Revolutionizing 2-Arylsulfonyl Quinoline Production: A Green Ultrasound-Assisted Aqueous Strategy for Commercial Scale-Up
Revolutionizing 2-Arylsulfonyl Quinoline Production: A Green Ultrasound-Assisted Aqueous Strategy for Commercial Scale-Up
The landscape of pharmaceutical intermediate manufacturing is undergoing a significant transformation driven by the urgent need for greener, more cost-effective synthetic methodologies. A pivotal advancement in this domain is detailed in Chinese Patent CN109096186B, which discloses a novel synthesis method for 2-arylsulfonyl quinoline derivatives. This technology represents a paradigm shift from traditional organometallic or high-temperature organic solvent-based protocols to a sustainable, ultrasound-assisted aqueous system. By leveraging the unique physical effects of cavitation generated by ultrasonic waves, this process enables the efficient coupling of 2-haloquinolines with arylsulfonyl chlorides in the presence of sulfites. For R&D directors and procurement managers seeking reliable pharmaceutical intermediate suppliers, this innovation offers a compelling value proposition: it drastically reduces reaction times from days to minutes while eliminating the need for toxic organic solvents, thereby aligning perfectly with modern environmental compliance standards and cost-reduction imperatives in API manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2-arylsulfonyl quinolines has been plagued by inefficient and environmentally burdensome protocols. As illustrated in prior art, such as the method reported by Merck researchers in 2011, the standard approach involved reacting 2-chloroquinoline with sodium p-toluenesulfinate in dimethylacetamide (DMAC) at 100°C for extended periods, often up to 24 hours. 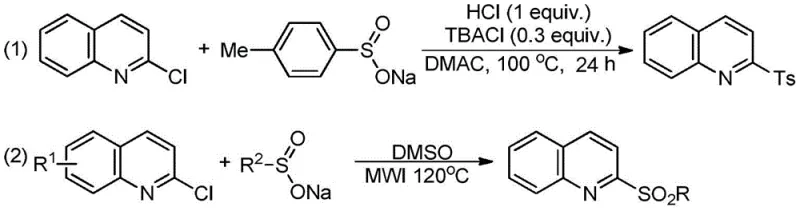 This legacy methodology suffers from multiple critical drawbacks that hinder commercial scalability. Firstly, the requirement for stoichiometric amounts of expensive phase-transfer catalysts like tetrabutylammonium chloride and strong acids increases the raw material bill of materials significantly. Secondly, the use of high-boiling polar aprotic solvents like DMAC creates severe downstream processing challenges, necessitating energy-intensive distillation for solvent recovery and generating hazardous waste streams. Furthermore, the pre-synthesis of sodium aryl sulfinates from arylsulfonyl chlorides adds an extra operational step, increasing labor costs and reducing overall atom economy. These factors collectively result in a process that is not only economically suboptimal but also difficult to scale safely in large-scale chemical reactors due to thermal hazards and solvent toxicity.
This legacy methodology suffers from multiple critical drawbacks that hinder commercial scalability. Firstly, the requirement for stoichiometric amounts of expensive phase-transfer catalysts like tetrabutylammonium chloride and strong acids increases the raw material bill of materials significantly. Secondly, the use of high-boiling polar aprotic solvents like DMAC creates severe downstream processing challenges, necessitating energy-intensive distillation for solvent recovery and generating hazardous waste streams. Furthermore, the pre-synthesis of sodium aryl sulfinates from arylsulfonyl chlorides adds an extra operational step, increasing labor costs and reducing overall atom economy. These factors collectively result in a process that is not only economically suboptimal but also difficult to scale safely in large-scale chemical reactors due to thermal hazards and solvent toxicity.
The Novel Approach
In stark contrast, the technology disclosed in CN109096186B introduces a streamlined, one-pot tandem reduction-coupling strategy that operates under remarkably mild conditions. This innovative route utilizes readily available arylsulfonyl chlorides directly, bypassing the need for pre-formed sulfinate salts. The reaction proceeds in pure water, a non-toxic, non-flammable, and essentially free solvent, which fundamentally alters the safety profile of the manufacturing process. By employing ultrasound irradiation at frequencies between 80KHz and 160KHz, the reaction kinetics are dramatically accelerated, allowing for completion within 10 to 30 minutes at ambient temperature. This rapid conversion is crucial because it effectively outcompetes the hydrolysis of the arylsulfonyl chloride, a side reaction that typically plagues aqueous chemistry. The result is a highly selective transformation that delivers the target 2-arylsulfonyl quinoline derivatives with exceptional yields, often exceeding 98%, and simplifies isolation to a mere filtration step, representing a substantial leap forward in process chemistry efficiency.
