predictable batch outcomes and reliable delivery timelines for customers. For procurement managers, this reliability translates into reduced safety stock requirements and improved cash flow management.
- Scalability and Environmental Compliance: Scaling this chemistry from gram to ton scale is facilitated by the absence of hazardous oxidants and the use of standard solvents like acetonitrile, which are well-understood in industrial settings. The process generates fewer toxic by-products, aligning with increasingly stringent global environmental regulations and corporate sustainability goals. The simplified workup procedure reduces the volume of wastewater and solid waste generated per kilogram of product, easing the burden on effluent treatment plants. This environmental compatibility not only future-proofs the manufacturing process against tightening regulations but also enhances the brand reputation of companies adopting green chemistry principles in their supply chains.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this copper-catalyzed sulfonylation technology. These answers are derived directly from the experimental data and technical disclosures within the patent documentation, providing a reliable foundation for decision-making. Understanding these nuances is critical for R&D teams evaluating the feasibility of this route for their specific pipeline candidates and for supply chain leaders assessing the long-term viability of the manufacturing process.
Q: What are the key advantages of this copper-catalyzed method over traditional oxidation routes?
A: Unlike traditional methods requiring strong oxidants, inert atmospheres, or hard-to-obtain precursors like alkynyl selenium salts, this patent-disclosed method utilizes readily available terminal alkynes and sodium sulfinates under mild, non-inert conditions with high atom economy.
Q: Can this synthesis be scaled for industrial production of pharmaceutical intermediates?
A: Yes, the process operates at moderate temperatures (60-120°C) without the need for specialized inert gas equipment or hazardous strong oxidants, making it highly suitable for large-scale commercial manufacturing with simplified safety protocols.
Q: What represents the optimal catalyst and additive system for maximum yield?
A: Experimental data indicates that using cuprous iodide (CuI) as the catalyst in acetonitrile solvent, combined with ethyl difluorobromoacetate as the additive, provides superior conversion rates and yields up to 85% compared to other copper salts or solvents.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,2-Bis(arylsulfonyl) Ethylene Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the copper-catalyzed synthesis method disclosed in CN110590621B for the production of high-value sulfone intermediates. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive consistent quality regardless of order volume. Our state-of-the-art facilities are equipped to handle the specific requirements of this chemistry, including precise temperature control and efficient solvent recovery systems, all while adhering to stringent purity specifications and rigorous QC labs to guarantee product integrity. We are committed to leveraging this advanced technology to deliver cost-effective solutions that accelerate our partners' drug development timelines.
We invite you to collaborate with us to explore how this innovative synthesis route can be tailored to your specific project needs. Our technical team is ready to provide a Customized Cost-Saving Analysis that quantifies the potential efficiencies of switching to this copper-catalyzed method for your target molecules. Please contact our technical procurement team today to request specific COA data for our existing sulfone inventory or to discuss route feasibility assessments for your custom synthesis projects, ensuring a seamless transition to a more efficient and sustainable supply chain.