Mechanistic Insights into Ultrasound-Assisted Tandem Reduction-Coupling
The success of this methodology relies on the synergistic interplay between ultrasonic cavitation and the specific redox properties of sulfites in an aqueous medium. Under ultrasound irradiation, the formation and collapse of microbubbles generate localized hot spots with extreme temperatures and pressures, along with intense shear forces. These physical phenomena enhance mass transfer between the organic substrates and the aqueous phase, effectively creating a pseudo-homogeneous reaction environment without the need for organic co-solvents. Mechanistically, the sulfite ion (SO3 2-) acts as a mild reducing agent, converting the arylsulfonyl chloride into a reactive arylsulfinic acid or sulfinate species in situ. This transient intermediate then immediately undergoes nucleophilic substitution with the 2-haloquinoline substrate. 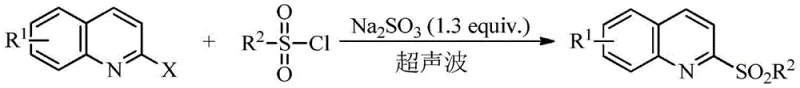 The rapid generation and consumption of the sulfinate species prevent its accumulation and subsequent degradation, ensuring high regioselectivity at the 2-position of the quinoline ring. This mechanistic pathway avoids the formation of complex by-products often seen in transition-metal catalyzed couplings, resulting in a cleaner impurity profile that is easier to control during quality assurance testing.
The rapid generation and consumption of the sulfinate species prevent its accumulation and subsequent degradation, ensuring high regioselectivity at the 2-position of the quinoline ring. This mechanistic pathway avoids the formation of complex by-products often seen in transition-metal catalyzed couplings, resulting in a cleaner impurity profile that is easier to control during quality assurance testing.
From an impurity control perspective, the aqueous nature of the reaction provides an inherent purification advantage. Most organic impurities and unreacted starting materials either remain dissolved in the aqueous phase or are removed during the ethanol wash step, while the product precipitates due to its low water solubility. This "crystallization-driven" isolation ensures that the final product achieves a purity greater than 98% without the need for column chromatography. For R&D teams, this means a robust process with a wide operating window where minor fluctuations in temperature or mixing speed do not significantly impact the impurity spectrum. The absence of transition metals also eliminates the risk of heavy metal contamination, a critical specification for pharmaceutical intermediates intended for eventual API synthesis, thereby reducing the burden on analytical QC labs to perform rigorous metal residue testing.
How to Synthesize 2-Arylsulfonyl Quinoline Efficiently
Implementing this synthesis route in a laboratory or pilot plant setting requires precise control over ultrasonic parameters to maximize yield and minimize side reactions. The process is designed to be operationally simple, involving the direct charging of solid reagents into water followed by the application of sonic energy. Detailed standardized operating procedures regarding equipment setup and safety protocols are essential for reproducibility.
- Charge a reaction vessel with 2-haloquinoline, arylsulfonyl chloride, and sodium sulfite in an aqueous medium.
- Apply ultrasonic irradiation (e.g., 60W, 140KHz) at ambient temperature for 10 to 30 minutes to facilitate the coupling.
- Filter the resulting precipitate directly and wash with ethanol to obtain the high-purity product without chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this ultrasound-assisted aqueous synthesis translates into tangible economic and logistical benefits that extend far beyond the laboratory bench. The elimination of expensive organic solvents like DMAC and DMSO removes a significant variable cost component, as well as the capital expenditure associated with solvent recovery systems and explosion-proof infrastructure. Moreover, the substitution of costly sodium aryl sulfinates with commodity-grade arylsulfonyl chlorides and sodium sulfite drastically lowers the raw material cost base. Since these reagents are widely available from bulk chemical suppliers, supply chain resilience is enhanced, reducing the risk of production stoppages due to specialty reagent shortages. The simplified workup procedure, which relies on filtration rather than extraction and distillation, significantly reduces cycle times, allowing for higher throughput in existing manufacturing facilities without the need for major equipment retrofitting.
- Cost Reduction in Manufacturing: The economic impact of this process is profound due to the removal of multiple cost drivers inherent in traditional methods. By avoiding the use of phase-transfer catalysts and expensive organic solvents, the direct material costs are significantly lowered. Additionally, the energy consumption is minimized because the reaction runs at ambient temperature rather than requiring prolonged heating at 100°C or microwave irradiation. The ability to isolate the product via simple filtration eliminates the need for energy-intensive solvent evaporation and chromatographic purification steps, leading to substantial operational expenditure savings. These cumulative efficiencies allow for a much more competitive pricing structure for high-purity pharmaceutical intermediates, providing a strategic advantage in cost-sensitive markets.
- Enhanced Supply Chain Reliability: Supply chain stability is bolstered by the use of universally available starting materials. Arylsulfonyl chlorides and sodium sulfite are commodity chemicals produced on a massive scale globally, ensuring consistent availability and price stability compared to specialized sulfinate salts. The robustness of the aqueous system also simplifies logistics, as there is no need to transport or store large volumes of flammable or toxic organic solvents, reducing regulatory compliance burdens and insurance costs. Furthermore, the short reaction time of 10 to 30 minutes enables rapid response to demand fluctuations, allowing manufacturers to operate on a just-in-time basis and reduce inventory holding costs for finished goods.
- Scalability and Environmental Compliance: Scaling this technology to industrial levels is straightforward due to the absence of thermal hazards and the use of water as the primary medium. The process aligns perfectly with green chemistry principles, generating minimal waste and avoiding the discharge of halogenated organic solvents into the environment. This environmental friendliness simplifies the permitting process for new manufacturing lines and reduces wastewater treatment costs. The high selectivity and yield ensure that raw materials are converted efficiently into product, maximizing atom economy and minimizing the volume of waste requiring disposal. For companies aiming to meet stringent ESG (Environmental, Social, and Governance) goals, this technology offers a clear pathway to sustainable manufacturing practices.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this ultrasound-assisted synthesis technology. These answers are derived directly from the experimental data and beneficial effects described in the patent literature, providing clarity on process capabilities and limitations. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this method into their existing production workflows.
Q: Why is water used as the solvent instead of organic solvents like DMAC?
A: Water is utilized to eliminate the environmental hazards and high costs associated with volatile organic solvents. Furthermore, the ultrasound acceleration allows the reaction to complete rapidly (10-30 mins), minimizing the hydrolysis of the arylsulfonyl chloride which typically occurs in aqueous environments over longer periods.
Q: How does this method improve upon the traditional Merck protocol?
A: Unlike the traditional method requiring 24 hours at 100°C in DMAC with expensive phase transfer catalysts, this ultrasound method operates at room temperature in just minutes. It also replaces costly sodium aryl sulfinates with cheaper arylsulfonyl chlorides, significantly lowering raw material expenses.
Q: Is the purification process complex for these derivatives?
A: No, the purification is exceptionally simple. Since the 2-arylsulfonyl quinoline products are insoluble in water, they precipitate out upon reaction completion. Simple filtration followed by an ethanol wash yields purity greater than 98%, avoiding energy-intensive distillation or column chromatography.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Arylsulfonyl Quinoline Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic technologies to maintain competitiveness in the global pharmaceutical market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovations like the ultrasound-assisted aqueous synthesis of 2-arylsulfonyl quinolines can be seamlessly transferred from the lab to the plant. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, guaranteeing that every batch conforms to the highest industry standards. Our capability to handle complex heterocyclic intermediates positions us as a strategic partner for clients seeking to optimize their supply chains with greener, more cost-effective solutions.
We invite you to collaborate with us to leverage this cutting-edge technology for your specific project needs. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your volume requirements. We encourage potential partners to contact us to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions based on hard data. Let us help you accelerate your development timelines and reduce manufacturing costs with our proven expertise in fine chemical synthesis.
